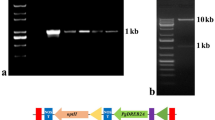
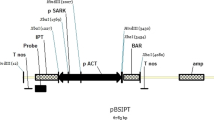
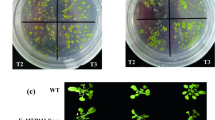
Abstract
Peanut is widely grown and provides protein and edible oil for millions of people. Peanut growth and productivity are frequently negatively affected by abiotic and biotic environmental factors. However, the research on improving peanut germplasm resources by genetic transformation is very limited. Here, the novel R2R3-MYB repressor GmMYB3a was introduced into peanut plants by Agrobacterium-mediated transformation for the first time for thorough evaluation of the function of GmMYB3a in drought stress plant responses. We generated GmMYB3a-transgenic peanut plants. The GmMYB3a-overexpressing lines showed significantly improved physiological responses and no yield loss non-transgenic plants, in terms of survival rates. Thus, the GmMYB3a-overexpressing plants showed better photosynthetic performance, higher relative water content, and greater water use efficiency, demonstrating their adaptive capacity to water deficit. We conclude that overexpression of GmMYB3a can improve drought tolerance and productivity in peanut.




Similar content being viewed by others
References
An JP, Zhang XW, Bi SQ, You CX, Wang XF, Hao YJ (2020) The ERF transcription factor MdERF38 promotes drought stress-induced anthocyanin biosynthesis in apple. Plant J 101(3):573–589. https://doi.org/10.1111/tpj.14555
Aninbon C, Jogloy S, Vorasoot N, Nuchadomrong S, Holbrook CC, Kvien C, Patanothai A (2019) Effect of terminal drought on arginine content in peanut genotypes with difference in levels of drought resistance. Int J Plant Prod 13(2):155–162. https://doi.org/10.1007/s42106-019-00043-x
Arunyanark A, Jogloy S, Akkasaeng C, Vorasoot N, Kesmala T, Nageswara Rao RC, Wright GC, Patanothai A (2008) Chlorophyll stability is an indicator of drought tolerance in peanut. J Agron Crop Sci 194(2):113–125. https://doi.org/10.1111/j.1439-037X.2008.00299.x
Banavath JN, Chakradhar T, Pandit V, Konduru S, Guduru KK, Akila CS, Podha S, Puli COR (2018) Stress Inducible Overexpression of AtHDG11 Leads to Improved Drought and Salt Stress Tolerance in Peanut (Arachis hypogaea L.). Front Chem 6:34. https://doi.org/10.3389/fchem.2018.00034
Banjara M, Zhu L, Shen G, Payton P, Zhang H (2011) Expression of an Arabidopsis sodium/proton antiporter gene (AtNHX1) in peanut to improve salt tolerance. Plant Biotechnol Rep 6(1):59–67. https://doi.org/10.1007/s11816-011-0200-5
Basu S, Ramegowda V, Kumar A, Pereira A (2016) Plant adaptation to drought stress [version 1; peer review: 3 approved]. F1000Research 5:1554. https://doi.org/10.12688/f1000research.7678.1
Bhatnagar-Mathur P, Devi MJ, Reddy DS, Lavanya M, Vadez V, Serraj R, Yamaguchi-Shinozaki K, Sharma KK (2007) Stress-inducible expression of At DREB1A in transgenic peanut (Arachis hypogaea L.) increases transpiration efficiency under water-limiting condition. Plant Cell Rep 26(12):2071–2082. https://doi.org/10.1007/s00299-007-0406-8)
Cuc LM, Mace ES, Crouch JH, Quang VD, Long TD, Varshney RK (2008) Isolation and characterization of novel microsatellite markers and their application for diversity assessment in cultivated groundnut (Arachis hypogaea). BMC Plant Biol 8(55):1. https://doi.org/10.1186/1471-2229-8-55
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28(3):350–356. https://doi.org/10.1021/ac60111a017
Elavarthi S, Martin B (2010) Spectrophotometric Assays for Antioxidant Enzymes in Plants. Methods Mol Biol (Clifton, N.J.) 639:273–280. https://doi.org/10.1007/978-1-60761-702-0_16
Gong DH, Wang GZ, Si WT, Zhou Y, Liu Z, Jia J (2018) Effects of salt stress on photosynthetic pigments and activity of ribulose-1,5-bisphosphate carboxylase/oxygenase in Kalidium foliatum. Russ J Plant Physiol 65(1):98–103. https://doi.org/10.1134/s1021443718010144
Hassanzadeh M, Ebadi A, Panahyanekivi M, Eshghi AG, Jamaatiesomarin S, Saeidi M, Zabihi-e-Mahmoodabad R (2009) Evaluation of drought stress on relative water content and chlorophyll content of sesame (Sesamum indicum L) genotypes at early flowering stage. Res J Environ Sci 3(3):345–350. https://doi.org/10.3923/rjes.2009.345.350
He Y, Chen Y, Yu CL, Lu KX, Jiang QS, Fu JL, Wang GM, Jiang DA (2016) Photosynthesis and yield traits in different soybean lines in response to salt stress. Photosynthetica 54(4):630–635. https://doi.org/10.1007/s11099-016-0218-6
He Y, Yang X, Xu C, Guo D, Niu L, Wang Y, Li J, Yan F, Wang Q (2018) Overexpression of a novel transcriptional repressor GmMYB3a negatively regulates salt-alkali tolerance and stress-related genes in soybean. Biochem Biophys Res Commun 498(3):586–591. https://doi.org/10.1016/j.bbrc.2018.03.026
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207(4):604–611. https://doi.org/10.2307/23385611
Hsu SY, Kao CH (2003) Differential effect of sorbitol and polyethylene glycol on antioxidant enzymes in rice leaves. Plant Growth Regul 39(1):83–90. https://doi.org/10.1023/A:1021830926902
Jain S, Kumar D, Jain M, Chaudhary P, Deswal R, Sarin NB (2011) Ectopic overexpression of a salt stress-induced pathogenesis-related class 10 protein (PR10) gene from peanut (Arachis hypogaea L.) affords broad spectrum abiotic stress tolerance in transgenic tobacco. Plant Cell Tissue Organ Culture (PCTOC) 109(1):19–31. https://doi.org/10.1007/s11240-011-0069-6
Kang C, Zhai H, He S, Zhao N, Liu Q (2019) A Novel Sweetpotato bZIP Transcription Factor Gene, IbbZIP1, is involved in salt and drought tolerance in transgenic Arabidopsis. Plant Cell Rep 38(11):1373–1382. https://doi.org/10.1007/s00299-019-02441-x
Khare T, Kishor PBK, Kumar V (2015) Na+ and Cl− ions show additive effects under NaCl stress on induction of oxidative stress and the responsive antioxidative defense in rice. Protoplasma 252(4):1149–1165. https://doi.org/10.1007/s00709-014-0749-2
Kim TH (2014) Mechanism of ABA signal transduction: agricultural highlights for improving drought tolerance. J Plant Biol 57(1):1–8. https://doi.org/10.1007/s12374-014-0901-8
Kiranmai K, Rao GL, Pandurangaiah M, Nareshkumar A, Reddy VA, Lokesh U, Venkatesh B, Johnson AMA, Sudhakar C (2018) A novel WRKY transcription factor, MuWRKY3 (Macrotyloma uniflorum Lam. Verdc.) enhances drought stress tolerance in transgenic groundnut (Arachis hypogaea L.) plants. Front Plant Sci 16(9):346. https://doi.org/10.3389/fpls.2018.00346
Koenigshofer H, Loeppert HG (2019) The up-regulation of proline synthesis in the meristematic tissues of wheat seedlings upon short-term exposure to osmotic stress. J Plant Physiol 237:21–29. https://doi.org/10.1016/j.jplph.2019.03.010
Kumar D, Kirti PB (2015) Pathogen-induced SGT1 of Arachis diogoiinduces cell death and enhanced disease resistance in tobacco and peanut. Plant Biotechnol J 13(1):73–84. https://doi.org/10.1111/pbi.12237
Lee SK, Jeon JS, Bornke F, Voll L, Cho JI, Goh CH, Jeong SW, Park YI, Kim SJ, Choi SB, Miyao A, Hirochika H, An G, Cho MH, Bhoo SH, Sonnewald U, Hahn TR (2008) Loss of cytosolic fructose-1,6-bisphosphatase limits photosynthetic sucrose synthesis and causes severe growth retardations in rice (Oryza sativa). Plant Cell Environ 31(12):1851–1863. https://doi.org/10.1111/j.1365-3040.2008.01890.x
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Mace ES, Phong DT, Upadhyaya HD (2006) SSR analysis of cultivated groundnut (Arachis hypogaea L) germplasm resistant to rust and late leaf spot diseases. Euphytica 152:317–330. https://doi.org/10.1007/s10681-006-9218-0
Meher Shivakrishna P, Reddy KA, Rao DM (2018) Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J Biol Sci 25(2):285–289. https://doi.org/10.1016/j.sjbs.2017.04.008
Mellacheruvu S, Tamirisa S, Vudem DR, Khareedu VR (2016) Pigeonpea hybrid-proline-rich protein (CcHyPRP) confers biotic and abiotic stress tolerance in transgenic rice. Front Plant Sci 6:1167. https://doi.org/10.3389/fpls.2015.01167
Monneveux P, Rekika D, Acevedo E, Merah O (2006) Effect of drought on leaf gas exchange, carbon isotope discrimination, transpiration efficiency and productivity in field grown durum wheat genotypes. Plant Sci 170(4):867–872. https://doi.org/10.1016/j.plantsci.2005.12.008
Ning W, Zhai H, Yu J, Liang S, Yang X, Xing X, Huo J, Pang T, Yang Y, Bai X (2017) Overexpression of Glycine soja WRKY20 enhances drought tolerance and improves plant yields under drought stress in transgenic soybean. Mol Breed 37(2):1. https://doi.org/10.1007/s11032-016-0614-4
Noman M, Jameel A, Qiang WD, Ahmad N, Liu WC, Wang FW, Li HY (2019) Overexpression of GmCAMTA12 enhanced drought tolerance in Arabidopsis and Soybean. Int J Mol Sci 20(19):4849. https://doi.org/10.3390/ijms20194849
Painawadee MS, Jogloy T, Kesmala C, Akkasaeng C, Patanothai A (2009) Identification of traits related to drought resistance in peanut (Arachis hypogaea L.). Asian J Plant Sci 8(2):120–128. https://doi.org/10.3923/ajps.2009.120.128
Peleg Z, Reguera M, Tumimbang E, Walia H, Blumwald E (2011) Cytokinin-mediated source/sink modifications improve drought tolerance and increase grain yield in rice under water-stress. Plant Biotechnol J 9(7):747–758. https://doi.org/10.1111/j.1467-7652.2010.00584.x
Pilon C, Snider JL, Sobolev V, Chastain DR, Sorensen RB, Meeks CD, Massa AN, Walk T, Singh B, Earl HJ (2018) Assessing stomatal and non-stomatal limitations to carbon assimilation under Progressive drought in peanut (Arachis Hypogaea L.). J Plant Physiol 231:124–134. https://doi.org/10.1016/j.jplph.2018.09.007
Qin H, Gu Q, Kuppu S, Sun L, Zhu X, Mishra N, Hu R, Shen G, Zhang J, Zhang Y, Zhu L, Zhang X, Burow M, Payton P, Zhang H (2012) Expression of the Arabidopsis vacuolar H + -pyrophosphatase gene AVP1 in peanut to improve drought and salt tolerance. Plant Biotechnol Rep 7(3):345–355. https://doi.org/10.1007/s11816-012-0269-5
Reddy TY, Reddy VR, Anbumozhi V (2003) Physiological responses of groundnut (Arachis hypogea L.) to drought stress and its amelioration:a critical review. Plant Growth Regul 41(1):75–88. https://doi.org/10.1023/a:1027353430164
Redillas MCFR, Park SH, Lee JW, Kim YS, Jeong JS, Jung H, Bang SW, Hahn TR, Kim JK (2011) Accumulation of trehalose increases soluble sugar contents in rice plants conferring tolerance to drought and salt stress. Plant Biotechnol Rep 6(1):89–96. https://doi.org/10.1007/s11816-011-0210-3
Richard M (2012) Traditional Plant Breeding vs. genetic engineering—a primer. Western Farm Press 34(22):15
Roychoudhury A, Paul S, Basu S (2013) Cross-talk between abscisic acid-dependent and abscisic acid-independent pathways during abiotic stress. Plant Cell Rep 32(7):985–1006. https://doi.org/10.1007/s00299-013-1414-5
Sharma KK, Anjaiah V (2000) An efficient method for the production of transgenic plants of peanut (Arachis hypogaea L.) through Agrobacterium tumefaciens-mediated genetic transformation. Plant Sci 159(1):7–19. https://doi.org/10.1016/s0168-9452(00)00294-6
So HA, Lee JH (2019) NAC Transcription factors from soybean (Glycine max L.) differentially regulated by abiotic stress. J Plant Biol 62(2):147–160. https://doi.org/10.1007/s12374-018-0285-2
Swapnil P, Rai AK (2018) Physiological responses to salt stress of salt-adapted and directly salt (NaCl and NaCl + Na2SO4 mixture)-stressed cyanobacterium Anabaena fertilissima. Protoplasma 255(3):963–976. https://doi.org/10.1007/s00709-018-1205-5
Tavakkoli E, Rengasamy P, McDonald GK (2010) High concentrations of Na+ and Cl− ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J Exp Bot 61(15):4449–4459. https://doi.org/10.1093/jxb/erq251
Troll W, Lindsley J (1955) A photometric method for the determination of proline. J Biol Chem 215(2):655–660. https://doi.org/10.2165/00128413-200816480-00062
Wei Q, Zhang F, Sun F, Luo Q, Wang R, Hu R, Chen M, Chang J, Yang G, He G (2017) A wheat MYB transcriptional repressor TaMyb1D regulates phenylpropanoid metabolism and enhances tolerance to drought and oxidative stresses in transgenic tobacco plants. Plant Sci 265:112–123. https://doi.org/10.1016/j.plantsci.2017.09.020
Xiong L, Zhu JK (2002) Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ 25(2):131–139. https://doi.org/10.1046/j.1365-3040.2002.00782.x
Xiong L, Schumaker KS, Zhu JK (2002) Cell signaling during cold, drought, and salt stress. Plant Cell 14:165–183. https://doi.org/10.1105/tpc.000596
Yang Z, Li JL, Liu LN, Xie Q, Sui N (2020) Photosynthetic regulation under salt stress and salt-tolerance mechanism of sweet sorghum. Front Plant Sci 10:1722. https://doi.org/10.3389/fpls.2019.01722
Yuan X, Wang H, Cai J, Bi Y, Li D, Song F (2019) Rice NAC transcription factor ONAC066 functions as a positive regulator of drought and oxidative stress response. BMC Plant Biol 19(1):278. https://doi.org/10.1186/s12870-019-1883-y
Zhang L, Xiao S, Li W, Feng W, Li J, Wu Z, Gao X, Liu F, Shao M (2011) Overexpression of a Harpin-encoding gene hrf1 in rice enhances drought tolerance. J Exp Bot 62(12):4229–4238. https://doi.org/10.1093/jxb/err131
Zhang YQ, Lu X, Zhao FY, Li QT, Niu SL, Wei W, Zhang WK, Ma B, Chen SY, Zhang JS (2016) Soybean GmDREBL increases lipid content in seeds of Transgenic Arabidopsis. Sci Rep 6(1):1. https://doi.org/10.1038/srep34307
Zhao X, Li C, Wan S, Zhang T, Yan C, Shan S (2018) Transcriptomic analysis and discovery of genes in the response of Arachis hypogaea to drought stress. Mol Biol Rep 45(2):119–131. https://doi.org/10.1007/s11033-018-4145-4
Acknowledgements
This work was supported by Department of Science and Technology of Jilin Province——Creation of high-quality germplasm resources and breeding of new varieties for peanut and sunflower (grant number: 2018020170NY). The Provincial Talent Development Fund Project——Demonstration and transformation of new peanut varieties with high yield and good quality (“Jihua 5″ and “Jihua 6”) (grant number: C82400002). The Fundamental research on crop germplasm resources in the 13th five-year plan (grant number: C92073505).
Author information
Authors and Affiliations
Contributions
YL designed the experiments. YH and ZH conducted the experiments and drafted the manuscript. SM and BW participated in the investigation of photosynthetic physiology and agronomic characters. YH and BW conducted Agrobacterium-mediated transformation experiments and qRT-PCR analyses. All authors participated in the revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, Y., Mu, S., He, Z. et al. Ectopic expression of MYB repressor GmMYB3a improves drought tolerance and productivity of transgenic peanuts (Arachis hypogaea L.) under conditions of water deficit. Transgenic Res 29, 563–574 (2020). https://doi.org/10.1007/s11248-020-00220-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-020-00220-z