Abstract
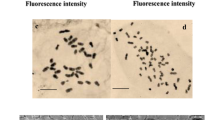
Papaver bracteatum, belonging to Papaveraceae, is considered to be a primary ingredient in the production of codeine and anti-drug drugs (e.g., naloxone and Naltrexone) without the involvement of morphine. For polyploidy induction, papaver seedlings were treated with various colchicine concentrations (0, 0.05, 0.1, 0.2 and 0.5% w/v) in three time durations (24, 48 and 72 h). The effects of polyploidy induction on morphological, physiological, alkaloid production and gene expression levels in the pathway of benzylisoquinoline alkaloids were analyzed. High rates of seedling survival were observed at lower colchicine concentrations (0.05 and 0.1% w/v) versus higher concentrations (0.2 and 0.5% w/v). The best polyploidy treatment to induce P. bracteatum shoot tips was observed in plants treated with 0.2% colchicine for 72 h. P. bracteatum-induced tetraploids showed higher stomata and an increase in leaf area (mm2), chlorophyll (a, b and total) and a decrease in stomata density, leaf length (cm) and petiole length. Flow cytometry and chromosome counting analysis revealed that chromosome number was 2n = 2x = 14 in diploid plants and 2n = 4x = 28 in tetraploid plants. Gene expression analysis through semi-quantitative reverse transcription-polymerase chain reaction (RT-PCR) showed that the transcription level of sat and tydc genes of the benzylisoquinoline biosynthesis pathway was enhanced in tetraploid plants. The tetraploid plants of P. bracteatum also showed 5.86 and 30.55 times higher thebaine and noscapine contents than the diploid plants, respectively. Regarding the higher commercial value of thebaine and noscapine, tetraploids can efficiently be used to improve the thebaine and noscapine production of P. bracteatum.









Similar content being viewed by others
References
Abdoli M, Moieni A, Badi HN (2013) Morphological, physiological, cytological and phytochemical studies in diploid and colchicine-induced tetraploid plants of Echinacea purpurea L. Acta Physiol Plant 35(7):1–9
Adams KL, Wendel JF (2005) Novel patterns of gene expression in polyploidy plants. Trends Genet 10(21):539–543
Adaniya S, Shirai D (2001) In vitro induction of tetraploid ginger (Zingiber officinale Roscoe) and its pollen fertility and germinability. Sci Hort. 88(4):277–287
Ajalin I, Kobza F, Dolezel J (2002) Ploidy identification of doubled chromosome number plants in Viola × witrockiana Gams. M1- generation. Hortic Sci. 29(1):35–40
Albuzio A, Spettoli P, Cacco G (2006) Changes in gene expression from diploid to autotetraploid status of Lycopersicon esculentum. Physiol Plant 44:77–80
Allum JF, Bringloe DH, Roberts AV (2007) Chromosome doubling in a Rosa rugosa Thunb. Hybrid by exposure of in vitro nodes to oryzalin: the effects of node length, oryzalin concentration and exposure time. Plant Cell Rep 26:1977–1984
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant physiol. 24:1–15
Berkov S (2001) Size and alkaloid content of seeds in induced autotetraploids of Datura innoxia, Datura stramonium and Hyoscyamus niger. Pharm Biol. 39(5):329–331
Berkov S, Philipov S (2002) Alkaloid production in diploid and autotetraploid plants of Datura stramonium. Pharm Biol. 40(8):617–621
Caruso I, Lepore L, De Tommasi N, Dal Piaz F, Frusciante L, Aversano R, Garramone R, Carputo D (2011) Secondary metabolite profile in induced tetraploids of wild Solanum commersonii Dun. Chem Biodivers 8(12):2226–2237
Cheng ZM, Korban S (2011) In vitro ploidy manipulation in the genomics era. Plant Cell, Tissue Organ Cult 104:281–282
Coffman CB, Bare CE, Gentner WA (1975) Thebaine variations between germplasm sources within one collection of Papaver bracteatum. B narcotics. 27:41–46
Dhooghe E, Van Laere K, Eeckhaut T, Leus V, Van Huylenbroeck J (2011) Mitotic chromosome doubling of plant tissues in vitro. Plant Cell Tiss Organ Cult 104:359–373
Facchini PJ, Penzes-Yost C, Samanani N, Kowalchuk N (1998) Expression Patterns Conferred by Tyrosine/Dihydroxyphenylalanine Decarboxylase Promoters from Opium Poppy Are Conserved in Transgenic Tobacco. Plant Physiol 118(1):69–81
Gao S, Zhu D, Cai Z, Xu D (1996) Autotetraploid plants from colchicine-treated bud culture of Salvia miltiorrhiza Bge. Plant Cell Tiss Organ Cult 47(1):73–77
Gao SL, Chen BJ, Zhu DN (2002) In vitro production and identification of autotetraploids of Scutellaria baicalensis. Plant Cell Tiss Organ Cult 70:289–293
Ghotbi Ravandi E, Rezanejad F, Zolala J, Dehghan E (2013) The effects of chromosome-doubling on selected morphological and phytochemical characteristics of Cichorium intybus L. J Hortic Sci Biotechnol. 88(8):701–709
Griesbach RJ (1990) Colchicine-induced polyploidy in Eustoma grandiflorum. Sci Hort. 25:1284–1286
Grothe T, Lenz R, Kutchan TM (2001) Molecular characterization of the salutaridinol 7-O-acetyltransferase involved in morphine biosynthesis in opium poppy Papaver somniferum. J Biol Chem 276(33):30717–30723
Guo M, Davis D, James A (1996) Birchler dosage effects on gene expression in a maize ploidy series. Genet Soc Am 142(4):1349–1355
Hamill S, Smith M, Dodd W (1992) In vitro induction of banana autotetraploids by colchicine treatment of micropropagated diploids. Aust J Bot 40(6):887–896
He M, Jiang L, Ren Z, Wang G, Wang J (2016) Noscapine targets EGFRp-Tyr1068 to suppress the proliferation and invasion of MG63 cells. Sci. Rep. https://doi.org/10.1038/srep37062
Javadian N, Karimzadeh G, Sharifi M, Moieni A, Behmanesh M (2017) In vitro polyploidy induction: changes in morphology, podophyllotoxin biosynthesis, and expression of the related genes in Linum album (Linaceae). Planta. https://doi.org/10.1007/s00425-017-2671-2
Kaensaksiri T, Soontornchainaksaeng P, Soonthornchareonnon N, Prathanturarug S (2011) In vitro induction of polyploidy in Centella asiatica (L.) Urban. Plant Cell Tiss Organ Cult 107(2):187–194
Kamada H, Okamura N, Satake M, Harada H, Shimomura K (1986) Alkaloid production by hairy root cultures in Atropa belladonna. Plant Cell Rep 5(4):239–242
Kerr D, Dietze P (2008) Kelly AM Intranasal naloxone for the treatment of suspected heroin overdose. Addiction 103(3):379–386
Kettenes-van de Bosch JJ, Salemink CA, Khan I (1981) Biological activity of the alkaloids of Papaver bracteatum Lind. J Ethnopharmacol 3(1):21–38
Kim CS, Park JB, Kim KJ, Chang SJ, Ryoo SW, Jeon BH (2002) Effect of Korea red ginseng (Panax ginseng) on cerebral blood flow and superoxide production. Acta Pharmacol. 23:1152–1156
Kondorosi E, Roudier F, Gendrau E (2000) Plant cell-size: growing by ploidy. Curr Opin Plant Biol 3:488–492
Lin X, Zhou Y, Zhang J, Lu X, Zhang F, Shen Q, Wu S, Chen Y, Wang T, Tang K (2011) Enhancement of artemisinin content in tetraploid Artemisia annua plants by modulating the expression of genes in artemisinin biosynthetic pathway. Biotechnol Appl Biochem 58(1):50–57
Liu S, Chen S, Chen Y, Guan Z, Yin D, Chen F (2011) In vitro induced tetraploid of Dendranthema nankingense (Nakai) Tzvel. Shows an improved level of abiotic stress tolerance. Sci Hort. 127(3):411–419
Machara A, Werner L, Ann Endoma-Arias M, Cox DF, Hudlicky T (2012) Improved synthesis of buprenorphine from thebaine and/or oripavine via palladium-catalyzed N-demethylation/acylation and/or concomitant O-demethylation. Adv Synth Catal 354:613–626
Madani H, Hosseini B, Dehghan E, Rezaei- chiyaneh E (2015) Enhanced production of scopolamine in induced autotetraploid plants of Hyoscyamus reticulatus L. Acta Physiol Plant 37:55
Mathura S, Fossey A, Beck SL (2006) Comparative study of chlorophyll content in diploid and tetraploid black wattle (Acacia mearnsii). Forestry 79:381–388
Milo J, Levy A, Palevitch D, Ladizinsky G (1987) Thebaine content and yield in induced tetraploid and triploid plants of Papaver bracteatum Lindl. Euphytica 36:361–367
Milo J, Levy A, Ladizinsky G, Palevitch D (1988) Phylogenetic and genetic studies in Papaver section Oxytona: cytogenetics, isozyme analysis and chloroplast DNA variation. Theor Appl Genet 75:795–802
Mishra BK, Pathak S, Sharma A, Trivedi PK, Shukla S (2010) Modulated gene expression in newly synthesized auto tetraploid of Papaver somniferum L. S Afr J Bot 76:447–452
Neubauer D, Mothes K (1963) Uber Papaver bracteatum I. Mitteilung, ein neuer weg zur gewinnung von morphinanen auf pflanztichef rohstoffbasis. Planta Med 11:387–391
Omidbaigi R, Mirzaee M, Hassani ME, Sedghi Moghadam M (2010a) Induction and identification of polyploidy in basil (Ocimum basilicum L.) medicinal plant by colchicine treatment. Int J Plant Prod. 4:1735–8043
Omidbaigi R, Yavari S, Hassani ME, Yavari S (2010b) Induction of autotetraploidy in dragonhead (Dracocephalum moldavica L.) by colchicine treatment. J. Fruit Ornam. Plant Res. 18:23–35
Palevitch D, Levy A (1983) Cultural and genetic factors affecting thebaine yield in Papaver bracteatum Lindl. Acta Hort 132:189–195
Rey H, Sansberro B, Collavino M, Davina J, Gonzalez A, Mroginski L (2002) Colchicine, trifluralin and oryzalin promoted development of somatic embryos in Ilex paraguariensis (Aquifoliaceae). Euphytica 123:49–56
Rinner U, Hudlicky T (2012) Synthesis of morphine alkaloids and derivatives. Top Curr Chem 309:33–66
Rowson JM (1949) Increased alkaloid contents of induced polyploid of Datura. Nature Rev Genet. 154:81–82
Rubuluza T, Nikolova RV, Smith MT, Hannweg K (2007) In vitro induction oftetraploids in Colophospermum mopane by colchicine. South Afri J Bot. 73:259–261
Sadat Noori SA, Norouzi M, Karimzadeh G, Shirkool K, Niazian M (2017) Effect of colchicine-induced polyploidy on morphological characteristics and essential oil composition of ajowan (Trachyspermum ammi L.). Plant Cell Tiss Organ Cult 130(3):543–551
Saminathan T, Nimmakayala P, Manohar S, Malkaram S, Almeida A, Cantrell R, Tomason Y, Abburi L, Rahman MA, Vajja VG, Khachane A, Kumar B, Rajasimha HK, Levi A, Wehner T, Reddy UK (2015) Differential gene expression and alternative splicing between diploid and tetraploid watermelon. J Exp Bot 66(5):1369–1385
Seddigh M, Jolliff GD, Calhoun W, Crane JM (1982) Papaver bracteatum, potential commercial source of codeine. Econ Bot 36:433–441
Shahriari-Ahmadi F, Dehghan E, Farsi M, Azizi M (2009) Tetraploid Induction of Hyoscyamus muticus L. using Colchicine Treatment. Pak J Biol Sci 11(24):2653–2659
Swanson CP (1957) Cytol cytogenet. Carl P, Swanson, p 596
Talebi SF, Saharkhiz MJ, Jafarkhani Kermani M, Sharafi Y, Raouf Fard F (2016) Effect of different antimitotic agents on polyploid induction of anise hyssop (Agastache foeniculum L.). Inte J Cytol Cytosyst Cytogen. 70(2):184–193
Tang ZQ, Chen DL, Song ZJ, He YC, Cai DT (2010) In vitro induction and identification of tetraploid plants of Paulownia tomentosa. Plant Cell Tiss Organ Cult 102(2):213–220
Tarkesh Esfahani S, Karimzadeh G, Naghavi MR (2016) 2C DNA value of persian poppy (Papaver bracteatum Lindl.) medicinal plant as revealed by flow cytometry analysis; a quick effective criteria for distinguishing unidentified papaver species. Int J Adv Biotechnol Res. 7(2):573–578
Wallaart TE, Pras N, Quax WJ (1999) Seasonal variations of artemisinin and its biosynthetic precursors in tetraploid Artemisia annua plants compared with the diploid wild-type. Planta Med 65:723–728
Wendel JF, Doyle JJ (2005) Polyploidy and evolution in plants. In: Henry RJ (ed) Plant diversity and evolution. CABI Publishing, Wallingford, UK, pp 97–117
Zahedi AA, Hosseini B, Fattahi M, Dehghan E, Parastar H, Madani H (2014) Overproduction of valuable methoxylated flavones in induced tetraploid plants of Dracocephalum kotschyi Boiss. Bot Stud. 55:22
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Y. Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Madani, H., Hosseini, B., Karimzadeh, G. et al. Enhanced thebaine and noscapine production and modulated gene expression of tyrosine/dopa decarboxylase and salutaridinol 7-O-acetyltransferase genes in induced autotetraploid seedlings of Papaver bracteatum Lindl. Acta Physiol Plant 41, 194 (2019). https://doi.org/10.1007/s11738-019-2984-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-019-2984-9