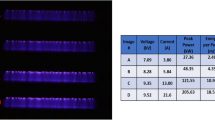
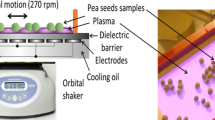
Abstract
Effects of a cold atmospheric pressure plasma (CAPP) treatment on the germination, production of biomass, vigor of seedlings, uptake of water of wheat seeds (Triticum aestivum L. cv. Eva) were investigated. The CAPP treatment influence on the inactivation of microorganisms occurring on the surface of wheat seeds was investigated also. The so-called Diffuse Coplanar Surface Barrier Discharge generating a cold plasma in ambient air with high power volume density of some 100 W/cm3 was used for the treatment of seeds at exposure times in the range of 10–600 s. The optical emission spectroscopy and the electrical measurements were used for estimation of CAPP parameters. The obtained results indicate that the germination rate, dry weight and vigor of seedlings significantly increased for plasma treatment from 20 to 50 s. The plasma treatment of seeds led to an extensive increase in wettability and faster germination comparing with the untreated seeds. The growth inhibition effect of CAPP on the surface microflora of wheat seeds increased with the increase of the treatment time. The efficiency of the treatment of wheat seeds artificially contaminated with pure cultures of filamentous fungi decreased in the following order: Fusarium nivale > F. culmorum > Trichothecium roseum > Aspergillus flavus > A. clavatus.










Similar content being viewed by others
References
Roth JR (2001) Industrial plasma engineering: applications to nonthermal plasma processing, vol 2. IOP Publishing Ltd., London
Laroussi M (2005) Low temperature plasma-based sterilization : overview and state-of-the-art. Plasma Processes Polym 2:391–400. doi:10.1002/ppap.200400078
Ben Gadri R, Roth JR, Montie TC et al (2000) Sterilization and plasma processing of room temperature surfaces with a one atmosphere uniform glow discharge plasma (OAUGDP). Surf Coat Technol 131:528–541. doi:10.1016/S0257-8972(00)00803-3
Morfill GE, Kong MG, Zimmermann JL (2009) Focus on plasma medicine. N J Phys 11:115011
Laroussi M, Leipold F (2004) Evaluation of the roles of reactive species, heat, and UV radiation in the inactivation of bacterial cells by air plasmas at atmospheric pressure. Int J Mass Spectrom 233:81–86. doi:10.1016/j.ijms.2003.11.016
Machala Z, Chládeková L, Pelach M (2010) Plasma agents in bio-decontamination by dc discharges in atmospheric air. J Phys D Appl Phys 43:222001
Lee K, Joo B, Hee D et al (2005) Sterilization of Escherichia coli and MRSA using microwave-induced argon plasma at atmospheric pressure. Surf Coat Technol 193:35–38. doi:10.1016/j.surfcoat.2004.07.034
Moisan M, Barbeau J, Moreau S et al (2001) Low-temperature sterilization using gas plasmas: a review of the experiments and an analysis of the inactivation mechanisms. Int J Pharm 226:1–21. doi:10.1016/S0378-5173(01)00752-9
Basaran P, Basaran-Akgul N, Oksuz L (2008) Elimination of Aspergillus parasiticus from nut surface with low pressure cold plasma (LPCP) treatment. Food Microbiol 25:626–632. doi:10.1016/j.fm.2007.12.005
Dobrynin D, Fridman G, Friedman G, Fridman A (2009) Physical and biological mechanisms of direct plasma interaction with living tissue. N J Phys 11:115020. doi:10.1088/1367-2630/11/11/115020
Kostov KG, Rocha V, Koga-Ito CY et al (2010) Bacterial sterilization by a dielectric barrier discharge (DBD) in air. Surf Coat Technol 204:2954–2959. doi:10.1016/j.surfcoat.2010.01.052
Koval’ová Z, Tarabová K, Hensel K, Machala Z (2013) Decontamination of Streptococci biofilms and Bacillus cereus spores on plastic surfaces with DC and pulsed corona discharges. Eur Phys J Appl Phys 61:24306. doi:10.1051/epjap/2012120449
Sinclair JB (1993) Control of seedborne pathogens and diseases of soybean seeds and seedlings. Pestic Sci 37:15–19. doi:10.1002/ps.2780370104
Michalíková A, Roháčik T, Kulichova R (1995) Efficacy of Vitavax 200 FF against diseases of spring barley caused by helminthosporioses. Agriculture 41:518–529
Oehrle NW, Karr DB, Kremer RJ, Emerich DW (2000) Enhanced attachment of Bradyrhizobium japonicum to soybean through reduced root colonization of internally seedborne microorganisms. Can J Microbiol 46:600–606. doi:10.1139/w00-030
Henselová M, Hudecová D (2001) Differences in the microflora of scarified and unscarified seeds of Karwinskia humboldtiana (Rhamnaceae). Folia Microbiol 46:543–548
Pietruszewski S (1996) Effects of magnetic biostimulation of wheat seeds on germination, yield and proteins. Int Agrophys 10:51–55
Volin JC, Denes FS, Young RA, Park SMT (2000) Modification of seed germination performance through cold plasma chemistry technology. Crop Sci 40:1706–1718
Meiqiang Yin, Mingjing Huang, Ma Buzhou MT (2005) Stimulating effects of seed treatment by magnetized plasma on tomato growth and yield. Plasma Sci Technol 7:3143
Marinković D, Borcean I (2009) Effect of cold electron plasma and extremely low frequency electron-magnetic field on wheat yield. Agric Sci 41:96–101
Lynikiene S, Pozeliene GR (2006) Influence of corona discharge field on seed viability and dynamics of germination. Int Agrophys 20:195–200
Borodin IF, Shcherbakov KN (1998) Electrophysical ways of stimulating plant growth. Mach Agric 5:35–36 (in Russian)
Palov I (2003) Research of influence of electromagnetic impact on maize seed and plants. Mach Agric 15:10–15 (in Bulgarian)
Dhayal M, Lee S, Park S (2006) Using low-pressure plasma for Carthamus tinctorium L. seed surface modification. Vacuum 80:499–506. doi:10.1016/j.vacuum.2005.06.008
Šerá B, Straňák V, Šerý M, Tichý M, Špatenka P (2008) Germination of Chenopodium albumin response to microwave plasma treatment. Plasma Sci Technol 10:506–511
Šerá B, Špatenka P, Šerý M et al (2010) Influence of plasma treatment on wheat and oat germination and early growth. IEEE Trans Plasma Sci 38:2963–2968
Živković S, Puač N, Giba Z et al (2004) The stimulatory effect of non-equilibrium (low temperature) air plasma pretreatment on light-induced germination of Paulownia tomentosa seeds. Seed Sci Technol 32:693–701
Dobrin D, Magureanu M, Mandache NB, Ionita M-D (2015) The influence of non-thermal plasma treatment on wheat germination. Innov Food Sci Emerg Technol 29:255–260. doi:10.1016/j.ifset.2015.02.006
Ksenz NV, Kaciesvili SV (2000) Electrostatic field and productivity of cereals. Mech Electrif Agric 6:18–19 (in Russian)
Huang H-H, Wang S-R (2008) The effects of inverter magnetic fields on early seed germination of mung beans. Bioelectromagnetics 29:649–657. doi:10.1002/bem.20432
Vashisth A, Nagarajan S (2008) Exposure of seeds to static magnetic field enhances germination and early growth characteristics in chickpea (Cicer arietinum L.). Bioelectromagnetics 29:571–578. doi:10.1002/bem.20426
Mitra A, Li Y-F, Klämpfl TG et al (2013) Inactivation of surface-borne microorganisms and increased germination of seed specimen by cold atmospheric plasma. Food Bioprocess Technol 7:645–653. doi:10.1007/s11947-013-1126-4
Jiang J, He X, Li L, Li J, Shao H, Xu Q, Ye R, Dong Y (2014) Effect of cold plasma treatment on seed germination and growth of wheat. Plasma Sci Technol 16:54–58
Deng X, Shi J, Kong MG (2006) Physical mechanisms of inactivation of Bacillus subtilis spores using cold atmospheric plasmas. IEEE Trans Plasma Sci 34:1310–1316. doi:10.1109/TPS.2006.877739
Jung H, Kim DB, Gweon B et al (2010) Enhanced inactivation of bacterial spores by atmospheric pressure plasma with catalyst TiO2. Appl Catal B 93:212–216. doi:10.1016/j.apcatb.2009.09.031
Ohkawa H, Akitsu T, Tsuji M, Kimura H (2006) Pulse-modulated, high-frequency plasma sterilization at atmospheric-pressure. Surf Coat Technol 200:5829–5835. doi:10.1016/j.surfcoat.2005.08.124
Gweon B, Kim DB, Moon SY, Choe W (2009) Escherichia coli deactivation study controlling the atmospheric pressure plasma discharge conditions. Curr Appl Phys 9:625–628. doi:10.1016/j.cap.2008.06.001
Selcuk M, Oksuz L, Basaran P (2008) Decontamination of grains and legumes infected with Aspergillus spp. and Penicillum spp. by cold plasma treatment. Bioresour Technol 99:5104–5109. doi:10.1016/j.biortech.2007.09.076
Arrus K, Blank G, Abramson D et al (2005) Aflatoxin production by Aspergillus flavus in Brazil nuts. J Stored Prod Res 41:513–527. doi:10.1016/j.jspr.2004.07.005
Yu MC, Yuan J-M (2004) Environmental factors and risk for hepatocellular carcinoma. Gastroenterology 127:S72–S78. doi:10.1016/j.gastro.2004.09.018
Park BJ, Takatori K, Sugita-Konishi Y et al (2007) Degradation of mycotoxins using microwave-induced argon plasma at atmospheric pressure. Surf Coat Technol 201:5733–5737. doi:10.1016/j.surfcoat.2006.07.092
Černák M, Kováčik D, Ráhel’ J et al (2011) Generation of a high-density highly non-equilibrium air plasma for high-speed large-area flat surface processing. Plasma Phys Control Fusion 53:124031. doi:10.1088/0741-3335/53/12/124031
Černák M, Černáková L, Hudec I et al (2009) Diffuse Coplanar Surface Barrier Discharge and its applications for in-line processing of low-added-value materials. Eur Phys J Appl Phys 47:22806. doi:10.1051/epjap/2009131
Šimor M, Ráhel’ J, Vojtek P et al (2002) Atmospheric-pressure diffuse coplanar surface discharge for surface treatments. Appl Phys Lett 81:2716. doi:10.1063/1.1513185
Homola T, Matoušek J, Medvecká V et al (2012) Atmospheric pressure diffuse plasma in ambient air for ITO surface cleaning. Appl Surf Sci 258:7135–7139. doi:10.1016/j.apsusc.2012.03.188
Navrátil Z, Trunec D, Šmíd R, Lazar L (2006) A software for optical emission spectroscopy: problem formulation and application to plasma diagnostics. Czech J Phys 56:944–951
Laux CO (2002) Radiation and nonequilibrium collisional-radiative models. In: Fletcher D, Charbonnier JM, Sarma GSR, Magin T (eds) von Karman Institute Lecture Series 2002–2007, Physico-chemical modeling of high enthalpy and plasma flows. Rhode-Saint-Genèse, Belgium
Abdul-Baki AA, Anderson JD (1973) Vigor determination in soybean seed by multiple criteria1. Crop Sci 13:630–633
Houghtby GB, Maturin LJ, Koenig EK, Wesser JW (1992) Microbial count methods. In: Marshall RT (ed) Standard methods for the examination of dairy products, 16th edn. American Public Health Association, Washington, DC, pp 213–216
Betina V, Baráthová H, Fargašová A et al (1987) Microbial laboratory methods. Alfa SNTL Publishing House, Bratislava (in Slovak)
Fassatiová O (1979) Moulds and filamentous fungi in technical microbiology. SNTL Publishing House, Praha (in Czech)
Hudecová D, Jantová S, Melník M, Uher M (1996) New azidometalkojates and their biological activity. Folia Microbiol 41:473–476. doi:10.1007/BF02814660
Puntner W (1981) Manual for field trials in plant protection. Ciba-Geigy Ltd, Basel
Rekanovic E, Potocnik I, Milijasevic-Marcic S et al (2010) Efficacy of seaweed concentrate from Ecklonia maxima (Osbeck) and conventional fungicides in the control of Verticillium wilt of pepper. Pesticidi i fitomedicina 25:319–324. doi:10.2298/PIF1004319R
Fischer G, Tausz M, Köck M, Grill D (2004) Effects of weak 16 3/2 Hz magnetic fields on growth parameters of young sunflower and wheat seedlings. Bioelectromagnetics 25:638–641. doi:10.1002/bem.20058
Weaver JC (1993) Electroporation: a general phenomenon for manipulating cells and tissues. J Cell Biochem 51:426–453
Muraji M, Asai T, Tatebe W (1998) Primary root growth rate of Zea mays seedlings grown in an alternating magnetic field of different frequencies. Bioelectrochem Bioenergy 44:271–273. doi:10.1016/S0302-4598(97)00079-2
Giba Z, Grubišic D, Konjevic R (2004) Nitric oxide signaling in higher plants. Studium Press, LLC, Houston
Kikuchi K, Koizumi M, Ishida N, Kano H (2006) Water uptake by dry beans observed by micro-magnetic resonance imaging. Ann Bot 98:545–553. doi:10.1093/aob/mcl145
Abebe AT, Modi AT (2009) Hydro-priming in dry bean (Phaseolus vulgaris L.). Res J Seed Sci 2:23–31. doi:10.3923/rjss.2009.23.31
Dubinov AE, Lazarenko EM, Selemir VD (2000) Effect of glow discharge air plasma on grain crops seed. IEEE Trans Plasma Sci 28:180–183
Henselová M, Slováková Ľ, Martinka M, Zahoranová A (2012) Growth, anatomy and enzyme activity changes in maize roots induced by treatment of seeds with low-temperature plasma. Biologia 67:490–497. doi:10.2478/s11756-012-0046-5
Stolárik T, Henselová M, Martinka M et al (2015) Effect of low-temperature plasma on the structure of seeds, growth and metabolism of endogenous phytohormones in pea (Pisum sativum L.). Plasma Chem Plasma Process. doi:10.1007/s11090-015-9627-8
Kobayashi DY, Palumbo JD (2000) Microbial endophytes. Marcel Dekker Inc., New York
Wachowska U, Stasiulewicz-Paluch AD, Głowacka K et al (2013) Response of epiphytes and endophytes isolated from winter wheat grain to biotechnological and fungicydal treatments. Pol J Environ Stud 22:267–273
Duan C, Wang X, Zhu Z, Wu X (2007) Testing of seedborne fungi in wheat germplasm conserved in the national crop genebank of China. Agric Sci China 6:682–687. doi:10.1016/S1671-2927(07)60100-X
Machala Z, Jedlovský I, Chládeková L et al (2009) DC discharges in atmospheric air for bio-decontamination: spectroscopic methods for mechanism identification. Eur Phys J D 54:195–204. doi:10.1140/epjd/e2009-00035-7
Sohbatzadeh F, Hosseinzadeh Colagar A, Mirzanejhad S, Mahmodi S (2010) E. coli, P. aeruginosa, and B. cereus bacteria sterilization using afterglow of non-thermal plasma at atmospheric pressure. Appl Biochem Biotechnol 160:1978–1984. doi:10.1007/s12010-009-8817-3
Suhem K, Matan N, Nisoa M, Matan N (2013) Inhibition of Aspergillus flavus on agar media and brown rice cereal bars using cold atmospheric plasma treatment. Int J Food Microbiol 161:107–111. doi:10.1016/j.ijfoodmicro.2012.12.002
Kim JE, Lee D-U, Min SC (2014) Microbial decontamination of red pepper powder by cold plasma. Food Microbiol 38:128–136. doi:10.1016/j.fm.2013.08.019
Acknowledgments
This study was supported by the Slovak Grant Agency for Science VEGA No. 1/0904/14. We wish to thank the Sedos, Krakovany in Slovakia for the samples of seeds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zahoranová, A., Henselová, M., Hudecová, D. et al. Effect of Cold Atmospheric Pressure Plasma on the Wheat Seedlings Vigor and on the Inactivation of Microorganisms on the Seeds Surface. Plasma Chem Plasma Process 36, 397–414 (2016). https://doi.org/10.1007/s11090-015-9684-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-015-9684-z