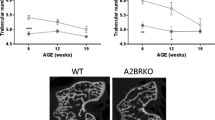
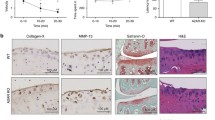
Abstract
There is growing recognition that bone serves important endocrine and immunologic functions that are compromised in several disease states. While many factors are known to affect bone metabolism, recent attention has focused on investigating the role of purinergic signaling in bone formation and regulation. Adenosine is a purine nucleoside produced intracellularly and extracellularly in response to stimuli such as hypoxia and inflammation, which then interacts with P1 receptors. Numerous studies have suggested that these receptors play a pivotal role in osteoblast, osteoclast, and chondrocyte differentiation and function. This review discusses the various ways by which adenosine signaling contributes to bone and cartilage homeostasis, while incorporating potential therapeutic applications of these signaling pathways.



Similar content being viewed by others
References
Fukumoto S, Martin TJ (2009) Bone as an endocrine organ. Trends Endocrinology Metab 20(5):230–236
Olsen BR, Reginato AM, Wang W (2000) Bone development. Annu Rev Cell Dev Biol 16(1):191–220
Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–1084
Auf’mkolk B, Hauschka PV, Schwartz ER (1985) Characterization of human bone cells in culture. Calcif Tissue Int 37(3):228–235
Schwarz M, Harbers K, Kratochwil K (1990) Transcription of a mutant collagen I gene is a cell type and stage-specific marker for odontoblast and osteoblast differentiation. Development 108(4):717–726
Termine JD (1988) Non-collagen proteins in bone. Cell and molecular biology of vertebrate hard tissues:178–190
Turner RT, Colvard DS, Spelsberg TC (1990) Estrogen inhibition of periosteal bone formation in rat long bones: down-regulation of gene expression for bone matrix proteins*. Endocrinology 127(3):1346–1351
Dougall WC (2012) Molecular pathways: osteoclast-dependent and osteoclast-independent roles of the RANKL/RANK/OPG pathway in tumorigenesis and metastasis. Clin Cancer Res 18(2):326–335
Nelson CA, Warren JT, Wang MWH, Teitelbaum SL, Fremont DH (2012) RANKL employs distinct binding modes to engage RANK and the osteoprotegerin decoy receptor. Structure 20(11):1971–1982
Burgess TL, Y-x Q, Kaufman S, Ring BD, Van G, Capparelli C, Kelley M, Hsu H, Boyle WJ, Dunstan CR (1999) The ligand for osteoprotegerin (OPGL) directly activates mature osteoclasts. J Cell Biol 145(3):527–538
Hofbauer LC, Schoppet M (2004) Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA 292(4):490–495
Augat P, Simon U, Liedert A, Claes L (2005) Mechanics and mechano-biology of fracture healing in normal and osteoporotic bone. Osteoporos Int 16(2):S36–S43
Asagiri M, Takayanagi H (2007) The molecular understanding of osteoclast differentiation. Bone 40(2):251–264
Jimi E, Shuto T, Koga T (1995) Macrophage colony-stimulating factor and interleukin-1 alpha maintain the survival of osteoclast-like cells. Endocrinology 136(2):808–811
Hoebertz A, Arnett TR, Burnstock G (2003) Regulation of bone resorption and formation by purines and pyrimidines. Trends Pharmacol Sci 24(6):290–297
Drury AN, Av S-G (1929) The physiological activity of adenine compounds with especial reference to their action upon the mammalian heart. J Physiol 68(3):213
Fredholm BB, Ijzerman AP, Jacobson KA, Linden J, Müller CE (2011) International union of basic and clinical pharmacology. LXXXI. Nomenclature and classification of adenosine receptors—an update. Pharmacol Rev 63(1):1–34
Shimegi S (1996) ATP and adenosine act as a mitogen for osteoblast-like cells (MC3T3-E1). Calcif Tissue Int 58(2):109–113
Manson D, Diamond L, Oudjhane K, Hussain FB, Roifman C, Grunebaum E (2013) Characteristic scapular and rib changes on chest radiographs of children with ADA-deficiency SCIDS in the first year of life. Pediatr Radiol 43(5):589–592
Sauer AV, Mrak E, Hernandez RJ, Zacchi E, Cavani F, Casiraghi M, Grunebaum E, Roifman CM, Cervi MC, Ambrosi A (2009) ADA-deficient SCID is associated with a specific microenvironment and bone phenotype characterized by RANKL/OPG imbalance and osteoblast insufficiency. Blood 114(15):3216–3226
Evans BAJ, Elford C, Pexa A, Francis K, Hughes AC, Deussen A, Ham J (2006) Human osteoblast precursors produce extracellular adenosine, which modulates their secretion of IL-6 and osteoprotegerin. J Bone Miner Res 21(2):228–236
Iser IC, Bracco PA, Gonçalves CEI, Zanin RF, Nardi NB, Lenz G, Battastini AMO, Wink MR (2014) Mesenchymal stem cells from different murine tissues have differential capacity to metabolize extracellular nucleotides. J Cell Biochem 115(10):1673–1682
Burnstock G, Kennedy C (1985) Is there a basis for distinguishing two types of P 2-purinoceptor? Gen Pharmacol 16(5):433–440
Ballarin M, Fredholm BB, Ambrosio S, Mahy N (1991) Extracellular levels of adenosine and its metabolites in the striatum of awake rats: inhibition of uptake and metabolism. Acta Physiol Scand 142(1):97–103
King AE, Ackley MA, Cass CE, Young JD, Baldwin SA (2006) Nucleoside transporters: from scavengers to novel therapeutic targets. Trends Pharmacol Sci 27(8):416–425
Linden J (2005) Adenosine in tissue protection and tissue regeneration. Mol Pharmacol 67(5):1385–1387
Takedachi M, Oohara H, Smith BJ, Iyama M, Kobashi M, Maeda K, Long CL, Humphrey MB, Stoecker BJ, Toyosawa S (2012) CD73‐generated adenosine promotes osteoblast differentiation. J Cell Physiol 227(6):2622–2631
Jacobson KA, Gao Z-G (2006) Adenosine receptors as therapeutic targets. Nat Rev Drug Discov 5(3):247–264
Elliott MR, Chekeni FB, Trampont PC, Lazarowski ER, Kadl A, Walk SF, Park D, Woodson RI, Ostankovich M, Sharma P (2009) Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461(7261):282–286
Lazarowski ER (2012) Vesicular and conductive mechanisms of nucleotide release. Purinergic Signalling 8(3):359–373
Rosenthal AK, Gohr CM, Mitton-Fitzgerald E, Lutz MK, Dubyak GR, Ryan LM (2013) The progressive ankylosis gene product ANK regulates extracellular ATP levels in primary articular chondrocytes. Arthritis Res Ther 15(5):R154. doi:10.1186/ar4337
Orriss IR, Knight GE, Utting JC, Taylor SEB, Burnstock G, Arnett TR (2009) Hypoxia stimulates vesicular ATP release from rat osteoblasts. J Cell Physiol 220(1):155–162
Brandao-Burch A, Burnstock G, Arnett TR, Orriss IR (2011) The P2X7 receptor is an important regulator of extracellular ATP levels. Bone 48:S131
MacDonald PE, Braun M, Galvanovskis J, Rorsman P (2006) Release of small transmitters through kiss-and-run fusion pores in rat pancreatic β cells. Cell Metab 4(4):283–290
Zhang Q, Li Y, Tsien RW (2009) The dynamic control of kiss-and-run and vesicular reuse probed with single nanoparticles. Science 323(5920):1448–1453
Young JD, Yao SYM, Sun L, Cass CE, Baldwin SA (2008) Human equilibrative nucleoside transporter (ENT) family of nucleoside and nucleobase transporter proteins. Xenobiotica 38(7–8):995–1021
Grenz A, Bauerle JD, Dalton JH, Ridyard D, Badulak A, Tak E, McNamee EN, Clambey E, Moldovan R, Reyes G (2012) Equilibrative nucleoside transporter 1 (ENT1) regulates postischemic blood flow during acute kidney injury in mice. J Clin Invest 122(2):693–710
Rose JB, Naydenova Z, Bang A, Ramadan A, Klawitter J, Schram K, Sweeney G, Grenz A, Eltzschig H, Hammond J (2011) Absence of equilibrative nucleoside transporter 1 in ENT1 knockout mice leads to altered nucleoside levels following hypoxic challenge. Life Sci 89(17):621–630
Ii H, Warraich S, Tenn N, Quinonez D, Holdsworth DW, Hammond JR, Dixon SJ, Séguin CA (2016) Disruption of biomineralization pathways in spinal tissues of a mouse model of diffuse idiopathic skeletal hyperostosis. Bone 90:37–49
Warraich S, Bone DBJ, Quinonez D, Ii H, Choi DS, Holdsworth DW, Drangova M, Dixon SJ, Séguin CA, Hammond JR (2013) Loss of equilibrative nucleoside transporter 1 in mice leads to progressive ectopic mineralization of spinal tissues resembling diffuse idiopathic skeletal hyperostosis in humans. J Bone Miner Res 28(5):1135–1149
Daniels G, Ballif BA, Helias V, Saison C, Grimsley S, Mannessier L, Hustinx H, Lee E, Cartron J-P, Peyrard T (2015) Lack of the nucleoside transporter ENT1 results in the Augustine-null blood type and ectopic mineralization. Blood 125(23):3651–3654
Hinton DJ, McGee-Lawrence ME, Lee MR, Kwong HK, Westendorf JJ, Choi D-S (2014) Aberrant bone density in aging mice lacking the adenosine transporter ENT1. PLoS One 9(2):e88818
Mishina M, Kimura Y, Naganawa M, Ishii K, Oda K, Sakata M, Toyohara J, Kobayashi S, Katayama Y, Ishiwata K (2012) Differential effects of age on human striatal adenosine A1 and A2A receptors. Synapse 66(9):832–839
Burnstock G (1976) Purinergic receptors. J Theor Biol 62(2):491–503
Tucker AL (1993) Short review cloned receptors and cardiovascular responses to adenosine. Cardiovasc Res 27:62–61
Verzijl D, Ijzerman AP (2011) Functional selectivity of adenosine receptor ligands. Purinergic Signalling 7(2):171–192
Kim S-H, Kim Y-K, Park H-W, Kim S-H, Kim S-H, Ye Y-M, Min K-U, Park H-S (2009) Adenosine deaminase and adenosine receptor polymorphisms in aspirin-intolerant asthma. Respir Med 103(3):356–363
Tang Z, Diamond MA, Chen JM, Holly TA, Bonow RO, Dasgupta A, Hyslop T, Purzycki A, Wagner J, McNamara DM (2007) Polymorphisms in adenosine receptor genes are associated with infarct size in patients with ischemic cardiomyopathy. Clin Pharmacol Ther 82(4):435–440
Hider SL, Thomson W, Mack LF, Armstrong DJ, Shadforth M, Bruce IN (2008) Polymorphisms within the adenosine receptor 2a gene are associated with adverse events in RA patients treated with MTX. Rheumatology 47(8):1156–1159
Hamilton SP, Slager SL, De Leon AB, Heiman GA, Klein DF, Hodge SE, Weissman MM, Fyer AJ, Knowles JA (2004) Evidence for genetic linkage between a polymorphism in the adenosine 2A receptor and panic disorder. Neuropsychopharmacol 29(3):558–565
Kara FM, Chitu V, Sloane J, Axelrod M, Fredholm BB, Stanley ER, Cronstein BN (2010) Adenosine A1 receptors (A1Rs) play a critical role in osteoclast formation and function. FASEB J 24(7):2325–2333
He W, Wilder T, Cronstein BN (2013) Rolofylline, an adenosine A1 receptor antagonist, inhibits osteoclast differentiation as an inverse agonist. Br J Pharmacol 170(6):1167–1176
He W, Cronstein BN (2012) Adenosine A1 receptor regulates osteoclast formation by altering TRAF6/TAK1 signaling. Purinergic Signalling 8(2):327–337
Kara FM, Doty SB, Boskey A, Goldring S, Zaidi M, Fredholm BB, Cronstein BN (2010) Adenosine A1 receptors regulate bone resorption in mice: Adenosine A1 receptor blockade or deletion increases bone density and prevents ovariectomy‐induced bone loss in adenosine A1 receptor–knockout mice. Arthritis Rheum 62(2):534–541
Mediero A, Kara FM, Wilder T, Cronstein BN (2012) Adenosine A 2A receptor ligation inhibits osteoclast formation. Am J Pathol 180(2):775–786
Mediero A, Perez‐Aso M, Cronstein BN (2013) Activation of adenosine A2A receptor reduces osteoclast formation via PKA‐and ERK1/2‐mediated suppression of NFkB nuclear translocation. Br J Pharmacol 169(6):1372–1388
Mediero A, Perez-Aso M, Cronstein BN (2013) Activation of adenosine A2A receptor reduces osteoclast formation via PKA- and ERK1/2-mediated suppression of NFkappaB nuclear translocation. Br J Pharmacol 169(6):1372–1388. doi:10.1111/bph.12227
Miyazaki T, Katagiri H, Kanegae Y, Takayanagi H, Sawada Y, Yamamoto A, Pando MP, Asano T, Verma IM, Oda H (2000) Reciprocal role of ERK and NF-kB pathways in survival and activation of osteoclasts. J Cell Biol 148(2):333–342
Saulnier N, Guihard S, Holy X, Decembre E, Jurdic P, Clay D, Feuillet V, Pagès G, Pouysségur J, Porteu F (2012) ERK1 regulates the hematopoietic stem cell niches. PLoS One 7(1):e30788
Pellegatti P, Falzoni S, Donvito G, Lemaire I, Di Virgilio F (2011) P2X7 receptor drives osteoclast fusion by increasing the extracellular adenosine concentration. FASEB J 25(4):1264–1274
Mediero A, Frenkel SR, Wilder T, He W, Mazumder A, Cronstein BN (2012) Adenosine A2A receptor activation prevents wear particle-induced osteolysis. Sci Transl Med 4(135):135ra165
Mediero A, Perez‐Aso M, Wilder T, Cronstein BN (2015) Brief report: methotrexate prevents wear particle–induced inflammatory osteolysis in mice via activation of adenosine A2A receptor. Arthritis Rheumatol 67(3):849–855
Teramachi J, Kukita A, Li Y-J, Ushijima Y, Ohkuma H, Wada N, Watanabe T, Nakamura S, Kukita T (2011) Adenosine abolishes MTX-induced suppression of osteoclastogenesis and inflammatory bone destruction in adjuvant-induced arthritis. Lab Investig 91(5):719–731
Rath-Wolfson L, Bar-Yehuda S, Madi L, Ochaion A, Cohen S, Zabutti A, Fishman P (2006) IB-MECA, an A. Clin Exp Rheumatol 24:400–406
Varani K, Vincenzi F, Targa M, Paradiso B, Parrilli A, Fini M, Lanza G, Borea PA (2013) The stimulation of A3 adenosine receptors reduces bone-residing breast cancer in a rat preclinical model. Eur J Cancer 49(2):482–491
Panjehpour M, Karami-Tehrani F (2007) Adenosine modulates cell growth in the human breast cancer cells via adenosine receptors. Oncol Res 16(12):575–585
Mediero A, Cronstein BN (2013) Adenosine and bone metabolism. Trends Endocrinol Metab 24(6):290–300
Carroll SH, Ravid K (2013) Differentiation of mesenchymal stem cells to osteoblasts and chondrocytes: a focus on adenosine receptors. Expert Reviews in Molecular Medicine 15. doi:10.1017/erm.2013.2
Gharibi B, Abraham AA, Ham J, Evans BAJ (2012) Contrasting effects of A1 and A2b adenosine receptors on adipogenesis. Int J Obes 36(3):397–406
Gharibi B, Abraham AA, Ham J, Evans BAJ (2011) Adenosine receptor subtype expression and activation influence the differentiation of mesenchymal stem cells to osteoblasts and adipocytes. J Bone Miner Res 26(9):2112–2124
D’Alimonte I, Nargi E, Lannutti A, Marchisio M, Pierdomenico L, Costanzo G, Iorio PD, Ballerini P, Giuliani P, Caciagli F, Ciccarelli R (2013) Adenosine A1 receptor stimulation enhances osteogenic differentiation of human dental pulp-derived mesenchymal stem cells via WNT signaling. Stem Cell Res 11(1):611–624. doi:10.1016/j.scr.2013.04.002
D’Alimonte I, Nargi E, Lannutti A, Marchisio M, Pierdomenico L, Costanzo G, Di Iorio P, Ballerini P, Giuliani P, Caciagli F (2013) Adenosine A1 receptor stimulation enhances osteogenic differentiation of human dental pulp-derived mesenchymal stem cells via WNT signaling. Stem Cell Res 11(1):611–624
Eijken M, Meijer IMJ, Westbroek I, Koedam M, Chiba H, Uitterlinden AG, Pols HAP, Van Leeuwen J (2008) Wnt signaling acts and is regulated in a human osteoblast differentiation dependent manner. J Cell Biochem 104(2):568–579
van der Horst G, van der Werf SM, Farih‐Sips H, van Bezooijen RL, Löwik CWGM, Karperien M (2005) Downregulation of Wnt signaling by increased expression of Dickkopf‐1 and‐2 is a prerequisite for late‐stage osteoblast differentiation of KS483 cells. J Bone Miner Res 20(10):1867–1877
De Mattei M, Caruso A, Traina GC, Pezzetti F, Baroni T, Sollazzo V (1999) Correlation between pulsed electromagnetic fields exposure time and cell proliferation increase in human osteosarcoma cell lines and human normal osteoblast cells in vitro. Bioelectromagnetics 20(3):177–182
Vincenzi F, Targa M, Corciulo C, Gessi S, Merighi S, Setti S, Cadossi R, Goldring MB, Borea PA, Varani K (2013) Pulsed electromagnetic fields increased the anti-inflammatory effect of A 2A and A 3 adenosine receptors in human T/C-28a2 Chondrocytes and hFOB 1.19 Osteoblasts. PLoS One 8(5):e65561
Mediero A, Wilder T, Perez-Aso M, Cronstein BN (2015) Direct or indirect stimulation of adenosine A2A receptors enhances bone regeneration as well as bone morphogenetic protein-2. FASEB J 29(4):1577–1590
He W, Mazumder A, Wilder T, Cronstein BN (2013) Adenosine regulates bone metabolism via A1, A2A, and A2B receptors in bone marrow cells from normal humans and patients with multiple myeloma. FASEB J 27(9):3446–3454
Glassman SD, Howard J, Dimar J, Sweet A, Wilson G, Carreon L (2011) Complications with recombinant human bone morphogenic protein-2 in posterolateral spine fusion: a consecutive series of 1037 cases. Spine 36(22):1849–1854
Costa MA, Barbosa A, Neto E, Sá‐e‐Sousa A, Freitas R, Neves JM, Magalhães‐Cardoso T, Ferreirinha F, Correia‐de‐Sá P (2011) On the role of subtype selective adenosine receptor agonists during proliferation and osteogenic differentiation of human primary bone marrow stromal cells. J Cell Physiol 226(5):1353–1366
Rao V, Shih Y-RV, Kang H, Kabra H, Varghese S (2015) Adenosine signaling mediates osteogenic differentiation of human embryonic stem cells on mineralized matrices. Front Bioeng Biotechnol 3:185
Trincavelli ML, Daniele S, Giacomelli C, Taliani S, Da Settimo F, Cosimelli B, Greco G, Novellino E, Martini C (2014) Osteoblast differentiation and survival: a role for A 2B adenosine receptor allosteric modulators. Biochim Biophys Acta 1843(12):2957–2966
Ham J, Evans BAJ (2012) An emerging role for adenosine and its receptors in bone homeostasis. Front Endocrinol 3:113
Hsiao EC, Boudignon BM, Chang WC, Bencsik M, Peng J, Nguyen TD, Manalac C, Halloran BP, Conklin BR, Nissenson RA (2008) Osteoblast expression of an engineered Gs-coupled receptor dramatically increases bone mass. Proc Natl Acad Sci 105(4):1209–1214
Hajjawi MOR, Patel JJ, Corcelli M, Arnett TR, Orriss IR (2016) Lack of effect of adenosine on the function of rodent osteoblasts and osteoclasts in vitro. Purinergic Signalling 12(2):247–258
Muir H (1995) The chondrocyte, architect of cartilage. Biomechanics, structure, function and molecular biology of cartilage matrix macromolecules. Bioessays 17(12):1039–1048
Sandell LJ, Aigner T (2001) Articular cartilage and changes in arthritis. An introduction: cell biology of osteoarthritis. Arthritis Res 3(2):107–113
Tesch AM, MacDonald MH, Kollias-Baker C, Benton HP (2002) Effects of an adenosine kinase inhibitor and an adenosine deaminase inhibitor on accumulation of extracellular adenosine by equine articular chondrocytes. Am J Vet Res 63(11):1512–1519
Mistry D, Chambers MG, Mason RM (2006) The role of adenosine in chondrocyte death in murine osteoarthritis and in a murine chondrocyte cell line. Osteoarthr Cartil 14(5):486–495
Schaeffer HJ, Schwender CF (1974) Enzyme inhibitors. 26. Bridging hydrophobic and hydrophilic regions on adenosine deaminase with some 9-(2-hydroxy-3-alkyl) adenines. J Med Chem 17(1):6–8
Nakamachi Y, Koshiba M, Nakazawa T, Hatachi S, Saura R, Kurosaka M, Kusaka H, Kumagai S (2003) Specific increase in enzymatic activity of adenosine deaminase 1 in rheumatoid synovial fibroblasts. Arthritis Rheum 48(3):668–674
Sari RA, Taysi S, Yilmaz O, Bakan N (2002) Correlation of serum levels of adenosine deaminase activity and its isoenzymes with disease activity in rheumatoid arthritis. Clin Exp Rheumatol 21(1):87–90
Tesch AM, MacDonald MH, Kollias-Baker C, Benton HP (2004) Endogenously produced adenosine regulates articular cartilage matrix homeostasis: enzymatic depletion of adenosine stimulates matrix degradation. Osteoarthr Cartil 12(5):349–359
Koolpe M, Pearson D, Benton HP (1999) Expression of both P1 and P2 purine receptor genes by human articular chondrocytes and profile of ligand‐mediated prostaglandin E2 release. Arthritis Rheum 42(2):258–267
Bitto A, Polito F, Irrera N, D’Ascola A, Avenoso A, Nastasi G, Campo GM, Micali A, Bagnato G, Minutoli L (2011) Polydeoxyribonucleotide reduces cytokine production and the severity of collagen‐induced arthritis by stimulation of adenosine A2A receptor. Arthritis Rheum 63(11):3364–3371
Campo GM, Avenoso A, D’Ascola A, Scuruchi M, Prestipino V, Nastasi G, Calatroni A, Campo S (2012) Adenosine A2A receptor activation and hyaluronan fragment inhibition reduce inflammation in mouse articular chondrocytes stimulated with interleukin‐1β. FEBS J 279(12):2120–2133
Mazzon E, Esposito E, Impellizzeri D, Di Paola R, Melani A, Bramanti P, Pedata F, Cuzzocrea S (2011) CGS 21680, an agonist of the adenosine (A2A) receptor, reduces progression of murine type II collagen-induced arthritis. J Rheumatol 38(10):2119–2129
Stefanovic V, Vlahovic P, Savic V, Ardaillou N, Ardaillou R (1993) Adenosine stimulates 5′‐nucleotidase activity in rat mesangial cells via A2 receptors. FEBS Lett 331(1–2):96–100
Morabito L, Montesinos MC, Schreibman DM, Balter L, Thompson LF, Resta R, Carlin G, Huie MA, Cronstein BN (1998) Methotrexate and sulfasalazine promote adenosine release by a mechanism that requires ecto-5′-nucleotidase-mediated conversion of adenine nucleotides. J Clin Investig 101(2):295
Tenenbaum J, Muniz O, Ralph Schumacher H, Good AE, Howell DS (1981) Comparison of phosphohydrolase activities from articular cartilage in calcium pyrophosphate deposition disease and primary osteoarthritis. Arthritis Rheum 24(3):492–500
Acknowledgments
This work was supported by grants from the National Institutes of Health (AR056672, AR068593) and the NYU-HHC Clinical and Translational Science Institute (UL1TR000038).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
Lauren Strazzulla, none. Bruce Cronstein, Consultations: AstraZeneca, Eli Lilly & Co., Bristol-Myers, Squibb. Grants: Celgene, AstraZeneca, Gilead. Equity: CanFite BioPharma. Intellectual Property: Multiple issued and pending patents assigned to NYU School of Medicine.
Rights and permissions
About this article
Cite this article
Strazzulla, L.C., Cronstein, B.N. Regulation of bone and cartilage by adenosine signaling. Purinergic Signalling 12, 583–593 (2016). https://doi.org/10.1007/s11302-016-9527-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-016-9527-2