Abstract
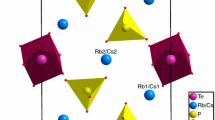
A new mixed compound, sodium rubidium phosphate tellurate, was elaborated using the slow evaporation method at room temperature, confirmed by X-ray single-crystal diffraction. The single-crystal X-ray diffraction structure of Na2.64Rb0.36PO4.Te(OH)6 is identified for the first time. At 298 K, structural analysis indicates that this compound crystallizes in the monoclinic system with P63 space group. The main feature of these atomic arrangements lies in the coexistence of two different anions (PO\(_{4}^{3 - }\) and TeO\(_{6}^{6 - }\) groups) in the unit cell, connected by hydrogen bonds which build up the crystal. The O–H…O hydrogen bonds between Te(OH)6 octahedrons and PO4 tetrahedral contribute to the cohesion and the stability of the atomic arrangement. The Na et Rb atoms are intercalating between their planes. The Fourier transform infrared spectrum exhibited characteristic bands corresponding to the tellurate and phosphate group. Thermal studies were carried out to examine the obtained material and yielded the presence of three phase transitions. The protonic conduction phase transition due to the presence of the breaking of O–H…O hydrogen bonds, was regarded as a technologically significant aspect for the new compound. These results corroborate that the new phosphate tellurate can be considered as a promising material in terms of enacting fruitful and constructive applications in advanced technology. The crystal structure was built up from corner-sharing TeOH6 octahedrons, which intercalated by PO4 tetrahedral units. These two groups are connected by hydrogen bonds which make up the building of the crystal, and the Na et Rb atoms are sandwitched between tetrahedral and Te(OH)6 octahedral planes.











Similar content being viewed by others
References
B. Bazan, J.L. Mesa, J.L. Pizarro, L. Lezama, A. Pena, M.L. Arriortua, T. Roja, J. Solid State Chem. 179, 1459 (2006)
R.K. Brow, J. Non-Cryst. Solids 263, 1 (2000)
S.W. Martin, J. Am. Ceram. Soc 74, 1767 (1991)
A. Kalaji, S. Skanthakumar, M.G. Kanatzidis, J.F. Mitchell, L. Soderholm, Inorg. Chem. 53, 6321 (2014)
A. Kalaji, L. Soderholm, Inorg. Chem. 53, 11252 (2014)
J. Lin, K. Diefenbach, J.N. Cross, J.M. Babo, T.E. Albrecht-Schmitt, Inorg. Chem. 52, 13278 (2013)
J. Lin, K. Diefenbach, N. Kikugawa, R.E. Baumbach, T.E. Albrecht-Schmitt, Inorg. Chem. 53, 8555 (2014)
L. Ktari, M. Dammak, A. Hadrich, A. Cousson, M. Nierlich, F. Romain, T. Mhiri, Solid State Sci. 6, 1393 (2004)
A. Elferjani, S. Garcia-Granda, M. Dammak, J. Res. Chem. Intermed. 45, 1357 (2018)
A. Elferjani, S. Garcia-Granda, M. Dammak, J. Alloys Compd. 749, 448 (2018)
H. Khemakhem, Ferroelectrics 234, 47 (1999)
L. Ktari, M. Dammak, A. Madani, T. Mhiri, Solid State Ion. 145, 225 (2001)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, J. Acta Cryst 36, 2743 (1980)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, Acta Cryst. B 37, 650 (1981)
R. Zilber, A. Durif, M.T. Averbuch-Pouchot, Acta Cryst. B 38, 1554 (1982)
M. Dammak, H. Khemakhem, T. Mhiri, A.W. Kolsi, A. Daoud, J. Alloys Compd. 280, 107 (1998)
M. Dammak, H. Khemakhem, T. Mhiri, J. Phys. Chem. Solids. 62, 2069 (2001)
M. Abdelhedi, M. Dammak, A. Cousson, M. Nierlich, A.W. Kolsi, Acta Cryst. E 61, 256 (2005)
L. Ktari, M. Dammak, T. Mhiri, A.W. Kolsi, Phys. Procedia. 2, 729 (2009)
F. B. Tahar, A. Elferjani, S. Garcia-Granda, M. Dammak, Inorg. Chem. Commun. 118, 108003 (2020)
K. Ghorbel, H. Litaiem, L. Ktari, S. Garcia-Granda, M. Dammak, J. Mol. Struct. 1079, 225 (2015)
H. Frikha, M. Abdelhedi, M. Dammak, S. Garcia-Granda, J. Saudi Chem. Soc. 21, 324 (2017)
H. Frikha, M. Abdelhedi, B. Louati, M. Dammak, S. Garcia-Granda, J. Therm. Anal. Calorim. 131, 2795 (2018)
M.T. Averbuch-Pouchout, A. Durif, J.C. Guitel, Mat. Res. Bull. 14, 1219 (1979)
M.T. Averbuch-Pouchot, Acta. Cryst. B36, 2405 (1980)
Nonius, in B.V. Nonius (Ed.), Kappa CCD Sever Software, Delft, The Netherlands (1999)
APEX2 version 1. 0–8, Bruker AXS, Madison, WI (2003)
D.J. Watkin, C.K. Prout, J.R. Carruthers, P.W. Betteridge, R.I. Cooper, CRYSTALS Issue 11 (Chemical Crystallography Laboratory, Oxford, UK, 2001)
G.M. Sheldrick, SHELXS-97 and SHELXL-97, Program for Crystal Structure Refinement (Univ. of Göttingen, Germany, 1970)
K. Brandenburg, M. Berndt, DIAMOND Version 2.1.b, Crystal Impact, Gb R, Bonn, Germany (1999)
E. Grech, Z. Malarski, W. Sawka-Dobrowolska, L. Sobczyk, J. Mol. Struct. 416, 227 (1997)
L.C. Thomas, R.A. Chittenden, H.E. Hartley, Nature 192, 1283 (1961)
L.C. Thomas, R.A. Chittenden, J. Spectrochim. Acta. 20, 489 (1964)
H.D. Lutz, B. Engelen, Trends. Appl. Spectrosc. 4, 355 (2002)
H.D. Lutz, J. Mol. Struct. 646, 227 (2003)
A. Novak, Hydrogen Bonding in Solids. (Spring-Verlag, Berlin, Heidelberg, New York, 1974) 18:177
K. Viswanathan, V.U. Nayar, G. Aruldhas, J. Infrared Phys 26, 353 (1986)
A. Hadrich, A. Lautie, T. Mhiri, F. Romain, Vib Spectrosc. 26, 51 (2001)
H. Naili, T. Mhiri, A. Daoud, Phase Transit. 71, 271 (1999)
D. Philip, G. Aruldhas, J. Raman Spectrosc. 20, 637 (1989)
J. Gaumt, Trans. Faraday Sot. 49, 112 (1953)
A. Elferjani, M. Abdelhedi, M. Dammak, A, W, Kolsi. J. Appl. Phys. A. 122, 742 (2016)
H. Litaiem, M. Dammak, L. Ktari, S. Kamoun, T. Mhiri, Phase Transit. 77, 929 (2004)
M. Dammak, H. Litaiem, T. Mhiri, J. Alloys Compd. 416, 228 (2006)
M. Dammak, H. Khemakhem, T. Mhiri, A.W. Kolsi, A. Daoud, J. Solid State Chem. 145, 612 (1999)
J. Faby, J. Loub, L. Feltl, J. Therm. Anal. 24, 95 (1982)
M. Dammak, H. Khemakhem, N. Zouari, T. Mhiri, A.W. Kolsi, Solid State Ion. 127, 125 (2000)
M. Djemel, M. Abdelhedi, L. Ktari, M. Dammak, J. Mol. Struct. 1033, 84 (2013)
Q.J. Jiao, Y.L. Zhu, H. Huang, H. Ren, J Therm Anal Calorim. 116, 1125 (2014)
R.L. Frost, A. Locke, W.N. Martens, J. Therm. Anal. Cal. 93, 993 (2008)
Acknowledgements
This work is supported by the Ministry of Higher Education and Research of Tunisia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elferjani, A., Zaidi, S., Garcia-Granda, S. et al. Synthesis, X-ray diffraction, thermal behavior and physicochemical studies of phase transitions before the decomposition in the new phosphate tellurate protonic conductor compound. Res Chem Intermed 47, 5099–5118 (2021). https://doi.org/10.1007/s11164-021-04584-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-021-04584-7