Abstract
High-temperature isothermal steam oxidation kinetic parameters of several ferritic alloys were determined by thermogravimetric analysis. The oxidation kinetic constant (k) was measured as a function of temperature from 900°C to 1200°C. The results show a marked increase in oxidation resistance compared to reference Zircaloy-2, with kinetic constants 3–5 orders of magnitude lower across the experimental temperature range. The results of this investigation supplement previous findings on the properties of ferritic alloys for use as candidate cladding materials and extend kinetic parameter measurements to high-temperature steam environments suitable for assessing accident tolerance for light water reactor applications.



Similar content being viewed by others
Notes
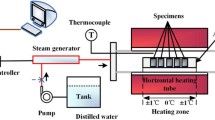
Testing was executed in Los Alamos, New Mexico, USA. The altitude of the laboratory results in a significant departure from 1 atm in any system maintained at local atmospheric pressure.
References
R. Gauntt, D. Kalinich, J. Cardoni, J. Phillips, A. Goldmann, S. Pickering, M. Francis, K. Robb, L. Ott, D. Wang, C. Smith, S. St.Germain, D. Schwieder, and C. Phelan, SAND2012-6173, Sandia National Laboratory (SNL) (2012)
Y. Yukinori, B.A. Pint, K.A. Terrani, Y. Yang, K.G. Field, and L.L. Snead, J. Nucl. Mater. 467, 703 (2015).
R.B. Rebak, P.L. Andresen, Y.J. Kim, and E.J. Dolley. IAEA TECDOC SERIES 79 (2016)
K. Barrett and S. Bragg-Sitton. No. INL/EXT-12-27090. Idaho National Laboratory (INL) (2012)
B.A. Pint, A.U. Kinga, and A.T. Kurt, Mater. High Temp. 32, 28 (2015).
S.J. Zinkle, K.A. Terrani, J.C. Gehin, L.J. Ott, and L.L. Snead, J. Nucl. Mater. 448, 374 (2014).
R.A. Shapiro, M.Y. Ian, and M. Fratoni, Trans. Am. Nucl. Soc. 109, 1351 (2013).
J.D. Hales and K.A. Gamble. No. INL/CON-14-33932. Idaho National Laboratory, (INL) (2015)
N.R. Brown and M. Todosow. No. BNL–107219-2014-CP. Brookhaven National Laboratory (BNL) (2014)
T. Cheng, J. Keiser, M. Brady, K.A. Terrani, and B.A. Pint, J. Nucl. Mater. 427, 396 (2012).
K.A. Terrani, S. Zinkle, and L.L. Snead, J. Nucl. Mater. 448, 420 (2014).
B.A. Pint, K.A. Terrani, M. Brady, T. Cheng, and J.J. Keiser, Nucl. Mater. 440, 420 (2013).
I.G. Write, B.A. Pint, and P.F. Tortorelli, Oxid. Metals 55, 333 (2001).
D. Sebastien, R.V. Put, and B.A. Pint, Oxid. Metals. 79, 627 (2013).
E.S. Wood, K.A. Terrani, and A.T. Nelson, J. Nucl. Mater. 477, 228 (2016).
R. Carter, J. Chem. Phys. 34, 2010 (1961).
C. Wagner, Z. Phys. Chem. B 21, 25 (1933).
J.V. Cathcart, R.E. Pawel, P.A. McKee, R.E. Druschel, G.J. Yurek, and J.J. Campbell. ORNL/NUREG-17,Oak Ridge National Laboratory (1977)
B.A. Pint, K.A. Terrani, Y. Yamamoto, and L.L. Snead, Metal. Mater. Trans. E. 2, 190 (2015).
L. Tan and B.A. Pint, Oakridge National Laboratory, ORNL/TM-2017/288 (2017)
D. Laverde, T. Comez-Acebo, and F. Castro, Corros. Sci. 46, 613 (2004).
G. Ostberg, Aktibolaget Atomenergi (Sweden: Stockholm, 1960).
C.S. Tedmon Jr., J. Electrochem. Soc. 113, 766 (1966).
N. Li, S.S. Parker, E.S. Wood, and A.T. Nelson, J. Nucl. Mater. Submitted September (2017)
B.A. Pint and J.W. McMurray, ORNL/LTR-2016/425 (2016)
A.T. Nelson, E.S. Sooby, Y.J. Kim, B. Cheng, and S.A. Maloy, J. Nucl. Mater. 448, 441 (2014)
Acknowledgements
Portions of this work were supported by the US Department of Energy, Office of Nuclear Energy Fuel Cycle Research and Development Program. This work was performed at Los Alamos National Laboratory under the auspices of the US Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parker, S.S., White, J., Hosemann, P. et al. Oxidation Kinetics of Ferritic Alloys in High-Temperature Steam Environments. JOM 70, 186–191 (2018). https://doi.org/10.1007/s11837-017-2639-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-017-2639-5