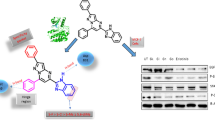
A series of benzimidazole-piperazine hybrids (14 – 37) were designed, synthesized and evaluated for their cytotoxic activity against human lung (A549) and breast (MCF-7) cancer cell lines. Preliminary evaluation revealed that most of these hybrid molecules (i.e., 16 – 25) exhibited noteworthy and preferential antiproliferative effect against human lung cancer (A549) with IC50 values of 2.8 – 7.8 μM. Among the synthesized molecules, compound 17 showed the most balanced cytotoxic effect against lung (A549) and breast (MCF-7) cancer cells with IC50 values of 5.4 and 4.2 μM, respectively. To explore the mechanistic aspects fundamental to the observed activity, further biological studies of compounds 16, 17 and 22 were carried out. In addition, these compounds induced PARP-1 cleavage and caspase 7 activation, caused morphological changes such as bleb formation in the treated cells, and significantly increased the nuclear fragmentation. Taken all together, our findings indicate that cytotoxic activities of newly synthesized benzimidazole-piperazine hybrids are mediated through the apoptotic cell death induction. These benzimidazole derivatives have the potential for further development as anticancer agents.





Similar content being viewed by others
References
Global Cancer Facts & Figures, 3rd Edition, Atlanta: American Cancer Society (2015).
R. S. Keri, A. Hiremathad, S. Budagumpi, et al., Chem. Biol. Drug Des., 86, 19 – 65 (2015).
P. Singla, V. Luxami, and K. Paul, Rsc. Adv., 4, 12422 – 12440 (2014).
G. Yadav and S. Ganguly, Eur. J. Med. Chem., 97, 419 – 443 (2015).
B. Caliskan, A. Yilmaz, I. Evren, et al., Med. Chem. Res., 22, 782 – 793 (2013).
B. D. Cheson and L. Leoni, Clin. Adv. Hematol. Oncol., 9, 1 – 11 (2011).
M. Gentile, A. G. Recchia, C. Mazzone, et al., Expert. Opin. Pharmacother., 14, 2263 – 2280 (2013).
F. C. Torres, M. E. Garcia-Rubino, C. Lozano-Lopez, et al., Curr. Med. Chem., 22, 1312 – 1323 (2015).
W. Wang, D. Kong, H. Cheng, et al., Bioorg. Med. Chem. Lett., 24, 4250 – 4253 (2014).
A. S. Alpan, S. Zencir, I. Zupko, et al., J. Enzym. Inhib. Med. Chem., 24, 844 – 849 (2009).
N. Shrivastava, M. J. Naim, M. J. Alam, et al., Arch Pharm., 350(6) (2017).
Y. K. Yoon, M. A. Ali, A. C. Wei, et al., Eur. J. Med. Chem., 83, 448 – 454 (2014).
Y. K. Yoon and T. S. Choon, Arch. Pharm., 349, 1 – 8 (2016).
M. K. Akkoc, M. Y. Yuksel, I. Durmaz, et al., Turk. J. Chem., 36, 515 – 525 (2012).
A. M. Saab, M. Dobmeier, B. Koenig, et al., Anticancer Res., 33, 3027 – 3032 (2013).
M. Tuncbilek, E. B. Guven, T. Onder, et al., J. Med. Chem., 55, 3058 – 3065 (2012).
A. M. Waszkielewicz, A. Gunia, N. Szkaradek, et al., Bioorg. Med. Chem. Lett., 23, 4419 – 4423 (2013).
E. H. Cho, S. G. Chung, S. H. Lee, et al., WO1998000402 A1 (1998).
J. H. Lee, D. W. Kang, H. S. Kwon, et al., Arch. Pharm. Res., 27, 436 – 441 (2004).
M. Tuncbilek, E. B. Guven, T. Onder, et al., Med. Chem., 55, 3058 – 3065 (2012).
E. Banoglu, B. Caliskan, S. Luderer, et al., Bioorg. Med. Chem., 20, 3728 – 3741 (2012).
E. Banoglu, E. Celikoglu, S. Volker, et al., Eur. J. Med. Chem., 113, 1 – 10 (2016).
S. N. Baytas, N. Inceler, A. Yilmaz, et al., Bioorg. Med. Chem., 22, 3096 – 3104 (2014).
S. Cankara Pirol, B. Caliskan, I. Durmaz, et al., Eur. J. Med. Chem., 87, 140 – 149 (2014).
A. Ozdemir,M. Yildiz, F. S. Senol, et al., Food. Chem. Toxicol., 109, 898 – 909 (2017).
P. K. Dubey, P. V. V. P. Reddy, and K. Srinivas, Synth. Commun., 37, 1675 – 1681 (2007).
R. U. Jänicke, Breast Cancer Res. Treat., 117(1), 219 – 21 (2009).
D. Boucher, V. Blais, and J. B. Denault, Proc. Natl. Acad. Sci. USA, 109(15), 5669 – 74 (2012).
Acknowledgments
This work was supported by the Scientific and Technological Research Council of Turkey (TUBITAK Grant No: 115S016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors state no conflict of interest.
Rights and permissions
About this article
Cite this article
Özdemir, A., Turanli, S., Çalişkan, B. et al. Evaluation of Cytotoxic Activity of New Benzimidazole-Piperazine Hybrids Against Human MCF-7 and A549 Cancer Cells. Pharm Chem J 53, 1036–1046 (2020). https://doi.org/10.1007/s11094-020-02119-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-020-02119-9