Abstract
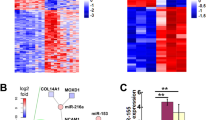
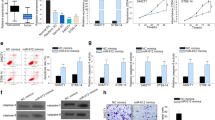
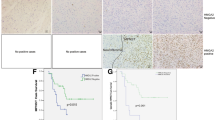
The dysregulation of microRNAs (miRNAs) is a crucial molecular signature of disease development. The potential implication of miRNAs in neurofibromatosis type 1 (NF1) remains poorly investigated. The expression levels of miR-27a-3p, miR-27b-3p, and neurofibromin 1 (NF1) were detected by real-time quantitative polymerase chain reaction (RT-qPCR) analysis. The functional roles of miR-27a-3p and miR-27b-3p in NF1 were explored by CCK8 (Cell Counting Kit-8), 5-ethynyl-2′-deoxyuridine (EdU), terminal deoxynucleoitidyl transferase dUTP nick-end labeling (TUNEL), and transwell assays. Luciferase reporter, RNA pull-down, and RNA binding protein immunoprecipitation (RIP) assays were employed to study the probable target relationship between miRNA and messenger RNA (mRNA). MiR-27b-3p and miR-27a-3p were upregulated in dermal and plexiform human Schwann cells (HSC) from NF1 neurofibromas as well as cell lines of malignant peripheral nerve sheath tumors (MPNSTs). MiR-27a-3p/miR-27b-3p mimics promoted the proliferative, migratory, and invasive ability of dermal HSC and MPNST cell ST88-14, while inhibiting the apoptotic capacity. MiR-27a-3p/miR-27b-3p inhibitors elicited the opposite impacts on the above cellular behaviors in dermal HSC and ST88-14. Intriguingly, NF1 was revealed to be the target of both miR-27a-3p and miR-27b-3p, and was negatively modulated by them. MiR-27a-3p/miR-27b-3p upregulation suppressed the expression of NF1 in dermal HSC and ST88-14. Furthermore, NF1 depletion counterbalanced the functional alteration induced by miR-27a-3p/miR-27b-3p inhibition. Our study suggests that both miR-27b-3p and miR-27a-3p are involved in upstream molecular activity responsible for the depletion of NF1, representing promising targets for therapeutic application in NF1.




Similar content being viewed by others
Data Availability
Research data and material are not shared.
Abbreviations
- NF1:
-
Neurofibromatosis type 1
- NF1:
-
Neurofibromin 1
- MPNSTs:
-
Malignant peripheral nerve sheath tumors
- HSC:
-
Human Schwann cells
- UTR:
-
Un-translated region
- mRNA:
-
Messenger RNA
- miRNAs:
-
MicroRNAs
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- FBS:
-
Fetal bovine serum
- RT-qPCR:
-
Real-time quantitative polymerase chain reaction
- CCK8:
-
Cell Counting Kit-8
- PFA:
-
Paraformaldehyde
- EdU:
-
5-Ethynyl-2′-deoxyuridine
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick-end labeling
- RIP:
-
RNA binding protein immunoprecipitation
- WT:
-
Wild-type
- Mut:
-
Mutant
- SD:
-
Standard deviation
- ANOVA:
-
Analysis of variance
References
An T et al (2019) Comparison of Alterations in miRNA Expression in Matched Tissue and Blood Samples during Spinal Cord Glioma Progression Scientific reports 9:9169. https://doi.org/10.1038/s41598-019-42364-x
Barkan B, Starinsky S, Friedman E, Stein R, Kloog Y (2006) The Ras inhibitor farnesylthiosalicylic acid as a potential therapy for neurofibromatosis type 1 Clinical cancer research : an official journal of the American Association for Cancer Research 12:5533–5542. https://doi.org/10.1158/1078-0432.Ccr-06-0792
Cappione AJ, French BL, Skuse GR (1997) A potential role for NF1 mRNA editing in the pathogenesis of NF1 tumors. Am J Hum Genet 60:305–312
Cimino PJ, Gutmann DH (2018) Neurofibromatosis type 1. Handb Clin Neurol 148:799–811. https://doi.org/10.1016/b978-0-444-64076-5.00051-x
Frassanito MA et al (2019) Bone marrow fibroblasts overexpress miR-27b and miR-214 in step with multiple myeloma progression, dependent on tumour cell-derived exosomes. J Pathol 247:241–253. https://doi.org/10.1002/path.5187
Gong M, Ma J, Li M, Zhou M, Hock JM, Yu X (2012) MicroRNA-204 critically regulates carcinogenesis in malignant peripheral nerve sheath tumors. Neuro-oncology 14:1007–1017. https://doi.org/10.1093/neuonc/nos124
Gutmann DH et al (1997) The diagnostic evaluation and multidisciplinary management of neurofibromatosis 1 and neurofibromatosis 2. Jama 278:51–57
Hoffmeyer S, Assum G, Griesser J, Kaufmann D, Nurnberg P, Krone W (1995) On unequal allelic expression of the neurofibromin gene in neurofibromatosis type 1. Hum Mol Genet 4:1267–1272. https://doi.org/10.1093/hmg/4.8.1267
Li Y et al (1995) Genomic organization of the neurofibromatosis 1 gene (NF1). Genomics 25:9–18. https://doi.org/10.1016/0888-7543(95)80104-t
Masliah-Planchon J et al (2013) MicroRNAome profiling in benign and malignant neurofibromatosis type 1-associated nerve sheath tumors: evidences of PTEN pathway alterations in early NF1 tumorigenesis BMC. Genomics 14:473. https://doi.org/10.1186/1471-2164-14-473
Metheny LJ, Cappione AJ, Skuse GR (1995) Genetic and epigenetic mechanisms in the pathogenesis of neurofibromatosis type I. J Neuropathol Exp Neurol 54:753–760. https://doi.org/10.1097/00005072-199511000-00001
Ottaviani S et al (2018) TGF-β induces miR-100 and miR-125b but blocks let-7a through LIN28B controlling PDAC progression. Nat Commun 9:1845. https://doi.org/10.1038/s41467-018-03962-x
Riccardi VM, Womack JE, Jacks T (1994) Neurofibromatosis and related tumors. Natural occurrence and animal models. Am J Pathol 145:994–1000
Rouhi A, Mager DL, Humphries RK, Kuchenbauer F (2008) MiRNAs, epigenetics, and cancer Mammalian genome : official journal of the International Mammalian Genome Society 19:517–525. https://doi.org/10.1007/s00335-008-9133-x
Skuse GR, Cappione AJ (1997) NA processing and clinical variability in neurofibromatosis type I (NF1). Hum Mol Genet 6:1707–1712
Sun X et al (2017) Glioma stem cells-derived exosomes promote the angiogenic ability of endothelial cells through miR-21/VEGF signal. Oncotarget 8:36137–36148. https://doi.org/10.18632/oncotarget.16661
Wang X et al (2019) LncRNA NEAT1 promotes extracellular matrix accumulation and epithelial-to-mesenchymal transition by targeting miR-27b-3p and ZEB1 in diabetic nephropathy. J Cell Physiol 234:12926–12933. https://doi.org/10.1002/jcp.27959
Xu Q, Cui Y, Luan J, Zhou X, Li H, Han J (2018) Exosomes from C2C12 myoblasts enhance osteogenic differentiation of MC3T3-E1 pre-osteoblasts by delivering miR-27a-3p . Biochem Biophys Res Commun 498:32–37. https://doi.org/10.1016/j.bbrc.2018.02.144
Xu W et al (2013) miR-24–3p and miR-27a-3p promote cell proliferation in glioma cells via cooperative regulation of MXI1. Int J Oncol 42:757–766. https://doi.org/10.3892/ijo.2012.1742
Yang X et al (2019) MiR-27b-3p promotes migration and invasion in colorectal cancer cells by targeting HOXA10. Bioscience Rep 39. https://doi.org/10.1042/bsr20191087
Zhang P et al (2014) EZH2-miR-30d-KPNB1 pathway regulates malignant peripheral nerve sheath tumour cell survival and tumourigenesis. J Pathol 232:308–318. https://doi.org/10.1002/path.4294
Zhao B, Zhang J, Chen X, Xu H, Huang B (2019) Mir-26b inhibits growth and resistance to paclitaxel chemotherapy by silencing the CDC6 gene in gastric cancer Archives of medical science : AMS 15:498–503. https://doi.org/10.5114/aoms.2018.73315
Acknowledgements
We appreciate the support of our experimenters.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare no relevant conflict of interests.
Ethics Approval
The human tissues utilized in the study were collected under the subject protocol approved by Institutional Review Boards of Rongcheng People’s Hospital of Shandong Province.
Consent to Participate
Written informed consent was provided by each subject or subject’s guardian.
Consent for Publication
The publication was approved by all the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12031_2020_1779_MOESM1_ESM.tif
Supplementary file1 (TIF 1316 KB) Figure S1. A. The expression of miR-27a-3p/miR-27b-3p in normal HSC was analyzed after transfection with miR-27a-3p/miR-27b-3p mimics. B. The cell vitality was studied by CCK8 after upregulation of miR-27b-3p and miR-27a-3p. C. Transwell assay was carried out to assess the migratory ability of the transfected normal HSC. **P < 0.01
12031_2020_1779_MOESM2_ESM.tif
Supplementary file2 (TIF 2089 KB) Figure S2. A. The expression of miR-27a-3p/miR-27b-3p in ST88-14 was analyzed after transfection with miR-27a-3p/miR-27b-3p mimics. B–C. The cell vitality and proliferation were studied by CCK8 and EdU assays, respectively, after upregulation of miR-27b-3p and miR-27a-3p. D. Transwell assay was carried out to assess the migratory ability of the transfected ST88-14. **P < 0.01
12031_2020_1779_MOESM3_ESM.tif
Supplementary file3 (TIF 1677 KB) Figure S3. A. The interference effectiveness of miR-27a-3p/miR-27b-3p inhibitor in ST88-14 was detected by RT-qPCR analysis. B–F. A series of functional experiments were performed to elucidate the effects of miR-27a-3p/miR-27b-3p inhibitor on proliferation, apoptosis, and migration by CCK8 (B), TUNEL (C), flow cytometry analysis (D), caspase-3 detection assay (E), and transwell assay (F). **P < 0.01.
12031_2020_1779_MOESM4_ESM.tif
Supplementary file4 (TIF 2903 KB) Figure S4. A. RT-qPCR assay was performed to study the expression of NF1 mRNA in the transfected ST88-14 with miR-27a-3p/miR-27b-3p mimics. B. RNA pull-down assay was completed to elucidate the molecular relationship between miR-27a-3p/miR-27b-3p and NF1. C. RIP assay was utilized to verify the interaction between target miRNAs and NF1. D-I. The rescue impact of NF1 depletion on miR-27b-3p/miR-27a-3p inhibitor-induced cell viability, proliferation, apoptosis, and migration phenotype was studied via CCK8 (D), EdU assay (E), TUNEL (F), flow cytometry analysis (G), caspase-3 detection assay (H), and transwell assay (I). **P < 0.01.
Rights and permissions
About this article
Cite this article
Lu, H., Liu, P. & Pang, Q. MiR-27a-3p/miR-27b-3p Promotes Neurofibromatosis Type 1 via Targeting of NF1 . J Mol Neurosci 71, 2353–2363 (2021). https://doi.org/10.1007/s12031-020-01779-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01779-2