Abstract
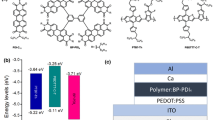
Non-fullerene organic acceptors have attracted increasing attention in recent years. One of the challenges in the synthesis of non-fullerene organic acceptors is to tune the absorption spectrum and molecular frontier orbitals, affording low bandgap molecules with improved absorption of the near-infrared solar photons. In this paper, we present the synthesis, optoelectronic and photovoltaic properties of a series of dimeric perylene diimide (PDI) based non-fullerene acceptors. These PDI dimers are bridged by oligothiophene (T) from 1T to 6T. With the increase of the oligothienyl size, the highest occupied molecular orbital (HOMO) energy is raised from −5.65 to −5.10 eV, while that of the lowest unoccupied molecular orbit (LUMO) is kept constant at −3.84 eV, affording narrow bandgap from 1.81 to 1.26 eV. The absorption from the oligothiophene occurs between 350 and 500 nm, which is complementary to that from its bridged PDI units, leading to a wide spectral coverage from 350 to 850 nm. The optimal dihedral angle between the bridged two perylene planes is dependent on the oligothienyl size, varying from 5° to 30°. The solubility of the dimers depends on the oligothienyl size and can be tuned by the alkyl chains on the bridged thienyl units. The possible applications as the solution-processable non-fullerene organic acceptor is primarily studied using commercial P3HT as the blend donor. The photovoltaic results indicate that 1T, 4T and 6T all yield a higher efficiency of ∼1.2%, whereas 2T, 3T and 5T all give a lower efficiency of <0.5%. The difference in the cell performance is related with the tradeoff between the differences of absorption, HOMO level and film-morphology between these dimers.
Similar content being viewed by others
References
Tang CW. Appl Phys Lett, 1986, 48: 183–185
Brunetti FG, Gong X, Tong M, Heeger AJ, Wudl F. Angew Chem Int Ed, 2010, 49: 532–536
Holcombe TW, Norton JE, Rivnay J, Woo CH, Goris L, Piliego C, Griffini G, Sellinger A, Bredas JL, Salleo A, Fréchet JM. J Am Chem Soc, 2011, 133: 12106–12114
Bloking JT, Han X, Higgs AT, Krastrop JP, Pandey L, Norton JE, Risko C, Chen CE, Brédas JL, Mc Gehee MD, Sellinger A. Chem Mater, 2011, 23: 5484–5490
Ahmed E, Ren G, Kim FS, Hollenbeck EC, Jenekhe SA. Chem Mater, 2011, 23: 4563–4577
Ren G, Ahmed E, Jenekhe SA. Adv Energy Mater, 2011, 1: 946–953
Wang X, Huang JH, Niu ZX, Sun YX, Zhan CL. Tetrahedron, 2014, 70: 4726–4731
Fernando R, Mao Z, Muller E, Ruan F, Sauvé G. J Phys Chem C, 2014, 118: 3433–3442
Zhou TL, Jia T, Kang BN, Li FH, Fahlman M, Wang Y. Adv Energy Mater, 2011, 1: 431–439
Zhou Y, Ding L, Shi K, Dai YZ, Ai N, Wang J, Pei J. Adv Mater, 2012, 24: 957–961
Zhou Y, Dai YZ, Zheng YQ, Wang XY, Wang JY, Pei J. Chem Commun, 2013, 49: 5802–5804
Pho TV, Toma FM, Chabinyc ML, Wudl F. Angew Chem Int Ed, 2013, 52: 1446–1451
Lin YZ, Li YF, Zhan XW. Adv Energy Mater, 2013, 3: 724–728
Shu Y, Lim YF, Li Z, Purushothaman B, Hallani R, Kim JE, Parkin SR, Malliaras GG, Anthony JE. Chem Sci, 2011, 2: 363–368
Winzenberg KN, Kemppinen P, Scholes FH, Collis GE, Shu Y, Singh TB, Bilic A, Forsythb CM, Watkins SE. Chem Commun, 2013, 49: 6307–6309
Zhan CL, Li ADQ. Curr Org Chem, 2011, 15: 1314–1339
Schmidt R, Oh JH, Sun YS, Deppisch M, Krause AM, Radacki K, Braunschweig H, Könemann M, Erk P, Bao ZN, Würthner F. J Am Chem Soc, 2009, 131: 6215–6228
Zhao Y, Guo YL, Liu YQ. Adv Mater, 2013, 25: 5372–5391
Würthner F. Chem Commun, 2004: 1564–1579
Rajaram S, Armstrong PB, Kim BJ, Fréchet JMJ. Chem Mater, 2009, 21: 1775–1777
Lu ZH, Zhang X, Zhan CL, Jiang B, Zhang XL, Chen LL, Yao JN. Phys Chem Chem Phys, 2013, 15: 11375–11385
Zhang X, Lu ZH, Ye L, Zhan CL, Hou JH, Zhang SQ, Jiang B, Zhao Y, Huang JH, Zhang SL, Liu Y, Shi Q, Liu YQ, Yao JN. Adv Mater, 2013, 25: 5791–5797
Jiang B, Zhang X, Zhan CL, Lu ZH, Huang JH, Ding XL, He SG, Yao JN. Polym Chem, 2013, 4: 4631–4638
Yan QF, Zhou Y, Zheng YQ, Pei J, Zhao DH. Chem Sci, 2013, 4: 4389–4394
Jiang W, Ye L, Li XG, Xiao CY, Tan F, Zhao WC, Hou JH, Wang ZH. Chem Commun, 2014, 50: 1024–1026
Shivanna R, Shoaee S, Dimitrov S, Kandappa SK, Rajaram S, Durrant JR, Narayan KS. Energy Environ Sci, 2014, 7: 435–441
Lin YZ, Wang JY, Dai SX, Li YF, Zhu DB, Zhan XW. Adv Energy Mater, 2014, 4: 1400420
Zhang X, Yao JN, Zhan CL. Chem Commun, 2015, 51: 1058–1061
Lin YZ, Wang YF, Wang JY, Hou JH, Li YF, Zhu DB, Zhan XW. Adv Mater, 2014, 26: 5137–5142
Zhang X, Jiang B, Zhang SQ, Hou JH, Yao JN, Zhan CL. Proc SPIE, 2014, 9184: 91840C
Sharenko A, Proctor CM, van der Poll TS, Henson ZB, Nguyen TQ, Bazan GC. Adv Mater, 2013, 25: 4403–4406
Ye TL, Singh R, Butt HJ, Floudas G, Keivanidis PE. ACS Appl Mater Interfaces, 2013, 5: 11844–11857
Singh R, Aluicio-Sarduy E, Kan Z, Ye TL, MacKenzie RCI, Keivanidis PE. J Mater Chem A, 2014, 2: 14348–14353
Zhang XL, Jiang B, Zhang X, Tang AL, Huang JH, Zhan CL, Yao JN. J Phys Chem C, 2014, 118: 24212–24220
Lu ZH, Jiang B, Zhang X, Tang AL, Chen LL, Zhan CL, Yao JN. Chem Mater, 2014, 26: 2907–2914
Zang Y, Li CZ, Chueh CC, Williams ST, Jiang W, Wang ZH, Yu JS, Jen AKY. Adv Mater, 2014, 26: 5708–5714
Zhong Y, Trinh MT, Chen RS, Wang W, Khlyabich PP, Kumar B, Xu QZ, Nam CY, Sfeir MY, Black C, Steigerwald ML, Loo YL, Xiao SX, Ng F, Zhu XY, Nuckolls C. J Am Chem Soc, 2014, 136: 15215–15221
Zhang X, Zhan CL, Yao JN. Chem Mater, 2015, 27: 166–173
Zhao JB, Li YK, Lin HR, Liu YH, Jiang K, Mu C, Ma TX, Lai JYL, Yan H. Energy Environ Sci, 2015, 8: 520–525
Lin YZ, Wang JY, Zhang ZG, Bai HT, Li YF, Zhu DB, Zhan XW. Adv Mater, 2015, 27: 1170–1174
Tan ZA, Zhang WQ, Zhang ZG, Qian DP, Huang Y, Hou JH, Li YF. Adv Mater, 2012, 24: 1476–1481
An ZS, Yu JS, Jones SC, Barlow S, Yoo S, Domercq B, Prins P, Siebbeles LDA, Kippelen B, Marder SR. Adv Mater, 2005, 17: 2580–2583
Zhang X, Pang SF, Zhang ZG, Ding XL, Zhang SL, He SG, Zhan CL. Tetrahedron Lett, 2012, 53: 1094–1097
Zhang X, Zhan CL, Zhang XL, Yao JN. Tetrahedron, 2013, 69: 8155–8160
Li YF. Sci China Chem, 2015, 58: 188
Wu HB. Sci China Chem, 2015, 58: 189
Huang F. Sci China Chem, 2015, 58: 190
Li YF. Sci China Chem, 2015, 58: 191
Author information
Authors and Affiliations
Corresponding author
Additional information
Citation: Zhang X, Yao JN, Zhan CL. Synthesis and photovoltaic properties of low bandgap dimeric perylene diimide based non-fullerene acceptors. Sci China Chem, doi: 10.1007/s11426-015-5485-8
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, X., Yao, J. & Zhan, C. Synthesis and photovoltaic properties of low bandgap dimeric perylene diimide based non-fullerene acceptors. Sci. China Chem. 59, 209–217 (2016). https://doi.org/10.1007/s11426-015-5485-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5485-8