Abstract
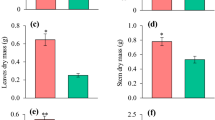
Micronutrients participate in various plant metabolic pathways, including the synthesis of carbohydrates, lignin, and phenols, which are necessary for plant growth and defense against pests and pathogens. In this study, we evaluated the effects of nutrient solutions deplete in B, Cu, or Zn on the nutrition, growth, and production of biochemical compounds in Eucalyptus urophylla cuttings. The experiment was carried out in a greenhouse for 90 days, with clonal cuttings of E. urophylla at four months of age. The treatments evaluated were complete fertilization (CF) and individual omissions of B, Cu, and Zn in the nutrient solution. The omission of Cu and Zn increased the concentration of carbohydrates in the leaves by 10% and reduced the rate of CO2 assimilation (A) by more than 30%. Cu omission decreased the lignification process compared to complete fertilization. Flavonoid production in plants with complete fertilization was 45–75% higher than that in plants maintained under B, Cu, or Zn deficiencies. Collectively, these results suggest that micronutrient deficiencies may reduce plant productivity as well as plant resistance to pests and pathogens.


Similar content being viewed by others
References
ABRAF (2010) Anuário estatístico da ABRAF 2010 ano base 2009/ ABRAF. – Brasília, p 140
Alvares CA, Sentelhas PC, Mattos EM, Miranda AC, Moraes WB, Silva PHM, Furtado EL, Stape JL (2017) Climatic favorability zones for Eucalyptus rust in Brazil. Forest Pathol 47:1–17
Battie-Laclau P, Laclau JP, Beri C, Mietton L, Muniz MRA, Arenque BC, Piccolo MC, Jordan-Meille L, Bouilet JP, Nouvellon V (2014) Photosynthetic and anatomical responses of Eucalyptus grandis leaves to potassium and sodium supply in a field experiment. Plant Cell Environ 37:70–81
Blount JW, Dixon RA, Paiva NL (1992) Stress responses in alfalfa (Medicago sativa L.) XVI. Antifungal activity of medicarpin and its biosynthetic precursors; implications for the genetic manipulation of stress metabolites. Physiol Mol Plant Pathol 41:333–349
Bonello P, Blodgett JT (2003) Pinus nigra–Sphaeropsis sapinea as a model pathosystem to investigate local and systemic effects of fungal infection of pines. Physiol Mol Plant Physiol 63:249–261
Chormova D, Fry SC (2016) Boron bridging of rhamnogalacturonan-II is promoted in vitro by cationic chaperones, including polyhistidine and wall glycoproteins. New Phytol 209:241–251
Chow PS, Landhausser SMA (2004) A method for routine measurements of total sugar and starch content in woody plant tissues. Tree Physiol 24:1129–1136
Dalcorso G, Manara A, Piasentina S, Furinia A (2014) Nutrient metal elements in plants. Metallomics 6(10):1770–1788
Dell B, Huang L (1997) Physiological responses of plants to low boron. Plant Soil 193:85–101
Dell B, Malajczuk N (1994) Boron deficiency in eucalypt plantations in China. Can J Res 24(12):2409–2416
Dell B, Robinson JM (1993) Symptoms of mineral nutrient deficiencies and the nutrient concentration ranges in seedlings of Eucalyptus maculata Hook. Plant Soil 155–156:255–261
Dell B, Xu D (1995) Diagnosis of zinc deficiency in seedlings of a tropical eucalypt (Eucalyptus urophylla S.T. Blake). Plant Soil 176:329–332
Dell B, Xu D, Rogers C, Huang L (2003) Micronutrient disorders in eucalypt plantations: causes, symptoms, identification, impact and management. In: Wei R, Xu D (eds) Eucalyptus plantations: research, management and development. World Scientific Publising, London, pp 241–252
Dell B, Hardy G, Burgess T (2008) Health and nutrition of plantation eucalypts in Asia. Southern Forests JForest Sci 70(2):131–138
dos Santos EF, S, Zanchim B J, Campos A G, Garrone, R F, Lavres, J, (2013) Photosynthesis rate, chlorophyll content and initial development of physic nut without micronutrient fertilization. R Bras Ci Solo 37:1334–1342
du Toit B, Smith CW, Little KM, Boreham G, Pallett RN (2010) Intensive, site-specific silviculture: Manipulating resource availability at establishment for improved stand productivity. A review of South African research. Forest Ecol Manag 259(9):1836–1845
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric methods for determination of sugars related substances. Anal Chem 28(3):350–356
Elmer WH, Datnoff LE (2014) Mineral nutrition and suppression of plant disease. In: van Alfen N (ed) Encyclopedia of agriculture and food systems, 4 edn. Elsevier, San Diego, pp 231–244
Epstein E, Bloom AJ (2005) Mineral nutrition of plants: principles and perspectives, 2, Sunderland Mass: Sinauer, p 400
Ferraz A, Mendonça R, Silva FT (2000) Organosolv delignification of white- and brown-rotted Eucalyptus grandis hardwood. J Chem Technol Biotechnol 75:18–24
Gonçalves JLM, Alvares CA, Higa RA, Silva LD, Alfenas AC, Stahl J, Ferraz SFB, Paula Lima W, Brancalion PHS, Hubner A, Bouillet JPD, Laclau JP, Nouvellon Y, Epron D (2013) Integrating genetic and silvicultural strategies to minimize abiotic and biotic constraints in Brazilian eucalypt plantations. For Ecol Manage 301:6–27
Graham RD (1980) The distribution of copper and soluble carbohydrates in wheat plants grown at high and low levels of copper supply. Zeitschrift Für Pflanzenernährung Und Bodenkunde 143(2):161–169
Hagen-Thorn A, Stjernquist I (2005) Micronutrient levels in some temperate European tree species: a comparative field study. Trees 19:572–579
Hajiboland R, Amirazad F (2010) Growth, photosynthesis and antioxidant defense system in Zn-deficient red cabbage plants. Plant, Soil Environ 56:209–217
Huber D, Römheld V, Weinmann M (2012) Relationship between nutrition, plant diseases and pests. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, San Diego, pp 283–298
IBA Relatório IBA (2017) [s.l: s.n.]. Disponível em:. https://iba.org/images/shared/Biblioteca/IBA_RelatorioAnual2017.pdf. Accessed 27 July 2020
Julkunen-Tiitto R (1985) Phenolic constituents in the leaves of northern willows: methods for the analysis of certain pehnolics. J Agric Food Chem 33(2):213–217
Kastori R, Plesnicar M, Pankovic D, Sakac Z (1995) Photosynthesis, chlorophyll flu- orescence and soluble carbohydrates in sunflower leaves as affected by boron deficiency. J Plant Nutr 18:1751–1763
Kim T, Wetzstein HY (2003) Cytological and ultrastructural evaluations of zinc deficiency in leaves. J Am Soc Hortic Sci 128:171–175
Kulbat K (2016) The role of phenolic compounds in plant resistance. Biotechnol Food Sci 80(2):97–108
Lehto T, Ruuhola T, Dell B (2010) Boron in forest trees and forest ecosystems. For Ecol Manage 260:2053–2069
Li JW, Richter DD, Mendoza A, Heine P (2008) Four-decade responses of soil trace elements to an aggrading old-field forest: B, Mn, Zn, Cu, and Fe. Ecology 89:2911–2923
Li DD, Li YJ, Liang J, Zhao CZ, Yin HJ, Yin CY, Cheng XY, Liu Q (2014) Responses of soil micronutrient availability to experimental warming in two constrasting forest ecosystems in the Eastern Tibetan Plateau, China. Journal of Soils and Sediments 14(6), 1050–1060. Ma D, Sun D, Wang C, Ding H, Qin H, Hou J, Huang X, Xie Y, Guo T (2017) Physiological responses and yield of wheat plants in zinc-mediated alleviation of drought stress. Front Plant Sci 8:1–12
Liakopoulos G, Karabourniotis G (2005) Boron deficiency and concentrations and composition of phenolic compounds in Olea europaea leaves: a combined growth chamber and field study. Tree Physiol 25(3):307–315
Mabry TJ, Markham KR, Thomas MB (1970) The Systematic Identification of the Flavonoids. Springer, New York, p 354
Malavolta E, Vitti GC, Oliveira SA (1997) Avaliação do estado nutricional das plantas. Princípios e aplicações. 2, POTAFOS, Piracicaba, p 319
Marschner H (2012) Mineral nutrition of higher plants. In: Marschner H, Marschner P (eds) Mineral nutrition of higher plants. Academic Press
Martín-Rejano EM, Camacho-Cristóbal JJ, Herrera-Rodríguez MB, Rexach J, Navarro-Gochicoa MT, González-Fontes A (2011) Auxin and ethylene are involved in the responses of root system architecture to low boron supply in Arabidopsis seedlings. Physiol Plant 142:170–178
Masullo LS, Ferraz AV, de Gonçalves JL, M, Camargo L E A, Ávila P A de, Drago J R, Esteves O, Dias C T dos S, (2020) Relationship between forest residue management and micronutrient fertilization with eucalyptus rust severity in Eucalyptus grandis. Forest Ecol Manag 475:118443
Mierziak J, Kostyn K, Kulma A (2014) Flavonoids as important molecules of plant interactions with the environment. Molecules 19:16240–16265
O’Neill MA, Ishii T, Albersheim P, Darvill AG (2004) Rhamnogalacturonan II: Structure and Function of a Borate Cross-Linked Cell Wall Pectic Polysaccharide. Annu Rev Plant Biol 55:109–139
Pandey N, Sharma CP (2000) Carbonic anhydrase activity and stomata morphology associated with zinc deficiency induced changes in faba bean. Phytomorphology 50:261–265
Prasad D, Singh A, Singh KP, Bist S, Tewari A, Singh UP (2010) The role of phenolic compounds in disease resistance in geranium. Arch Phytopathol Plant Protect 43(7):615–623
Printz B, Lutts S, Hausman JF, Sergean K (2016) Copper trafficking in plants and its implication on cell wall dynamics. Front Plant Sci 7:1–16
Rocha JHT, Wenzel AVA, Melo EASC et al (2019) Responses of coppiced Eucalyptus to macro- and micronutrient application. New for 50:717–731
Santos RM, Fortes GAC, Ferri PH, Santos CS (2011) Influence of foliar nutrients on phenol level in leaves of Eugenia uniflora. Brazilian J Pharmacogn 21:581–586
Sasaki H, Hirose T, Watanabe Y, Ohsugi R (1998) Carbonic anhydrase activity and CO2-transfer resistance in Zn-deficient rice leaves. Plant Physiol 118(3):929–934
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Sekimoto H, Hoshi M, Nomura T, Yokota T (1997) Zinc deficiency affects the levels of endogenous gibberellins in Zea mays L. Plant Cell Physiol 38:1087–1090
Sequeira CH, Barros NF, Neves JCL, Novais RF, Silva IR, Alley M (2011) Micronutrient soil-test levels and eucalyptus foliar contents. Commun Soil Sci Plant Anal 42(4):475–488
Seth K, Aery NC (2014) Effect of boron on the contents of chlorophyll, carotenoid, phenol and soluble leaf protein in mung Bean, Vigna radiata (L) Wilczek. Proc. Natl. Acad Sci India Sect B Biol Sci 84:713–719
Sharma CP (2006) Plant micronutrients. Science Publishers, Enfield, p 265
Shi C, Wang J, Hu R, Zhou G, O’Neill MA, Kong Y (2017) Boron-bridged RG-II and calcium are required to maintain the pectin network of the Arabidopsis seed mucilage ultrastructure. Plant Mol Biol 94(3):267–280
Silveira RLVA, Higashi EN (2003) Aspectos nutricionais envolvidos na ocorrência de doenças com ênfase para o eucalipto. Circular Técnica IPEF 200:1–13
Silveira RLVA, Higashi EN, Gonçalves NA, Moreira A (2005) Evaluation of the nutritional status of eucalypts: visual and foliar diagnoses and their interpretation. In: Gongalves JLM, Benedetti V (eds) Forest nutrition and fertilization. IPEF, Piracicaba/BR, pp 85–111
Smith AH, Gill WM, Pinkard EA, Mohammed CL (2007) Anatomical and histochemical defense responses induced in juvenile leaves of Eucalyptus globulus and Eucalyptus nitens by Mycosphaerella infection. For Pathol 37:361–373
Tavares GA, Bendassolli JA, Souza G, Nolasco FR (2004) Implantação de uma estação de produção de água desionizada para uso nos laboratórios do Cena / Usp empregando resinas de troca-iônica. Revista Analytica 10(7):36–42
Thomas G, Andresen E, Mattusch J, Hubáček T, Küpper H (2016) Deficiency and toxicity of nanomolar copper in low irradiance - A physiological and metalloproteomic study in the aquatic plant Ceratophyllum demersum. Aquat Toxicol 177:226–236
Turnbull CRA, Beadle CL, West PW, Crome RN (1994) Copper deficiency a probable cause of stem deformity in fertilised Eucalyptus nitens. Can J for Res 24(7):1434–1439
Wang N, Yang C, Pan Z, Liu Y, Peng S (2015) Boron deficiency in woody plants: various responses and tolerance mechanisms. Front Plant Sci 6:1–14
Waterman PG (1992) Roles for secondary metabolites in plants. Ciba Found Symp 171:255–269
Whitham FH, Blaydes DF, Devlin RM (1971) Experiments in plant physiology. Van Nostrand Reinhold, New York, p 254p
Wimmer MA, Abreu I, Bell RW, Bienert MD, Brown PH, Dell B, Fujiwara T, Goldbach HE, Lehto T, Mock HP, von Wirén N, Bassil E, Bienert GP (2019) Boron: an essential element for vascular plants: a comment on Lewis (2019) “Boron: the essential element for vascular plants that never was.” New Phytol 1232–1237
Funding
This study received support from São Paulo Research Foundation—FAPESP (Process: 2018/07294–3), The Brazilian National Council for Scientific and Technological Development (CNPq) and Silviculture and Management Thematic Program (PTSM-IPEF).
Author information
Authors and Affiliations
Contributions
LSM, AVF and JLMG designed this research. LSM, VDD, GRM, PAA and ALF performed this research. LSM, VDD and JHTR analyzed the data. JLMG, AVF, CLA, JL and JHTR revised this draft by rewriting, discussion and commenting. All authors contributed in the same way to the preparation of all the parts of this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Masullo, L.S., Derisso, V.D., Manarim, G.R. et al. Modulation of structural carbohydrates, phenol compounds and lignin content in Eucalyptus urophylla cuttings grown under boron, copper and zinc induced-deficiency. New Forests 53, 337–352 (2022). https://doi.org/10.1007/s11056-021-09859-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-021-09859-w