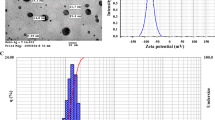
Abstract
Micropropagation has proven to be an effective method for large-scale plant production in a short time and a useful tool for plant breeding. Microbial contamination is one of the most difficult micropropagation challenges, resulting in reduced plant quality and loss of valuable stocks. Therefore, sterilization of culture media is a critical step in plant micropropagation. However, sterilized media might reduce the activity of plant growth regulators and nutritional components of culture media. The sterilization effects of silver nanoparticles (AgNP) on the growth of explants and culture media were examined. The treatment with 250 ppm AgNP for 15 to 20 min of 4-wk-old ex vitro leaves proved optimal for controlling the contamination. Furthermore, the Murashige and Skoog medium containing 4 ppm AgNP resulted in 100% medium disinfection (no contamination) after 4 wk of culture. The plantlets obtained from non-sterilized MS medium (NoM) containing 4 ppm AgNP and 4 g L−1 agar gave similar results as the control medium with 8 g L−1 agar and the absence of AgNP. Large scale culture systems using NoM in large plastic containers of two different sizes (NoM1 and NoM2) could produce quality plantlets. Chrysanthemum plantlets in the NoM1 system showed higher antioxidant enzyme activities of ascorbate peroxidase and superoxide dismutase than plantlets in the autoclaved medium. Furthermore, the plantlets from NoM were better acclimatized under greenhouse conditions than those from the autoclaved medium (AuM) system. The developmental stages (flower buds and blooming time) of NoM1 and NoM2 plantlets, were 1 wk earlier than those from the AuM system. The successful use of AgNP as a sterilizer and as a component of culture media would reduce the cost of micropropagation and improve plants' quality.





Similar content being viewed by others
References
Arab MM, Yadollahi A, Hosseini-Mazinani M (2014) Effects of antimicrobial activity of silver nanoparticles on in vitro establishment of GXN15 (hybrid of almond x peach) rootstock. J Gen Eng Biotech 12:103–110. https://doi.org/10.1016/j.jgeb.2014.10.002
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566. https://doi.org/10.1016/0003-2697(87)90489-1
Brondani GE, de Oliveira LS, Bergonci T, Brondani AE, França FAM, da Silva ALL, Goncalves AN (2013) Chemical sterilization of culture medium: a low-cost alternative to in vitro establishment of plants. Sci For 41:257–264
Cardoso JC, Gerald LTS, Silva JAT (2018) Micropropagation in the twenty-first century. In: Loyola-Vargas V, Ochoa-Alejo N (eds) Plant Cell Culture Protocols. Springer, Switzerland, pp 17–46. https://doi.org/10.1007/978-1-61779-818-4
Carrillo A, Puente ME, Bashan Y (1996) Application of diluted chlorine dioxide to radish and lettuce nurseries in significantly reduced plant development. Ecot Envir Saf 35:57–66. https://doi.org/10.1006/eesa.1996.0081
Cassells AC (1991) Problems in tissue culture: culture contamination. In: Debergh PC, Zimmerman RH (eds) Micropropagation: technology and application. Springer, Dordrecht, pp 31–44. https://doi.org/10.1007/978-94-009-2075-0
Chen C (2016) Cost analysis of plant micropropagation of Phalaenopsis. Plant Cell Tiss Org Cult 126:167–175. https://doi.org/10.1007/s11240-016-0987-4
Duncan DB (1955) Multiple range and multiple F test. Biometrics 11:1–42. https://doi.org/10.2307/3001478
Fakhrfeshani M, Bagheri A, Sharifi A (2012) Disinfecting effects of nano silver fluids in Gerbera (Gerbera jamesonii) capitulum tissue culture. J Biol Environ Sci 6:121–127
Gamborg OL (2002) Plant tissue culture: biotechnology milestones in vitro. Cell Dev Biol - Plant 38:84–92
George EF, De Klerk G-J (2008) The components of plant tissue culture media: I: macro- and micro-nutrients. In: George EF, Hall MA, De Klerk G-J (eds) Plant propagation by tissue culture, 3rd edn. Springer, Dordrecht, pp 65–114
Gupta DS (2011) Role of free radicals and antioxidants in in vitro morphogenesis. In: Gupta DS (ed) Reactive oxygen species and antioxidants in higher plants. CRC Press, New York, pp 229–247
Haq IU, Khan J, Alam M, Khattak MS (1998) In vitro culture of Chrysanthemum Sarhad. J Agric For 14:211–213
Ines M, Krunoslav D, Vesna T, Marija V, Ankica P, Zlatko C, Boris P, Zorica J (2013) In vitro sterilization procedures for micropropagation of Oblaciska sour cherry. J Agric Sci 58:117–126. https://doi.org/10.2298/JAS1302117M
Kim DH, Gopal J, Sivanesan L (2017) Nanomaterials in plant tissue culture: the disclosed and undisclosed. RSC Adv 7:36392–36505
Kim JS, Kuk E, Yu KN, Kim J, Park SJ, Lee HJ, Kim SH, Park YK, Park YH, Hwany CY, Kim YK, Lee SY, Jeong DH, Cho MH (2007) Antimicrobial effects of silver nanoparticles. Nanomedicine 3:95–101. https://doi.org/10.1016/j.nano.2006.12.001
Kole C, Kumar DS, Khodakovskaya MV (eds) (2016) Plant nanotechnology: principles and practices. Springer, Switzerland
Kumar PP, Lakshmanan P, Thorpe TA (1998) Regulation of morphogenesis in plant tissue culture by ethylene in vitro. Cell Develop Biol – Plant 34:94–103
Maiti S, Krishnan D, Barman G, Ghosh SK, Laha JK (2014) Antimicrobial activities of silver nanoparticles synthesized from Lycopersicon esculentum extract. J Anal Sci Technol 5:1–7. https://doi.org/10.1186/S40543-014-0040-3
McDaniel BK, Binder BM (2012) Ethylene Receptor1 (ETR1) is sufficient and has the predominant role in mediating inhibition of ethylene responses by silver in Arabidopsis thaliana. J Biol Chem 287:26094–26103. https://doi.org/10.1074/jbc.M112.383034
Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, Ramírez JT, Yacaman MJ (2005) The bactericidal effect of silver nanoparticles. Nanotechnology 16:2346–2353. https://doi.org/10.1088/0957-4484/16/10/059
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Pais AK, Silva AP, Souza JC, Teixeira SL, Ribeiro JM, Peixoto AR, Paz CD (2016) Sodium hypochlorite sterilization of culture medium in micropropagation of Gerbera hybrid cv. Essandre. Afr J Biotech 15:1995–1998. https://doi.org/10.5897/AJB2016.15405
Proctor AG (1977) Mycological method. In: Collins CH, Lyne PM (eds) Microbiological methods, 4th edn, Butter Worths, UK. https://doi.org/10.1002/jobm.19770170512
Rostami AA, Shahsavar A (2009) Nano-silver particles eliminate the in vitro contaminations of olive 'Mission' explants. Asian J Plant Sci 8:505–509. https://doi.org/10.3923/ajps.2009.505.509
Saha N, Gupta D (2018) Promotion of shoot regeneration of Swertia chirata by biosynthesized silver nanoparticles and their involvement in ethylene interceptions and activation of antioxidant activity. Plant Cell Tissue Organ Cult 134:289–300. https://doi.org/10.1007/s11240-018-1423-8
Sanzari I, Leone A, Ambrosone A (2019) Nanotechnology in plant science: to make a long story short. Front Bioeng Biotechnol 7:1–12. https://doi.org/10.3389/fbioe.2019.00120
Sarmast MK, Salehi H (2016) Silver nanoparticles: an influential element in plant nanobiotechnology. Mol Biotechnol 58:441–449. https://doi.org/10.1007/s12033-016-9943-0
Sharma P, Bhatt D, Zaidi MG, Saradhi PP, Khanna PK, Arora S (2012) Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica juncea. Appl Biochem Biotechnol 167:2225–2233. https://doi.org/10.1007/s12010-012-9759-8
Siddiqui MH, Al-Whaibi MH, Firoz M, Al-Khaishany MY (2015) Role of nanoparticles in plants. In: Siddiqui MH (ed) Nanotechnology and plant sciences. Springer, Basel, pp 19–35
Sondi I, Salopek-Sondi B (2004) Silver nanoparticles as antimicrobial agent: a case study as a model for gram negative bacteria. J Colloid Interface Sci 275:177–182. https://doi.org/10.1016/j.jcis.2004.02.012
Syu YY, Hung JH, Chen JC, Chuang HW (2014) Impacts of size and shape of silver nanoparticles on Arabidopsis plant growth and gene expression. Plant Physiol Biochem 83C:57–64. https://doi.org/10.1016/j.plaphy.2014.07.010
da Silva Teixeira JA, Fukai S (2003) Chrysanthemum organogenesis through thin cell layer technology and plant growth regulator control. Asian J Plant Sci 2:505–514. https://doi.org/10.3923/ajps.2003.505.514
Tung HT, Nam NB, Huy NP, Luan VQ, Hien VT, Phuong TTB, Dung LT, Nhut DT (2018) A system for large scale production of Chrysanthemum using microponics with the supplement of silver nanoparticles under light-emitting diodes. Sci Hortic 232:153–161. https://doi.org/10.1016/j.scienta.2017.12.063
Vargas DP, Formoso RS, Dutra LF, Newton AM, Santos J, Ueno B (2016) Chemical sterilization of in vitro culture for peach rootstock. Colloq Agr 12:1–6
Wang XJ, Hsiao KC (1995) Sugar degradation during autoclaving: effects of duration and solution volume on breakdown of glucose. Physiol Plant 94:415–418. https://doi.org/10.1111/j.1399-3054.1995.tb00947.x
Zhang J, Chen S, Liu R, Jiang J, Chen F, Fang W (2013) Chrysanthemum cutting productivity and rooting ability are improved by grafting. Sci World J 2013:286328. 1-7. https://doi.org/10.1155/2013/286328
Acknowledgments
The authors would like to thank Professor C.C. Chinnappa (University of Calgary, Canada) for critical reading of the manuscript.
Funding
This research is funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 106.01-2019.301.
Author information
Authors and Affiliations
Contributions
Hoang Thanh Tung acquired data and wrote the manuscript. Huynh Gia Bao, Do Manh Cuong, Ha Thi My Ngan, Vu Thi Hien, Vu Quoc Luan, Bui Van The Vinh, Hoang Thi Nhu Phuong, Nguyen Ba Nam, Le Ngoc Trieu, Nguyen Khoa Truong, Pham Nguyen Duc Hoang participated in the interpretation of data and revision for intellectual content. Duong Tan Nhut and Hoang Thanh Tung conceptualized and designed the study. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
The authors declare no competing interests.
Additional information
Editor: Charles Armstrong
Rights and permissions
About this article
Cite this article
Tung, H.T., Bao, H.G., Cuong, D.M. et al. Silver nanoparticles as the sterilant in large-scale micropropagation of chrysanthemum. In Vitro Cell.Dev.Biol.-Plant 57, 897–906 (2021). https://doi.org/10.1007/s11627-021-10163-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-021-10163-7