Abstract
Purpose
Transient hypocalcaemia after thyroid surgery and its possible predictors have not been extensively described in the elderly. This study aimed to establish the frequency of postsurgical transient hypocalcaemia according to the extent of thyroid surgery in older adults and to assess mineral metabolism biochemical parameters as its predictors.
Methods
All patients ≥60 years undergoing thyroid surgery were prospectively included. Type of surgery (hemithyroidectomy(HT) or total thyroidectomy(TT)); and preoperative 25OH Vitamin D (25OHD) and pre and 6 (only TT), 24 h and 6 months postsurgical serum levels of calcium, magnesium, phosphate and parathormone (PTH) were considered. Postsurgical hypoparathyroidism (hPTpost) was defined at PTH levels ≤11 pg/mL.
Results
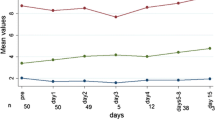
Out of 46 patients (87% female), age (mean ± SD) 70.1 ± 6.2 years, 24 h postsurgical hypocalcaemia was found in ten patients (22%). In 25 (54%) TT patients, 36% and 16% had postsurgical hypocalcaemia at 6 and 24 h respectively; 28% hPTpost but no definitive hPT was recorded and 44% had 25OHD deficiency. Lower 24 h magnesium levels were found in those TT patients with 24 h hypocalcaemia (1.6 ± 0.1 vs 1.9 ± 0.1 mg/dL (p = 0.005)). Among 21 (46%) HT patients, 28.6% had 24 h postsurgical hypocalcaemia; 9.5% had hPTpost. A positive correlation was observed between preoperative 25OHD and 24 h calcaemia (r:0.51,p = 0.02). 43% of the patients were 25OHD deficient, in whom 55% had 24 h hypocalcaemia vs only 9% in the 25OHD sufficient group (p = 0.049).
Conclusion
Postsurgical hypocalcaemia was common in elderly thyroidectomized patients. After TT, lower magnesium levels were found in those patients with 24 h hypocalcaemia. In the HT group, preoperative 25OHD deficiency predicted lower postsurgical calcium levels.


Similar content being viewed by others
Data availability
All data and material was collected at Dr. Cesar Milstein Care Unit, Buenos Aires.
Change history
26 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12020-021-02943-3
References
A.L. Schafer, D. Shoback, Hypocalcemia: Definition, Etiology, Pathogenesis, Diagnosis, and Management. In: Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism, 9th edn. ed. by J.P. Bilezikian, (American Society for Bone and Mineral Research, John Wiley & Sons, Inc., 2019), p. 646–653.
J. Turner, N. Gittoes, P. Selby, __. Society for endocrinology endocrine emergency guidance: emergency management of acute hypocalcaemia in adult patients. Endocr. Connect. (2016). https://doi.org/10.1530/EC-16-0056
E.B.Y. Filho, R.V. Machry, R. Mesquita, R.S. Scheffel, A.L. Maia, The timing of parathyroid hormone measurement defines the cut-off values to accurately predict postoperative hypocalcemia: a prospective study. Endocrine. (2018). https://doi.org/10.1007/s12020-018-1601-9
O. Edafe, R. Antakia, N. Laskar, L. Uttley, S.P. Balasubramanian, Systematic review and meta-analysis of predictors of post-thyroidectomy hypocalcaemia. Br. J. Surg. (2014). https://doi.org/10.1002/bjs.9384
F. Dip, J. Falco, S. Verna, et al. Randomized Controlled Trial Comparing White Light with Near-Infrared Autofluorescence for Parathyroid Gland Identification During Total Thyroidectomy. J. Am. Colleg. Surg. (2019). https://doi.org/10.1016/j.jamcollsurg.2018.12.044
Z. Al-Qurayshi, C.B. Sullivan, A. Schwalje, et al. Presentation and Outcomes of Elderly Patients Undergoing Head and Neck Surgeries: a National Perspective. Otolaryngol. Head Neck Surg. (2020). https://doi.org/10.1177/0194599820911727
V.D. Tarasova, R.M. Tuttle. Current Management of Low Risk Differentiated Thyroid Cancer and Papillary Microcarcinoma. Clin. Oncol. (2017). https://doi.org/10.1016/j.clon.2016.12.009
Z. Wang, C.M. Vyas, O. Van Benschoten, et al. Quantitative Analysis of the Benefits and Risk of Thyroid Nodule Evaluation in Patients ≥70 Years Old. Thyroid. (2018). https://doi.org/10.1089/thy.2017.0655
M.C. Zatelli, L. Lamartina, D. Meringolo, et al. Thyroid nodule recurrence following lobo-isthmectomy: incidence, patient’s characteristics, and risk factors. J. Endocrinol. Investig. (2018). https://doi.org/10.1007/s40618-018-0946-5
M.F. Holick. Vitamin D Deficiency. N. Engl J. Med. (2007). https://doi.org/10.1056/NEJMra070553
C.S. Anast, J.M. Mohs, S.L. Kaplan, T.W. Burns. Evidence for Parathyroid Failure in Magnesium Deficiency. Science. (1972). https://doi.org/10.1126/science.177.4049.606
M.F. Holick, N.C. Binkley, H.A. Bischoff-Ferrari, et al., Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 96(7), 1911–1930 (2011). https://doi.org/10.1210/jc.2011-0385
R.V. Lloyd, R.Y. Osamura, G. Klöppel, J. Rosai, WHO Classification of Tumours of Endocrine Organs, 4th edn. (IARC print publications. WHO Press, World Health Organization, Switzerland, 2017).
A.E.L. McMurran, R. Blundell, V. Kim, Predictors of post-thyroidectomy hypocalcaemia: a systematic and narrative review. J. Laryngol. Otol. 134(6), 541–552 (2020). https://doi.org/10.1017/S0022215120001024
H.M. Mehanna, A. Jain, H. Randeva, J. Watkinson, A. Shaha, Postoperative hypocalcemia-The difference a definition makes. Head Neck. Published online 2009:NA-NA. https://doi.org/10.1002/hed.21175
D.R. Chadwick, Hypocalcaemia and permanent hypoparathyroidism after total/bilateral thyroidectomy in the BAETS Registry. Gland Surg. (2017) https://doi.org/10.21037/gs.2017.09.14
D.M. Shoback, J.P. Bilezikian, A.G. Costa, et al. Presentation of Hypoparathyroidism: Etiologies and Clinical Features. J. Clin. Endocrinol. Metab. (2016) https://doi.org/10.1210/jc.2015-3909
S.M. Suh, A.H. Tashjian, N. Matsuo, D.K. Parkinson, D. Fraser, Pathogenesis of Hypocalcemia in Primary Hypomagnesemia: Normal End-Organ Responsiveness to Parathyroid Hormone, Impaired Parathyroid Gland Function. J. Clin. Investig. (1973) https://doi.org/10.1172/JCI107159
A. Garrahy, M.S. Murphy, P. Sheahan, Impact of postoperative magnesium levels on early hypocalcemia and permanent hypoparathyroidism after thyroidectomy: Magnesium Levels and Postthyroidectomy Hypocalcemia. Head Neck. (2016) https://doi.org/10.1002/hed.23937
W. Wang, C. Meng, Q. Ouyang, J. Xie, X. Li, Magnesemia: an independent risk factor of hypocalcemia after thyroidectomy. CMAR. (2019) https://doi.org/10.2147/CMAR.S218179
C.S.P. Soares, J.V. Tagliarini, G.M.F.S. Mazeto, Preoperative vitamin D level as a post-total thyroidectomy hypocalcemia predictor: a prospective study. Braz. J. Otorhinolaryngol. (2021) https://doi.org/10.1016/j.bjorl.2019.07.001
Y. Erbil, U. Barbaros, B. Temel, et al. The impact of age, vitamin D3 level, and incidental parathyroidectomy on postoperative hypocalcemia after total or near total thyroidectomy. Am. J. Surg. (2009) https://doi.org/10.1016/j.amjsurg.2008.01.032
T. Al-Khatib, A.M. Althubaiti, A. Althubaiti, H.H. Mosli, R.O. Alwasiah, L.M. Badawood, Severe Vitamin D Deficiency: A Significant Predictor of Early Hypocalcemia after Total Thyroidectomy. Otolaryngol. Head Neck Surg. (2015) https://doi.org/10.1177/0194599814561209
Y. Lin, H.L. Ross, C.D. Raeburn, et al. Vitamin D deficiency does not increase the rate of postoperative hypocalcemia after thyroidectomy. Am. J. Surg. (2012) https://doi.org/10.1016/j.amjsurg.2012.10.001
G. Manzini, F. Malhofer, T. Weber, Can preoperative vitamin D deficiency predict postoperative hypoparathyroidism following thyroid surgery? Langenbecks Arch. Surg. (2019) https://doi.org/10.1007/s00423-019-01748-3
B. Oliveri, L. Plantalech, A. Bagur, et al. High prevalence of vitamin D insufficiency in healthy elderly people living at home in Argentina. Eur. J. Clin. Nutr. (2004). https://doi.org/10.1038/sj.ejcn.1601786
M. Tripathi, R. Karwasra, S. Parshad, Effect of preoperative vitamin D deficiency on postoperative hypocalcemia after thyroid surgery. Thyroid Res. (2014). https://doi.org/10.1186/1756-6614-7-8
L.A. Orloff, S.M. Wiseman, V.J. Bernet, et al. American Thyroid Association Statement on Postoperative Hypoparathyroidism: Diagnosis, Prevention, and Management in Adults. Thyroid. (2018) https://doi.org/10.1089/thy.2017.0309
L. Davies, L.G.T. Morris, M. Haymart, et al. American Association of Clinical Endocrinologists and American College of Endocrinology Disease State Clinical Review: the Increasing Incidence of Thyroid Cancer. Endocr. Pract. (2015) https://doi.org/10.4158/EP14466.DSCR
J.J. Díez, A. Oleaga, C. Álvarez-Escolá, T. Martín, J.C. Galofré, Guía clínica para el manejo de pacientes con carcinoma diferenciado de tiroides de bajo riesgo. Endocrinología y Nutrición. (2015) https://doi.org/10.1016/j.endonu.2015.02.006
S. Ahmadi, J.M. Gonzalez, M. Talbott, et al. Patient Preferences Around Extent of Surgery in Low-Risk Thyroid Cancer: a Discrete Choice Experiment. Thyroid. (2020) https://doi.org/10.1089/thy.2019.0590
K. Matsuzu, K. Sugino, K. Masudo, et al. Thyroid Lobectomy for Papillary Thyroid Cancer: long-term Follow-up Study of 1088 Cases. World J. Surg. (2014) https://doi.org/10.1007/s00268-013-2224-1
B.M. Barney, Y.J. Hitchcock, P. Sharma, D.C. Shrieve, J.D. Tward, Overall and cause-specific survival for patients undergoing lobectomy, near-total, or total thyroidectomy for differentiated thyroid cancer. Head Neck. (2011) https://doi.org/10.1002/hed.21504
A.H. Mendelsohn, D.A. Elashoff, E. Abemayor, M.A. St John, Surgery for Papillary Thyroid Carcinoma: Is Lobectomy Enough? Arch. Otolaryngol. Head Neck Surg. (2010) https://doi.org/10.1001/archoto.2010.181
I.J. Nixon, I. Ganly, S.G. Patel, et al. Thyroid lobectomy for treatment of well differentiated intrathyroid malignancy. Surgery. (2012) https://doi.org/10.1016/j.surg.2011.08.016
M.A. Adam, J. Pura, L. Gu, et al. Extent of Surgery for Papillary Thyroid Cancer Is Not Associated With Survival: an Analysis of 61,775 Patients. Ann. Surg. (2014) https://doi.org/10.1097/SLA.0000000000000925
Y. Erbil, Predictive Value of Age and Serum Parathormone and Vitamin D3 Levels for Postoperative Hypocalcemia After Total Thyroidectomy for Nontoxic Multinodular Goiter. Arch. Surg. (2007) https://doi.org/10.1001/archsurg.142.12.1182
Acknowledgements
We would like to thank the Endocrine and Surgery residents of Dr. Cesar Milstein Unit Care for their support in the follow-up of patients and collection of data.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by [B.M.P., S.N., M.A.Y., L.M., S.L., S.C., M.T., F.C., F.M.P. and B.G.]. The first draft of the paper was written by [B.M.P., M.E., S.C. and B.G.] and all authors commented on previous versions of the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
All participants gave a written voluntary consent to participate.
Consent for publication
Yes.
Ethical approval
The protocol was approved by the Ethics Committee for human studies of Dr. Cesar Milstein Care Unit, Buenos Aires.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Barbier, M.P., Mingote, E., Sforza, N. et al. Incidence and predictive factors of postoperative hypocalcaemia according to type of thyroid surgery in older adults. Endocrine 75, 276–283 (2022). https://doi.org/10.1007/s12020-021-02840-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02840-9