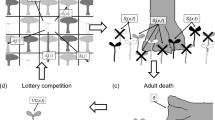
Abstract
The densities of a species and its specialist enemies are expected to covary, giving rare species an advantage that may promote species coexistence. This hypothesis has been particularly applied to tree species coexistence, and theoretical work has investigated how the spatial scale of dispersal and degree of enemy specialization influence coexistence outcomes. A simplifying assumption of this modeling work is that enemies are ubiquitous and host individuals always colonized. Yet recent studies have suggested that this assumption may not be justified in natural systems. Here we build a multispecies spatial model in which both the host species and enemy species are demographically dynamic. Exploring a range of spatial scales of enemy dispersal and seed dispersal, we find that the lag between seedling recruitment and colonization by enemies generally results in reduced species richness compared with models assuming ubiquitous enemies. This result depends on the spatial dynamics of enemy dispersal, and holds whether or not species diversity is supported by migration from outside the study area, as is often assumed in community ecology models. In light of our findings, we suggest that a full understanding of how enemies promote species coexistence must account for the colonization dynamics of enemies.



Similar content being viewed by others
References
Adler FR, Muller-Landau HC (2005) When do localized natural enemies increase species richness?. Ecol Lett 8:438–447
Bagchi R, Swinfield T, Gallery RE, Lewis OT, Gripenberg S, Narayan L, Freckleton RP (2010) Testing the Janen-Connell mechanism: pathogens cause overcompensating density dependence in a tropical tree. Ecol Lett 13:1262–1269
Bagchi R, Gallery RE, Gripenberg S, Gurr SJ, Narayan L, Addis CE, Freckleton RP, Lewis OT (2014) Pathogens and insect herbivores drive rainforest plant diversity and composition. Nature 506:85–90
Beckman NG, Neuhauser C, Muller-Landau HC (2012) The interacting effects of clumped seed dispersal and distance- and density-dependent mortality on seedling recruitment patterns. J Ecol 100:862–873
Berryman AA, Stenseth NC, Isaev AS (1987) Natural regulation of herbivorous forest insect populations. Oecologia 71:174–184
Bever JD, Mangan SA, Alexander HM (2015) Maintenance of plant species diversity by pathogens. Annu Rev Ecol Evol Syst 46:305–325
Bonsall MB, Hassell MP (1998) Population dynamics of apparent competition in a host-parasitoid assemblage. J Anim Ecol 67:918–929
Chen L, Swenson NG, Ji N, Mi X, Ren H, Guo L, Ma K (2019) Differential soil fungus accumulation and density dependence of trees in a subtropical forest. Science 366:124–128
Comita LS, Muller-Landau HC, Aguilar S, Hubbell SP (2010) Asymmetric density dependence shapes species abundances in a tropical tree community. Science 329:330–332
Comita LS, Queenborough SA, Murphy SJ, Eck JL, Xu K, Krishnadas M, Beckman N, Zhu Y (2014) Testing predictions of the Janzen,-Connell hypothesis: a meta-analysis of experimental evidence for distance- and densitiy-dependent seed and seedling survival. J Ecol 102:845–856
Connell JH (1971) Dynamics of populations, chap. on the role of natural enemies in preventing competitive exclusion in some marine animals and rain forest trees. Centre for Agricultural Publishing and Documentation, Wageningen, pp 298–312
Fricke EC, Tewksbury JJ, Rogers HS (2014) Multiple natural enemies cause distance-dependent mortality at the seed-to-seedling transition. Ecol Lett 17:593–598
Gripenberg S, Ovaskainen O, Morriën E, Roslin T (2008) Spatial population structure of a specialist leaf-mining moth. J Anim Ecol 77:757–767
Hart SP, Usinowicz J, Levine JM (2017) The spatial scales of species coexistence. Nat Ecol Evol 1:1066–1073
Holeski LM, Jander G, Agrawal AA (2012) Transgenerational defense induction and epigenetic inheritance in plants. Trends Ecol Evol 27:618–626
Janzen DH (1970) Herbivores and the number of tree species in tropical forests. Am Nat 104:501–528
Keeling MJ, Wilson HB, Pacala SW (2000) Reinterpreting space, time lags, and functional responses in ecological models. Science 290:1758–1761
LaManna JA, Mangan SA, Alonso A, Bourg NA, Brockelman WY, Bunyavejchewin S, Chang L-W, Chang J-M, Chuyong GB et al (2017) Plant diversity increases with the strength of negative density dependence at the global scale. Science 356:1389– 1392
Liu Y, Fang S, Chesson P, He F (2015) The effect of soil-borne pathogens depends on the abundance of host tree species. Nat Commun 6:1–7
Mangan SA, Schnitzer SA, Herre EA, Mack KML, Valencia MC, Sanchez EI, Bever JD (2010) Negative plant-soil feedback predicts tree-species relative abundance in a tropical forest. Nature 466:752–756
Marden JH, Mangan SA, Peterson MP, Wafula E, Fescemyer HW, Der JP, Depamphilis CW, Comita LS (2017) Ecological genomics of tropical trees: how local population size and allelic diversity of resistance genes relate to immune responses, cosusceptibility to pathogens, and negative density dependence. Mol Ecol 26:2498–2513
Mauch-Mani B, Baccelli I, Luna E, Flors V (2017) Defense priming: An adaptive part of induced resistance. Annu Rev Plant Biol 68:485–512
Ostfeld RS, Keesing F (2012) Effects of host diversity on infectious disease. Annu Rev Ecol Evol S 43:157–182
Sedio BE, Ostling AM (2013) How specialised must natural enemies be to facilitate coexistence among plants?. Ecol Lett 16:995– 1003
Stump SM, Chesson P (2015) Distance-responsive predation is not necessary for the Janzen-Connell hypothesis. Theor Popul Biol 106:60–70
Tack AJM, Ovaskainen O, Pulkkinen P, Roslin T (2010) Spatial location dominates over host plant genotype in stucturing an herbivore community. Ecol 91:2660–2672
Ylioja T, Roininen H, Ayres MP, Rousi M, Price PW (1999) Host-driven population dynamics in an herbivorous insect. Proc Natl Acad Sci USA 96:10,735–10,740
Acknowledgments
We thank Ben Gilbert for discussion, Hamish G. Spencer for feedback on the manuscript and for the idea of controlling for enemy abundance, and Simon Stump for helpful feedback on the manuscript and model, for suggesting the alternative method to control for migration rate with changing enemy dispersal, and for raising the issue of the interaction between scale and enemy dispersal distance on species richness. We thank Leithen M’Gonigle for sharing code with us that we drew on for our code. We thank Brian Sedio, Annette Ostling and Fred Adler for responding to questions about their models.
Funding
Financial support was provided by the Marsden Fund of the Royal Society of New Zealand, contract UOO1612, to Hamish G. Spencer.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Greenspoon, P., Wadhawan, K. Colonization limitation of specialized enemies reduces species richness. Theor Ecol 14, 1–8 (2021). https://doi.org/10.1007/s12080-020-00474-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-020-00474-7