Abstract
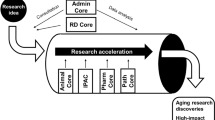
Understanding basic mechanisms of aging holds great promise for developing interventions that prevent or delay many age-related declines and diseases simultaneously to increase human healthspan. However, a major confounding factor in aging research is the heterogeneity of the aging process itself. At the organismal level, it is clear that chronological age does not always predict biological age or susceptibility to frailty or pathology. While genetics and environment are major factors driving variable rates of aging, additional complexity arises because different organs, tissues, and cell types are intrinsically heterogeneous and exhibit different aging trajectories normally or in response to the stresses of the aging process (e.g., damage accumulation). Tackling the heterogeneity of aging requires new and specialized tools (e.g., single-cell analyses, mass spectrometry-based approaches, and advanced imaging) to identify novel signatures of aging across scales. Cutting-edge computational approaches are then needed to integrate these disparate datasets and elucidate network interactions between known aging hallmarks. There is also a need for improved, human cell-based models of aging to ensure that basic research findings are relevant to human aging and healthspan interventions. The San Diego Nathan Shock Center (SD-NSC) provides access to cutting-edge scientific resources to facilitate the study of the heterogeneity of aging in general and to promote the use of novel human cell models of aging. The center also has a robust Research Development Core that funds pilot projects on the heterogeneity of aging and organizes innovative training activities, including workshops and a personalized mentoring program, to help investigators new to the aging field succeed. Finally, the SD-NSC participates in outreach activities to educate the general community about the importance of aging research and promote the need for basic biology of aging research in particular.



Similar content being viewed by others
References
Shock NW. The physiology of aging. Sci Am. 1961;206(1):100–11.
Yashin AI, Arbeev KG, Arbeeva LS, Wu D, Akushevich I, Kovtun M, et al. How the effects of aging and stresses of life are integrated in mortality rates: insights for genetic studies of human health and longevity. Biogerontology. 2016;17(1):89–107. https://doi.org/10.1007/s10522-015-9594-8.
Boisen L, Kristensen P. Confronting cellular heterogeneity in studies of protein metabolism and homeostasis in aging research. Adv Exp Med Biol. 2010;694:234–44. https://doi.org/10.1007/978-1-4419-7002-2_16.
Carnes BA, Olshansky SJ. Heterogeneity and its biodemographic implications for longevity and mortality. Exp Gerontol. 2001;36(3):419–30. https://doi.org/10.1016/s0531-5565(00)00254-0.
Herndon LA, Schmeissner PJ, Dudaronek JM, Brown PA, Listner KM, Sakano Y, et al. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature. 2002;419(6909):808–14. https://doi.org/10.1038/nature01135.
Mendenhall A, Crane MM, Tedesco PM, Johnson TE, Brent R. Caenorhabditis elegans genes affecting interindividual variation in life-span biomarker gene expression. J Gerontol A Biol Sci Med Sci. 2017;72(10):1305–10. https://doi.org/10.1093/gerona/glw349.
Mitnitski A, Howlett SE, Rockwood K. Heterogeneity of human aging and its assessment. J Gerontol A Biol Sci Med Sci. 2017;72(7):877–84. https://doi.org/10.1093/gerona/glw089.
Martin GM. Stochastic modulations of the pace and patterns of ageing: impacts on quasi-stochastic distributions of multiple geriatric pathologies. Mech Ageing Dev. 2012;133(4):107–11. https://doi.org/10.1016/j.mad.2011.09.001.
Tierney MT, Sacco A. Satellite cell heterogeneity in skeletal muscle homeostasis. Trends Cell Biol. 2016;26(6):434–44. https://doi.org/10.1016/j.tcb.2016.02.004.
Rattan SI. Increased molecular damage and heterogeneity as the basis of aging. Biol Chem. 2008;389(3):267–72. https://doi.org/10.1515/BC.2008.030.
Gladyshev VN. On the cause of aging and control of lifespan: heterogeneity leads to inevitable damage accumulation, causing aging; control of damage composition and rate of accumulation define lifespan. Bioessays. 2012;34(11):925–9. https://doi.org/10.1002/bies.201200092.
Gustafsson K, Scadden DT. Growing old in the age of heterogeneity: the perils of shifting clonality. Curr Opin Hematol. 2019;26(4):222–7. https://doi.org/10.1097/MOH.0000000000000513.
Stone ME, Lin J, Dannefer D, Kelley-Moore JA. The continued eclipse of heterogeneity in gerontological research. J Gerontol B Psychol Sci Soc Sci. 2017;72(1):162–7. https://doi.org/10.1093/geronb/gbv068.
Mertens J, Reid D, Lau S, Kim Y, Gage FH. Aging in a dish: iPSC-derived and directly induced neurons for studying brain aging and age-related neurodegenerative diseases. Annu Rev Genet. 2018;52:271–93. https://doi.org/10.1146/annurev-genet-120417-031534.
Bersini S, Schulte R, Huang L, Tsai H, Hetzer MW (2020) Direct reprogramming of human smooth muscle and vascular endothelial cells reveals defects associated with aging and Hutchinson-Gilford progeria syndrome. Elife. 9. https://doi.org/10.7554/eLife.54383
Mertens J, Herdy JR, Traxler L, Schafer ST, Schlachetzki JCM, Bohnke L, et al (2021) Age-dependent instability of mature neuronal fate in induced neurons from Alzheimer’s patients. Cell Stem Cell https://doi.org/10.1016/j.stem.2021.04.004
Lancaster MA, Knoblich JA. Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc. 2014;9(10):2329–40. https://doi.org/10.1038/nprot.2014.158.
Lancaster MA, Renner M, Martin CA, Wenzel D, Bicknell LS, Hurles ME, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501(7467):373–9. https://doi.org/10.1038/nature12517.
Pasca AM, Sloan SA, Clarke LE, Tian Y, Makinson CD, Huber N, et al. Functional cortical neurons and astrocytes from human pluripotent stem cells in 3D culture. Nat Methods. 2015;12(7):671–8. https://doi.org/10.1038/nmeth.3415.
Braganza A, Corey CG, Santanasto AJ, Distefano G, Coen PM, Glynn NW, et al (2019) Platelet bioenergetics correlate with muscle energetics and are altered in older adults. JCI Insight. 5. https://doi.org/10.1172/jci.insight.128248
Fligor CM, Langer KB, Sridhar A, Ren Y, Shields PK, Edler MC, et al. Three-dimensional retinal organoids facilitate the investigation of retinal ganglion cell development, organization and neurite outgrowth from human pluripotent stem cells. Sci Rep. 2018;8(1):14520. https://doi.org/10.1038/s41598-018-32871-8.
Spence JR, Mayhew CN, Rankin SA, Kuhar MF, Vallance JE, Tolle K, et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470(7332):105–9. https://doi.org/10.1038/nature09691.
McCracken KW, Cata EM, Crawford CM, Sinagoga KL, Schumacher M, Rockich BE, et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516(7531):400–4. https://doi.org/10.1038/nature13863.
Kim Y, Kim H, Ko UH, Oh Y, Lim A, Sohn JW, et al (2016) Islet-like organoids derived from human pluripotent stem cells efficiently function in the glucose responsiveness in vitro and in vivo. Sci Rep. 6:35145. https://doi.org/10.1038/srep35145
Zhang HC, Kuo CJ. Personalizing pancreatic cancer organoids with hPSCs. Nat Med. 2015;21(11):1249–51. https://doi.org/10.1038/nm.3992.
Wu F, Wu D, Ren Y, Huang Y, Feng B, Zhao N, et al. Generation of hepatobiliary organoids from human induced pluripotent stem cells. J Hepatol. 2019;70(6):1145–58. https://doi.org/10.1016/j.jhep.2018.12.028.
Morizane R, Lam AQ, Freedman BS, Kishi S, Valerius MT, Bonventre JV. Nephron organoids derived from human pluripotent stem cells model kidney development and injury. Nat Biotechnol. 2015;33(11):1193–200. https://doi.org/10.1038/nbt.3392.
Czerniecki SM, Cruz NM, Harder JL, Menon R, Annis J, Otto EA, et al. High-throughput screening enhances kidney organoid differentiation from human pluripotent stem cells and enables automated multidimensional phenotyping. Cell Stem Cell. 2018;22(6):929–40. https://doi.org/10.1016/j.stem.2018.04.022.
Miller JD, Ganat YM, Kishinevsky S, Bowman RL, Liu B, Tu EY, et al. Human iPSC-based modeling of late-onset disease via progerin-induced aging. Cell Stem Cell. 2013;13(6):691–705. https://doi.org/10.1016/j.stem.2013.11.006.
Hoang P, Wang J, Conklin BR, Healy KE, Ma Z. Generation of spatial-patterned early-developing cardiac organoids using human pluripotent stem cells. Nat Protoc. 2018;13(4):723–37. https://doi.org/10.1038/nprot.2018.006.
Angelidis I, Simon LM, Fernandez IE, Strunz M, Mayr CH, Greiffo FR, et al. An atlas of the aging lung mapped by single cell transcriptomics and deep tissue proteomics. Nat Commun. 2019;10(1):963. https://doi.org/10.1038/s41467-019-08831-9.
Haghverdi L, Buettner F, Theis FJ. Diffusion maps for high-dimensional single-cell analysis of differentiation data. Bioinformatics. 2015;31(18):2989–98. https://doi.org/10.1093/bioinformatics/btv325.
Haghverdi L, Buttner M, Wolf FA, Buettner F, Theis FJ. Diffusion pseudotime robustly reconstructs lineage branching. Nat Methods. 2016;13(10):845–8. https://doi.org/10.1038/nmeth.3971.
La Manno G, Soldatov R, Zeisel A, Braun E, Hochgerner H, Petukhov V, et al. RNA velocity of single cells. Nature. 2018;560(7719):494–8. https://doi.org/10.1038/s41586-018-0414-6.
Schiebinger G, Shu J, Tabaka M, Cleary B, Subramanian V, Solomon A, et al. Optimal-transport analysis of single-cell gene expression identifies developmental trajectories in reprogramming. Cell. 2019;176(4):928–43. https://doi.org/10.1016/j.cell.2019.01.006.
Gao F, Elliott NJ, Ho J, Sharp A, Shokhirev MN, Hargreaves DC. Heterozygous mutations in SMARCA2 reprogram the enhancer landscape by global retargeting of SMARCA4. Mol Cell. 2019;75(5):891–904. https://doi.org/10.1016/j.molcel.2019.06.024.
Thrane K, Eriksson H, Maaskola J, Hansson J, Lundeberg J. Spatially resolved transcriptomics enables dissection of genetic heterogeneity in stage III cutaneous malignant melanoma. Cancer Res. 2018;78(20):5970–9. https://doi.org/10.1158/0008-5472.CAN-18-0747.
Ong SE, Blagoev B, Kratchmarova I, Kristensen DB, Steen H, Pandey A, et al. Stable isotope labeling by amino acids in cell culture, SILAC, as a simple and accurate approach to expression proteomics. Mol Cell Proteomics. 2002;1(5):376–86. https://doi.org/10.1074/mcp.m200025-mcp200.
Welle KA, Zhang T, Hryhorenko JR, Shen S, Qu J, Ghaemmaghami S. Time-resolved analysis of proteome dynamics by tandem mass tags and stable isotope labeling in cell culture (TMT-SILAC) hyperplexing. Mol Cell Proteomics. 2016;15(12):3551–63. https://doi.org/10.1074/mcp.M116.063230.
Zecha J, Meng C, Zolg DP, Samaras P, Wilhelm M, Kuster B. Peptide level turnover measurements enable the study of proteoform dynamics. Mol Cell Proteomics. 2018;17(5):974–92. https://doi.org/10.1074/mcp.RA118.000583.
Fang L, Monroe F, Novak SW, Kirk L, Schiavon CR, Yu SB, et al. Deep learning-based point-scanning super-resolution imaging. Nat Methods. 2021;18(4):406–16. https://doi.org/10.1038/s41592-021-01080-z.
Schiavon CR, Zhang T, Zhao B, Moore AS, Wales P, Andrade LR, et al. Actin chromobody imaging reveals sub-organellar actin dynamics. Nat Methods. 2020;17(9):917–21. https://doi.org/10.1038/s41592-020-0926-5.
Sandhu C, Qureshi A, Emili A. Panomics for precision medicine. Trends Mol Med. 2018;24(1):85–101. https://doi.org/10.1016/j.molmed.2017.11.001.
Huang S, Chaudhary K, Garmire LX (2017) More is better: recent progress in multi-omics data integration methods. Front Genet 8:84. https://doi.org/10.3389/fgene.2017.00084
Lin E, Lane HY (2017) Machine learning and systems genomics approaches for multi-omics data. Biomark Res 5:2. https://doi.org/10.1186/s40364-017-0082-y
Ginsburg GS, Phillips KA. Precision medicine: from science to value. Health Aff (Millwood). 2018;37(5):694–701. https://doi.org/10.1377/hlthaff.2017.1624.
D’Argenio V (2018) The high-throughput analyses era: are we ready for the data struggle? High Throughput. 7(1). https://doi.org/10.3390/ht7010008
Packer J, Trapnell C. Single-cell multi-omics: an engine for new quantitative models of gene regulation. Trends Genet. 2018;34(9):653–65. https://doi.org/10.1016/j.tig.2018.06.001.
Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM, 3rd, et al (2019) Comprehensive integration of single-cell data. Cell. 177(7):1888–902 e21. https://doi.org/10.1016/j.cell.2019.05.031.
Cannoodt R, Saelens W, Saeys Y. Computational methods for trajectory inference from single-cell transcriptomics. Eur J Immunol. 2016;46(11):2496–506. https://doi.org/10.1002/eji.201646347.
Cao J, Spielmann M, Qiu X, Huang X, Ibrahim DM, Hill AJ, et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature. 2019;566(7745):496–502. https://doi.org/10.1038/s41586-019-0969-x.
Mirza B, Wang W, Wang J, Choi H, Chung NC, Ping P (2019) Machine learning and integrative analysis of biomedical big data. Genes (Basel).10(2). https://doi.org/10.3390/genes10020087
Garreta R (2013) MG Learning scikit-learn: machine learning in Python: Packt Publishing Ltd
Kumar A (2019) Pre-processing and modelling using Caret package in R. Int J Comput Appl. 39–42. https://doi.org/10.5120/ijca2018917530
Fleischer JG, Schulte R, Tsai HH, Tyagi S, Ibarra A, Shokhirev MN, et al. (2018)Predicting age from the transcriptome of human dermal fibroblasts. Genome Biol. 19(1):221. https://doi.org/10.1186/s13059-018-1599-6
Drigo RA, e. EG, Tyagi S, Capitanio J, Lyon J, Spigelman AF, Bautista A, Manning Fox JE, Shokhirev M, MacDonald PE, Hetzer MW (2019) Aging of human endocrine pancreatic cell types is heterogeneous and sex-specific. bioRxiv. https://doi.org/10.1101/729541
Kurita M, Araoka T, Hishida T, O’Keefe DD, Takahashi Y, Sakamoto A, et al. In vivo reprogramming of wound-resident cells generates skin epithelial tissue. Nature. 2018;561(7722):243–7. https://doi.org/10.1038/s41586-018-0477-4.
Tang H, Jiang X, Wang X, Wang S, Sofia H, Fox D, et al. Protecting genomic data analytics in the cloud: state of the art and opportunities. BMC Med Genomics. 2016;9(1):63. https://doi.org/10.1186/s12920-016-0224-3.
Schulz WL, Nelson BG, Felker DK, Durant TJS, Torres R. Evaluation of relational and NoSQL database architectures to manage genomic annotations. J Biomed Inform. 2016;64:288–95. https://doi.org/10.1016/j.jbi.2016.10.015.
Wang X, Allen WE, Wright MA, Sylwestrak EL, Samusik N, Vesuna S, et al (2018) Three-dimensional intact-tissue sequencing of single-cell transcriptional states. Science. 361(6400). 10.1126/science.aat5691
Jylhava J, Pedersen NL. Hagg S Biological age predictors. EBioMedicine. 2017;21:29–36. https://doi.org/10.1016/j.ebiom.2017.03.046.
Boguna M, Papadopoulos F, Krioukov D (2010) Sustaining the internet with hyperbolic mapping. Nat Commun. 1:62. https://doi.org/10.1038/ncomms1063
Zhou Y, Smith BH, Sharpee TO (2018) Hyperbolic geometry of the olfactory space. Sci Adv. 4(8):eaaq1458. https://doi.org/10.1126/sciadv.aaq1458
Giusti C, Ghrist R, Bassett DS. Two’s company, three (or more) is a simplex: algebraic-topological tools for understanding higher-order structure in neural data. J Comput Neurosci. 2016;41(1):1–14. https://doi.org/10.1007/s10827-016-0608-6.
Zhou Y, Sharpee TO (2021) Hyperbolic geometry of gene expression. iScience. 24(3):102225. https://doi.org/10.1016/j.isci.2021.102225
Zhou YS (2018) T.O. Using global t-SNE to preserve inter-cluster data structure. bioRxiv. https://doi.org/10.1101/331611.
Acknowledgements
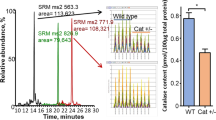
Dr. Malene Hansen was the original director of the SD-NSC Research Development Core and responsible for formulating its overall structure, innovative approaches, and integration with the annual LJAM meeting. Her tireless efforts were also instrumental in getting the entire SD-NSC off the ground successfully and transitioning seamlessly to the new Research Development Core Director Dr. Alessandra Sacco. The authors also wish to acknowledge Lara Avila at the Salk Institute for her steadfast and excellent coordination of SD-NSC workshops and other center activities and events. The authors wish to thank Rafael Arrojo E Drigo and Galena Erikson for allowing the use of preliminary data and other help to generate Fig. 3. This work is supported by the National Institute of Aging of the National Institutes of Health award number P30AG068635. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Shadel, G.S., Adams, P.D., Berggren, W.T. et al. The San Diego Nathan Shock Center: tackling the heterogeneity of aging. GeroScience 43, 2139–2148 (2021). https://doi.org/10.1007/s11357-021-00426-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-021-00426-x