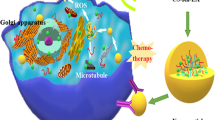
Abstract
Purpose
Paclitaxel is a first-line drug for the therapy of lung cancer, however, drug resistance is a serious limiting factor, related to overexpression of anti-apoptotic proteins like survivin. To overcome this phenomenon, developing novel ultrasound responsive nanobubbles - nanosized drug delivery system- for the delivery of paclitaxel and siRNA in order to silence survivin expression in the presence of ultrasound was aimed.
Methods
Paclitaxel-carrying nanobubble formulation was obtained by modifying the multistep method. Then, the complex formation of the nanobubbles - paclitaxel formulation with survivin-siRNA, was examined in terms of particle size, polydispersity index, zeta potential, and morphology. Furthermore, siRNA binding and protecting ability, cytotoxicity, cellular uptake, gene silencing, and induction of apoptosis studies were investigated in terms of lung cancer cells.
Results
Developed nanobubbles have particle sizes of 218.9–369.6 nm, zeta potentials of 27–34 mV, were able to protect siRNA from degradation and delivered siRNA into the lung cancer cells. Survivin expression was significantly lower compared with the control groups and enhanced apoptosis was induced by the co-delivery of survivin-siRNA and paclitaxel. Furthermore, significantly higher effects were obtained in the presence of ultrasound induction.
Conclusion
The ultrasound responsive nanobubble system carrying paclitaxel and survivin-siRNA is a promising and effective approach against lung cancer cells.







Similar content being viewed by others
Abbreviations
- DDAB:
-
Dimethyldiocta-decylammonium bromide
- DMEM:
-
Dulbecco’s Modified Eagle’s Medium
- ELISA:
-
Enzyme-linked immunosorbent assay
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- NB:
-
Nanobubbles
- NB-PTX:
-
Paclitaxel loaded nanobubbles
- NB-PTX:siRNA:
-
siRNA carrying paclitaxel loaded nanobubbles
- NSCLC:
-
Non-small cell lung cancer
- OD:
-
Optical density
- PDI:
-
Polydispersity index
- SCLC:
-
Small cell lung cancer
- SDS:
-
Sodium dodecyl sulfate
- SEM:
-
Scanning electron microscopy
- siRNA:
-
Small interfering RNA
- PBS:
-
Phosphate buffered saline
- PFP:
-
Perfluoropentane
- PTX:
-
Paclitaxel
- TBS:
-
Tris-buffered saline
- TEM:
-
Transmission electron microscopy
- US:
-
Ultrasound
- XTT:
-
2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide
- ZP:
-
Zeta potential
References
Mehta A, Dalle Vedove E, Isert L, Merkel OM. Targeting KRAS mutant lung Cancer cells with siRNA-loaded bovine serum albumin nanoparticles. Pharm Res. 2019;36:133.
Zhang M, Li G, Wang Y, Zhao S, Haihong P, Zhao H, et al. Expression in lung cancer and its correlation with driver mutations: a meta-analysis. Sci Rep. 2017;7:1–10.
Ettinger DS, Aisner DL, Wood DE, Akerley W, Bauman J, Chang JY, et al. NCCN guidelines insights: non–small cell lung Cancer, version 5.2018. J Natl Compr Cancer Netw. 2018;16:807–21.
Tiseo M, Gelsomino F, Alfieri R, Cavazzoni A, Bozzetti C, De Giorgi AM, et al. FGFR as potential target in the treatment of squamous non small cell lung cancer. Cancer Treat Rev. 2015;41:527–39.
Ekinci M, Ilem-Ozdemir D, Gundogdu E, Asikoglu M. Methotrexate loaded chitosan nanoparticles: preparation, radiolabeling and in vitro evaluation for breast cancer diagnosis. J Drug Deliv Sci Technol. 2015;30:107–13.
Akbaba H, Kantarcı AG, Erel AG. Development and in vitro evaluation of positive-charged solid lipid nanoparticles as nucleic acid delivery system in glioblastoma treatment. Marmara Pharm J. 2018;22:299–306.
Messaoudi K, Saulnier P, Boesen K, Benoit J-P, Lagarce F. Anti-epidermal growth factor receptor siRNA carried by chitosan-transacylated lipid nanocapsules increases sensitivity of glioblastoma cells to temozolomide. Int J Nanomedicine. 2014;9:1479–90.
Chakraborty C, Sharma AR, Sharma G, Doss CGP, Lee S-S. Therapeutic miRNA and siRNA: moving from bench to clinic as next generation medicine. Mol Ther - Nucleic Acids. 2017;8:132–43.
Chen J, Tang Y, Liu Y, Dou Y. Nucleic acid-based therapeutics for pulmonary diseases. AAPS PharmSciTech AAPS PharmSciTech. 2018;19:3670–80.
Ezrahi S, Aserin A, Garti N. Basic principles of drug delivery and microemulsions – the case of paclitaxel. Adv Colloid Interf Sci. 2019;263:95–130.
Nguyen CN, Tran BN, Do TT, Nguyen H, Nguyen TN. D-optimal optimization and data-analysis comparison between a DoE software and artificial neural networks of a chitosan coating process onto PLGA nanoparticles for lung and cervical cancer treatment. J Pharm Innov. 2018;14:206–20.
Xu Y, Zheng Y, Wu L, Zhu X, Zhang Z, Huang Y, et al. Novel solid lipid nanoparticle with Endosomal escape function for Oral delivery of insulin. ACS Nano. 2017;5:1–15.
Patel K, Doddapaneni R, Patki M, Sekar V, Bagde A, Singh M. Erlotinib-Valproic acid Liquisolid formulation: evaluating Oral bioavailability and cytotoxicity in Erlotinib-resistant non-small cell lung Cancer cells. AAPS PharmSciTech. 2019;20:1–11.
Thakur SS, Chen YS, Houston ZH, Fletcher N, Barnett NL, Thurecht KJ, et al. Ultrasound-responsive nanobubbles for enhanced intravitreal drug migration: an ex vivo evaluation. Eur J Pharm Biopharm. 2019;136:102–7.
Cavalli R, Bisazza A, Giustetto P, Civra A, Lembo D, Trotta G, et al. Preparation and characterization of dextran nanobubbles for oxygen delivery. Int J Pharm. 2009;381:160–5.
Argenziano M, Banche G, Luganini A, Finesso N, Allizond V, Gulino GR, et al. Vancomycin-loaded nanobubbles: a new platform for controlled antibiotic delivery against methicillin-resistant Staphylococcus aureus infections. Int J Pharm. 2017;523:176–88.
Boissenot T, Bordat A, Fattal E, Tsapis N. Ultrasound-triggered drug delivery for cancer treatment using drug delivery systems: from theoretical considerations to practical applications. J Control Release. 2016;241:144–63.
Perera RH, Solorio L, Wu H, Gangolli M, Silverman E, Hernandez C, et al. Nanobubble ultrasound contrast agents for enhanced delivery of thermal sensitizer to tumors undergoing radiofrequency ablation. Pharm Res. 2014;31:1407–17.
Lentacker I, De Geest BG, Vandenbroucke RE, Peeters L, Demeester J, De Smedt SC, et al. Ultrasound-responsive polymer-coated microbubbles that bind and protect DNA. Langmuir. 2006;22:7273–8.
Cavalli R, Bisazza A, Lembo D. Micro- and nanobubbles: a versatile non-viral platform for gene delivery. Int J Pharm. 2013;456:437–45.
Hoosein MM, Barnes D, Khan AN, Peake MD, Bennett J, Purnell D, et al. The importance of ultrasound in staging and gaining a pathological diagnosis in patients with lung cancer—a two year single centre experience. Thorax. 2011;3:8–11.
Miller A. Practical approach to lung ultrasound. BJA Educ. 2016;16:39–45.
Andolfi M, Potenza R, Capozzi R, Liparulo V, Puma F. The role of bronchoscopy in the diagnosis of early lung cancer : a review. J Thorac Dis. 2016;8:3329–37.
Dooms C, Muylle I, Yserbyt J, Ninane V. Endobronchial ultrasound in the management of nonsmall cell lung cancer. Eur Respir Rev. 2013;22:169–77.
Zhang X, Zheng Y, Wang Z, Huang S, Chen Y, Jiang W, et al. Methotrexate-loaded PLGA nanobubbles for ultrasound imaging and synergistic targeted therapy of residual tumor during HIFU ablation. Biomaterials. 2014;35:5148–61.
Alheshibri M, Craig VSJ. Armoured nanobubbles; ultrasound contrast agents under pressure. J Colloid Interface Sci. 2019;537:123–31.
Zhang X, Wu M, Zhang Y, Zhang J, Su J, Yang C. Molecular imaging of atherosclerotic plaque with lipid nanobubbles as targeted ultrasound contrast agents. Colloids Surfaces B Biointerfaces. 2020;189:110861.
Xenariou S, Griesenbach U, Liang H, Zhu J, Farley R, Somerton L, et al. Use of ultrasound to enhance nonviral lung gene transfer in vivo. Gene Ther. 2007;14:768–74.
Sugiyama MG, Mintsopoulos V, Raheel H, Goldenberg NM, Batt JE, Brochard L, et al. Lung ultrasound and microbubbles enhance aminoglycoside efficacy and delivery to the lung in Escherichia coli –induced pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2018;198:404–8.
Li J, Xi A, Qiao H, Liu Z. Ultrasound-mediated diagnostic imaging and advanced treatment with multifunctional micro / nanobubbles. Cancer Lett. 2020;475:92–8.
Başpınar Y, Erel-Akbaba G, Kotmakçı M, Akbaba H. Development and characterization of nanobubbles containing paclitaxel and survivin inhibitor YM155 against lung cancer. Int J Pharm. 2019;566:149–56.
Kotmakçı M, Akbaba H, Erel G, Ertan G, Kantarcı G. Improved method for solid lipid nanoparticle preparation based on hot microemulsions: preparation, characterization, cytotoxicity, and Hemocompatibility evaluation. AAPS PharmSciTech. 2017;18:1355–65.
Patil ML, Zhang M, Minko T. Multifunctional triblock nanocarrier (PAMAM-PEG-PLL) for the efficient intracellular siRNA delivery and gene silencing. ACS Nano. 2011;5:1877–87.
Erel G, Kotmakçı M, Akbaba H, Sözer Karadağlı S, Kantarcı AG. Nanoencapsulated chitosan nanoparticles in emulsion-based oral delivery system: in vitro and in vivo evaluation of insulin loaded formulation. J Drug Deliv Sci Technol. 2016;36:161–7.
Lu M, Zhao X, Xing H, Xun Z, Zhu S, Lang L, et al. Comparison of exosome-mimicking liposomes with conventional liposomes for intracellular delivery of siRNA. Int J Pharm. 2018;550:100–13.
Başpinar Y, Akbaba H, Bayraktar O. Encapsulation of paclitaxel in electrosprayed chitosan nanoparticles. J Res Pharm. 2019;23:886–96.
Bi YZ, Zhang YF, Cui CY, Ren LL, Jiang XY. Gene-silencing effects of anti-survivin siRNA delivered by RGDV-functionalized nanodiamond carrier in the breast carcinoma cell line MCF-7. Int J Nanomedicine. 2016;11:5771–87.
Bulbake U, Kommineni N, Bryszewska M, Ionov M, Khan W. Cationic liposomes for co-delivery of paclitaxel and anti-Plk1 siRNA to achieve enhanced efficacy in breast cancer. J Drug Deliv Sci Technol. 2018;48:253–65.
Popova P, Notabi MK, Code C, Arnspang EC, Andersen MØ. Co-delivery of siRNA and etoposide to cancer cells using an MDEA esterquat based drug delivery system. Eur J Pharm Sci. 2019;127:142–50.
Lyu H, Wang S, Huang J, Wang B, He Z, Liu B. Survivin-targeting miR-542-3p overcomes HER3 signaling-induced chemoresistance and enhances the antitumor activity of paclitaxel against HER2-overexpressing breast cancer. Cancer Lett. 2018;420:97–108.
Chen X, Zhang Y, Tang C, Tian C, Sun Q, Su Z, et al. Co-delivery of paclitaxel and anti-survivin siRNA via redox-sensitive oligopeptide liposomes for the synergistic treatment of breast cancer and metastasis. Int J Pharm. 2017;529:102–15.
Tabatabaei SN, Derbali RM, Yang C, Superstein R, Hamel P, Chain JL, et al. Co-delivery of miR-181a and melphalan by lipid nanoparticles for treatment of seeded retinoblastoma. J Control Release. 2019;298:177–85.
Akbaba H, Erel Akbaba G, Kantarcı AG. Development and evaluation of antisense shRNA-encoding plasmid loaded solid lipid nanoparticles against 5-α reductase activity. J Drug Deliv Sci Technol. 2018;44:270–7.
Cavalli R, Argenziano M, Vigna E, Giustetto P, Torres E, Aime S, et al. Preparation and in vitro characterization of chitosan nanobubbles as theranostic agents. Colloids Surfaces B Biointerfaces. 2015;129:39–46.
Mehta SK. Bhawna, Kaur K, Bhasin KK. Micellization behavior of cationic surfactant dodecyldimethylethylammonium bromide (DDAB) in the presence of papain. Colloids Surfaces A Physicochem Eng Asp. 2008;317:32–8.
Parodi A, Molinaro R, Sushnitha M, Evangelopoulos M, Martinez JO, Arrighetti N, et al. Bio-inspired engineering of cell- and virus-like nanoparticles for drug delivery. Biomaterials. 2017;147:155–68.
Fernandez-Piñeiro I, Pensado A, Badiola I, Sanchez A. Development and characterisation of chondroitin sulfate- and hyaluronic acid-incorporated sorbitan ester nanoparticles as gene delivery systems. Eur J Pharm Biopharm. 2018;125:85–94.
Erel-Akbaba G, Carvalho LA, Tian T, Zinter M, Akbaba H, Obeid PJ, et al. Radiation-induced targeted nanoparticle-based gene delivery for brain tumor therapy. ACS Nano. 2019;13:4028–40.
Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature. 2004;432:173–8.
Tai W. Chemical modulation of siRNA lipophilicity for efficient delivery. J Control Release. 2019;307:98–107.
Büyükköroğlu G, Şenel B, Başaran E, Yenilmez E, Yazan Y. Preparation and in vitro evaluation of vaginal formulations including siRNA and paclitaxel-loaded SLNs for cervical cancer. Eur J Pharm Biopharm. 2016;109:174–83.
Yang F, Huang W, Li Y, Liu S, Jin M, Wang Y, et al. Anti-tumor effects in mice induced by survivin-targeted siRNA delivered through polysaccharide nanoparticles. Biomaterials. 2013;34:5689–99.
Fröhlich E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int J Nanomedicine. 2012;7:5577–91.
Xiong H, Yu S, Zhuang L, Xiong H. Changes of survivin mRNA and protein expression during paclitaxel treatment in breast cancer cells. J Huazhong Univ Sci Technol. 2007;27:65–7.
Rauch A, Hennig D, Schäfer C, Wirth M, Marx C, Heinzel T, et al. Survivin and YM155: how faithful is the liaison? Biochim Biophys Acta - Rev Cancer. 1845;2014:202–20.
Li Z, Zhang L, Tang C, Yin C. Co-delivery of doxorubicin and Survivin shRNA-expressing plasmid via microenvironment-responsive dendritic mesoporous silica nanoparticles for synergistic cancer therapy. Pharm Res Pharmaceutical Research. 2017;34:2829–41.
Acknowledgments
This study has been financially supported by the Scientific and Technological Research Council of Turkey (TUBITAK) under grant code 116S213.
The authors would like to thank Ege University, Faculty of Pharmacy, Department of Pharmaceutical Technology (for DLS measurement), Ege University, Pharmaceutical Sciences Research Center (for Varioskan Multiplate Reader), Izmir Katip Celebi University, Central Research Labs (for SEM imaging) and Middle East Technical University, Central Laboratory (for TEM imaging).
Funding
This study was funded by the Scientific and Technological Research Council of Turkey (TUBITAK) under grant code 116S213.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent is not applicable to this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akbaba, H., Erel-Akbaba, G., Kotmakçı, M. et al. Enhanced Cellular Uptake and Gene Silencing Activity of Survivin-siRNA via Ultrasound-Mediated Nanobubbles in Lung Cancer Cells. Pharm Res 37, 165 (2020). https://doi.org/10.1007/s11095-020-02885-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-020-02885-x