Abstract
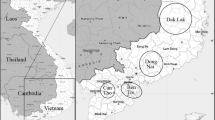
Vietnam has the potential to become a larger cocoa producer as the country experiences climatic conditions favorable to the growth of the crop. However, to enable a high cacao quality, genetic methods are required to identify and conserve promising cacao cultivars. Previous research as discussed by Everaert et al. (2017), using 14 microsatellite (SSR) markers, showed a genetic characterization of Vietnamese cacao, but additional research is required to further clarify the genetic background of the different Vietnamese cacao cultivars. Therefore, in this study, the Vietnamese dataset was enlarged, and a classification model was developed. Firstly, we compared the classification strength of the 14 previously applied SSR markers and 42 additional single nucleotide polymorphism (SNP) markers, either separately or combined, using 53 international reference cacao cultivars. Secondly, the most classifying marker set was used to examine the genetic associations of the Vietnamese cultivars to the reference cultivars, using Bayesian clustering and principal coordinate analysis (PCoA). The classification method based on the combined marker set performed best and could subdivide the Vietnamese cultivars in three clusters. The first cluster was strongly related to Nanay, the second to Criollo/Trinitario, and the third predominantly to Amelonado and Criollo/Trinitario. For all marker sets, an apparent high genetic diversity could be observed for the total Vietnam collection. The obtained knowledge provides a strong genetic basis for the Vietnamese cacao industry. In addition, the proposed marker set and the analysis workflow could be applied worldwide for future breeding programs and further production of origin chocolate.




Similar content being viewed by others
References
Bartra TE (1993) Caracterización botánica-agronómica ex situ de 20 clones de cacao (Theobroma cacao L.) colectados en la cuenca del rıo Huallaga. Dissertation, Universidad Nacional de la Selva
Boza EJ, Irish BM, Meerow AW, Tondo CL, Rodríguez OA, Ventura-López M, Gómez JA, Moore JM, Zhang D, Motamayor JC, Schnell RJ (2013) Genetic diversity, conservation, and utilization of Theobroma cacao L.: genetic resources in the Dominican Republic. Genet Resour Crop Ev 60(2):605–619. https://doi.org/10.1007/s10722-10012-19860-10724
Cornejo OE et al (2018) Population genomic analyses of the chocolate tree, Theobroma cacao L., provide insights into its domestication process. Communications Biology 1:1–12. https://doi.org/10.1038/s42003-42018-40168-42006
Cosme S, Cuevas HE, Zhang D, Oleksyk TK, Irish BM (2016) Genetic diversity of naturalized cacao (Theobroma cacao L.) in Puerto Rico. Tree Genet Genomes 12:88. https://doi.org/10.1007/s11295-11016-11045-11294
De Wever J, Everaert H, Coppieters F, Rottiers H, Dewettinck K, Lefever S, Messens K (2019) The development of a novel SNP genotyping assay to differentiate cacao clones. Sci Rep_UK 9(1):1–10. https://doi.org/10.1038/s41598-41019-45884-41598
Dinarti D, Susilo AW, Meinhardt LW, Ji K, Motilal LA, Mischke S, Zhang D (2015) Genetic diversity and parentage in farmer selections of cacao from southern Sulawesi, Indonesia revealed by microsatellite markers. Breeding Sci 65:438–446. https://doi.org/10.1270/jsbbs.1265.1438
Earl D, vonHoldt B (2012) Structure harvester: a website and program for visualising structure output and implementing the Evanno method. Conservation Genetic Resources 4:359–361. https://doi.org/10.1007/s12686-12011-19548-12687
Emanuelli F, Lorenzi S, Grzeskowiak L, Catalano V, Stefanini M, Troggio M, Myles S, Martinez-Zapater JM, Zyprian E, Moreira FM, Grando MS (2013) Genetic diversity and population structure assessed by SSR and SNP markers in a large germplasm collection of grape. BMC Plant Biol 13:39. https://doi.org/10.1186/1471-2229-1113-1139
Engels J (1981) Genetic resources of cacao. A catalogue of the CATIE Collection. Tropical Agricultural Research and Training Centre, CATIE, Turrialba, Costa Rica. Technical Bulletin No 7: pp. 191
Enriquez GA Characteristics of cacao “Nacional” of Ecuador. In: International workshop on Conservation, Characterisation and Utilisation of Cocoa Genetic Resources in the 21st century, Cocoa Research Unit (CRU), Trinidad and Tobago, 1992
Evanno G, Regnauts S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-2294x.2005.02553.x
Everaert H et al (2017) Molecular characterization of Vietnamese cocoa genotypes (Theobroma cacao L.) using microsatellite markers. Tree Genet Genomes 13:99. https://doi.org/10.1007/s11295-11017-11180-11296
Fang W, Meinhardt LW, Mischke S, Bellato CM, Motilal L, Zhang D (2013) Accurate determination of genetic identity for a single cacao bean, using molecular markers with a nanofluidic system, ensures cocoa authentication. J Agr Food Chem 62:481–487. https://doi.org/10.1021/jf404402v
Figueira A, Lambert S, Carpenter D, Pires J, Cascardo JCM, Romanczyk L (1997) The similarity of cocoa flavour of fermented seeds from fingerprinted genotypes of Theobroma cacao L. from Brazil and Malaysia. Trop Agr 74:132–139
González LM (1996) Caracterización botánico-agronómica ex situ de 25 clones internacionales de cacao (Theobroma cacao L.). Dissertation, Universidad Nacional Agraria de la Selva
Guest D, Keane P (2007) Vascular-streak dieback: a new encounter disease of cacao in Papua New Guinea and Southeast Asia caused by the obligate basidiomycete Oncobasidium theobromae. Phytopathology 97:1654–1657. https://doi.org/10.1094/phyto-1697-1612-1654
Hamblin MT, Warburton ML, Buckler ES (2007) Empirical comparison of simple sequence repeats and single nucleotide polymorphisms in assessment of maize diversity and relatedness. PLoS One 2(12):e1367. https://doi.org/10.1371/journal.pone.0001367
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332. https://doi.org/10.1111/j.1755-0998.2009.02591.x
ICCO (2019) Fine or flavor cocoa. International Cocoa Organization. https://www.icco.org/about-cocoa/fine-or-flavour-cocoa.html.
ICCO (2020) ICCO quarterly bulletin of cocoa statistics, Vol. XLVI, no.1, cocoa year 2019/20. https://www.icco.org/statistics/production-and-grindings/production.html.
Lam Thi VH et al (2017) Phylogenetic relationships among Vietnamese cocoa accessions using a non-coding region of the chloroplast DNA. Pak J of B 49:133–142
Lefever S et al. (2019) Cost-effective and robust genotyping using double-mismatch allele-specific quantitative PCR. Sci Rep_UK 9(1):2150. https://doi.org/10.1038/s41598-41019-38581-z
Livingstone DS et al (2011) Development of single nucleotide polymorphism markers in Theobroma cacao and comparison to simple sequence repeat markers for genotyping of Cameroon clones. Mol Breeding 27:93–106. https://doi.org/10.1007/s11032-11010-19416-11032
Livingstone DSI et al (2012) Optimization of a SNP assay for genotyping Theobroma cacao under field conditions. Mol Breeding 30:33–52. https://doi.org/10.1007/s11032-11011-19596-11034
Loor RG, Risterucci AM, Courtois B, Fouet O, Jeanneau M, Rosenquist E, Amores F, Vasco A, Medina M, Lanaud C (2009) Tracing the native ancestors of the modern Theobroma cacao L. population in Ecuador. Tree Genet Genomes 5(3):421–433. https://doi.org/10.1007/s11295-11008-10196-11293
Lukman ZD, Susilo AW, Dinarti D, Bailey B, Mischke S, Meinhardt LW (2014) Genetic identity, ancestry and parentage in farmer selections of cacao from Aceh, Indonesia revealed by single nucleotide polymorphism (SNP) markers. Trop Plant Biol 7:133–143. https://doi.org/10.1007/s12042-12014-19144-12046
Martínez IB, Nelson MR, Flamand MC, Bertin P (2015) Genetic diversity and population structure of anciently introduced Cuban cacao Theobroma cacao plants. Genet Resour Crop Ev 62(1):67–84. https://doi.org/10.1007/s10722-10014-10136-z
Monteiro WR, Lopes UV, Clement D (2009) Genetic improvement in cocoa. In: Jain SM, Priyadarshan PM (eds) Breeding plantation tree crops: tropical species. Springer, New York, pp 589–626
Motamayor JC, Lachenaud P, e Mota JWDS, Loor R, Kuhn DN, Brown JS, Schnell RJ (2008) Geographic and genetic population differentiation of the Amazonian chocolate tree (Theobroma cacao L.). PloS One 3(10):e3311. https://doi.org/10.1371/journal.pone.0003311
Motilal LA, Zhang D, Umaharan P, Mischke S, Mooleedhar V, Meinhardt LW (2010) The relic Criollo cacao in Belize-genetic diversity and relationship with Trinitario and other cacao clones held in the International Cocoa Genebank, Trinidad. Plant Genet Resour _C 8(2):106–115. https://doi.org/10.1017/s1479262109990232
Müller BS et al (2015) An operational SNP panel integrated to SSR marker for the assessment of genetic diversity and population structure of the common bean. Plant Mol Biol Rep 33(6):1697–1711. https://doi.org/10.1007/s11105-11015-10866-x
N'Goran JA, Laurent V, Risterucci AM, Lanaud C (1994) Comparative genetic diversity studies of Theobroma cacao L. using RFLP and RAPD markers. Heredity 73(6):589–597. https://doi.org/10.1038/hdy.1994.1166
Padi FK et al (2015) The impact of SNP fingerprinting and parentage analysis on the effectiveness of variety recommendations in cacao. Tree Genet Genomes 11:1–14. https://doi.org/10.1007/s11295-11015-10875-11299
Padi FK, Ofori A, Arthur A (2017) Genetic variation and combining abilities for vigour and yield in a recurrent selection programme for cacao. J Agr Sci 155:444–464. https://doi.org/10.1017/s0021859616000459
Peakall R, Smouse PE (2006) Genalex 6: genetic analysis in excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in excel. Population genetic software for teaching and research - an update. Bioinformatics 28:2537–2539. https://doi.org/10.1093/bioinformatics/bts2460
Pham HDP, Luong MN, Nguyen CT, Valenghi D, Nguyen LG (2016) Feasibility study organic and fairtrade cocoa in Vietnam. Helvetas Swiss Intercooperation. https://www.helvetas.org/Vietnam/wEnglish/Documents/Organic-cocoa-feasibility_final_report_EN.pdf. Accessed on 16 August 2016
Pokou ND, Motamayor JC, Tondo CL, Tahi M, Schnell R (2014) Molecular characterization of resistant accessions of cocoa (Theobroma cocoa L.) to Phytophtora pod rot selected on-farm in Côte-d’Ivoire. Int J Agric Biosci 3(5):236–240
Pound FJ (1938) Cacao and witchbroom disease (Marasmius perniciosus) of South America. With notes on other species of Theobroma. Report by Dr. FJ Pound on a visit to Ecuador, the Amazon Valley, and Colombia. April 1937-April 1938
Pound FJ (1943) Cacao and witches’ broom disease (Marasmius perniciosus). Report on a recent visit to the Amazon territory of Peru, September, 1942-February, 1943
Pound FJ (1945) A note on the cocoa population of South America. In: Cocoa Conference, London, Report and Proceedings of the 1945 Cocoa Conference. pp 131–133
Pridmore R et al (2000) Genomics, molecular genetics and the food industry. J Biotechnol 78:251–258. https://doi.org/10.1016/s0168-1656(1000)00202-00209
Pritchard J, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Rottiers H, Everaert H, Boeckx P, Limba G, Baert G, de Wever J, Maebe K, Smagghe G, Dewettinck K, Messens K (2018) Unraveling the genetic background of the Yangambi Research Center cacao germplasm collection, DR Congo. Tree Genet Genomes 14(5):68. https://doi.org/10.1007/s11295-11018-11285-11296
Saunders JA, Mischke S, Leamy EA, Hemeida AA (2004) Selection of international molecular standards for DNA fingerprinting of Theobroma cacao. Theor Appl Genet 110(1):41–47. https://doi.org/10.1007/s00122-00004-01762-00121
Singh N, Choudhury DR, Singh AK, Kumar S, Srinivasan K, Tyagi RK, Singh NK, Singh R (2013) Comparison of SSR and SNP markers in estimation of genetic diversity and population structure of Indian rice varieties. PLoS One 8(12):e84136. https://doi.org/10.81371/journal.pone.0084136
Soria VJ (1970) Principal varieties of cocoa cultivated in tropical America Cocoa Growers’ Bulletin 15
Susilo AW, Zhang D, Motilal L, Mischke S, Meinhardt LW (2011) Assessing genetic diversity in Java fine-flavor cocoa (Theobroma cacao L.) germplasm by using simple sequence repeat (SSR) markers. Tropical and Agricultural Development 55(2):84–92. https://doi.org/10.22302/iccri.jur.pelitaperkebunan.v22329i22301.22186
Takrama J et al (2012) Applying SNP marker technology in the cacao breeding programme in Ghana. Afr Crop Sci J 20:67–75
Takrama J, Kun J, Reinhardt L, Mischke S, Opoku SY, Padi FK, Zhang D (2014) Verification of genetic identity of introduced cacao germplasm in Ghana using single nucleotide polymorphism (SNP) markers. Afr J Biotechnol 13:2127–21361. https://doi.org/10.25897/ajb22013.13331
Thondaiman V, Rajamani K, Senthil N, Shoba N, Joel AJ (2013) Genetic diversity in cocoa (Theobroma cacao L.) plus trees in Tamil Nadu by simple sequence repeat (SSR) markers. Afr J Biotechnol 12(30):4747–4753. https://doi.org/10.5897/ajb2013.12423
Trinidad D (n.d.) Description of clonal cacao trees (mainly ICS clones). Unpublished notebook, Department of Agriculture, Trinidad. Department of Agriculture, Trinidad, Trinidad. International cocoa germplasm database (ICGD) [online database]. Accessed on 16 July 2019. (2019) http://www.icgd.reading.ac.uk. Accessed 16 july 2019
Turnbull, CJ and Hadley, P (2019) International cocoa germplasm database (ICGD) [online database]. http://www.icgd.reading.ac.uk. Accessed 16 July 2019
Van Inghelandt D, Melchinger AE, Lebreton C, Stich B (2010) Population structure and genetic diversity in a commercial maize breeding program assessed with SSR and SNP markers. Theor Appl Genet 120(7):1289–1299. https://doi.org/10.1007/s00122-00009-01256-00122
Wickramasuriya AM, Dunwell JM (2018) Cacao biotechnology: current status and future prospects. Plant Biotechnol J 16(1):4–17. https://doi.org/10.1111/pbi.12848
Würschum T, Langer SM, Longin CFH, Korzun V, Akhunov E, Ebmeyer E, Schachschneider R, Schacht J, Kazman E, Reif JC (2013) Population structure, genetic diversity and linkage disequilibrium in elite winter wheat assessed with SNP and SSR markers. Theor Appl Genet 126(6):1477–1486
Xahn C (2014) Cocoa seedlings. Company, Cao Nguyen Xanh (CNX) Joint Stock http://www.cnx.com.vn/en/Products/.
Zhang D, Motilal L (2016) Origin, dispersal, and current global distribution of cacao genetic diversity. In: Cacao diseases. Springer, Cham, pp 3–33. https://doi.org/10.1007/1978-1003-1319-24789-24782_24781
Acknowledgments
We would like to thank Chris Turnbull and Andrew Daymond of the International Cacao Quarantine Centre (ICQC) (University of Reading, Reading, UK) for providing the leaves of the reference cultivars. Further, we thank the Center for Forestry Research and Technology Transfer of Nong Lam University (NLU-CEFORTT), Ho Chi Minh City, Vietnam, for granting us access to and assisting us during sampling of the Vietnamese cultivars. We are grateful to the Molecular Biology lab at the Biotechnology research development institute (BiRDI), Can Tho University (CTU), for allowing us to perform the DNA extraction. We also thank Dapeng Zhang (USDA-ARS) and Donald Livingstone (Mars, Inc.) for providing input on possible SNP targets.
Data archiving statement
The generated SSR and SNP datasets for the reference and Vietnamese cultivars (Online Resource 1 and 2) will be available in ICGD (35), hosted by the University of Reading.
Funding
This research was funded by a Lotus Unlimited of Erasmus Mundus Action 2 grant (2013-2535/001-001-EM Action2) to HE and a doctoral fellowship from the Special Research Fund (BOF) of Ghent University (BOF16/DOC/338) to JDW. The funding body did not play a role in data analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
HE wrote the main manuscript and performed the sampling of the Vietnamese samples, guided by TKHT and TLAV. HE and JDW conceived and designed the study, performed the experiments and data analysis equally. SL, JDW, and HE analyzed and interpreted the data from the SNP analysis, and HE and HR from the SSR analysis. KeM and GS gave essential advice on the statistical data analysis. KDW and KaM supported this study by providing the required tools and resources. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by A. De La Torre
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 2
Details of 53 included international reference cultivars showing the accession name (Imperial College Selections (ICS), Pound, MAnaus (MA), Parinari (PA), London Cocoa Trade Estacion Experimental Napo (LCT EEN), Iquitos Mixed Calabacillo (IMC), Amazonas (AMAZ), Nanay (NA), Morona (MO), Estacion Experimental Tropical (EET), Institut Francais du Café et Cacao (IFC), COCAriver (COCA), Criollo, Expedicion Botanico Caqueta (EBC), FSC, Guiana (GU), River KERinioutou (KER), Napo Oriente (NAPO), RioBranco (RB), Sabino Contamana (SCA), SPECimen (SPEC), Ucayali (U) and United Fruitselections (UF)), genetic group according to Turnbull and Hadley (2019) and Motamayor et al. (2008) and the sampling source CEFORTT, the ICQC is mentioned. (PDF 507 kb)
ESM 3
Mean key descriptive parameters calculated for the separate VN clusters and the total VN collection using (1) 14 SSR markers. (2) 42 SNP markers. (3) Combination of 14 SSR markers and 42 SNP markers (PDF 290 kb)
ESM 4
Bayesian clustering of the VN group and correct reference cultivars combining 14 SSR and 42 SNP markers using STRUCTURE (K = 10) (2nd simulation). Nine reference populations and one VN population is shown: (1) Marañon, (2) Criollo/Trinitario, (3) Purús, (4) Nanay, (5) Guiana, (6) Vietnam, (7) Iquitos, (8) Contamana, (9) Amelonado and (10) Curaray. One vertical line represents one cultivar (PDF 347 kb)
ESM 5
Bayesian clustering of the VN group and correct reference cultivars combining 14 SSR and 42 SNP markers using STRUCTURE (K = 12) (3rd simulation). Nine reference populations and one VN population is shown: (1) Marañon, (2) Criollo/Trinitario, (3) Purús, (4) Nanay, (5) Guiana, (6) VN-2, (7) Iquitos, (8) Contamana, (9) Amelonado, (10) Curaray, (11) VN-3 and (12) VN-1. One vertical line represents one cultivar (PDF 175 kb)
Rights and permissions
About this article
Cite this article
Everaert, H., De Wever, J., Tang, T.K.H. et al. Genetic classification of Vietnamese cacao cultivars assessed by SNP and SSR markers. Tree Genetics & Genomes 16, 43 (2020). https://doi.org/10.1007/s11295-020-01439-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-020-01439-x