Abstract
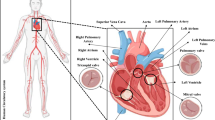
Brain artery occlusion is a major cause of cerebral stroke. During a stroke, perfusion decreases at tissues distal to the occlusion, but ischemia or hypoperfusion at tissues supplied by arterial branches proximal to the occlusion is not expected. However, ischemia proximal to the occlusion site has been observed without mechanical occlusion of the proximal branches. The mechanism of this unexpected ‘proximal ischemia’ is not well understood yet. In this study, transient three-dimensional computational fluid dynamics simulations of hemodynamic flow were performed to investigate ‘proximal ischemia’ using three non-Newtonian models and Newtonian model of a simple blood vessel consisting of an artery with one branch. Blood velocity of the artery dropped sharply after occlusion, resulting in shear rates low enough to cause red blood cell aggregation. The average shear rate in a cross-section of the artery during diastole was around 0.1 s−1, resulting in large red blood cell clots. Large aggregates formed in the artery have the potential to block the artery or the entrance of the branch.
Similar content being viewed by others
References
N. R. Sims and H. Muyderman. Mitochondria, oxidative metabolism and cell death in stroke, Biochim. Biophys. Acta., 1802 (1) (2010) 80–91.
J. A. Sirs, The flow of human blood through capillary tubes, J. Physiol. 442 (1) (1991) 569–583.
B. K. Lee, T. Alexy, R. B. Wenby and H. J. Meiselman, Red blood cell aggregation quantitated via Myrenne aggregometer and yield shear stress, Biorheology, 44 (1) (2007) 29–35.
J. B. Bederson, L. H. Pitts, M. Tsuji, M. C. Nishimura, R. L. Davis and H. Bartkowski, Rat middle cerebral artery occlusion: Evaluation of the model and development of a neurologic examination, Stroke, 17 (3) (1986) 472–476.
W. Reinke, P. Gaehtgens and P. C. Johnson, Blood viscosity in small tubes: Effect of shear rate, aggregation and sedimentation, Am. J. Physiol., 253 (2) (1987) H540–H547.
H. G. Morales, I. Larrabide, A. J. Geers, M. L. Aguilar and A. F. Frangi, Newtonian and non-newtonian blood flow in coiled cerebral aneurysms, J. Biomech., 46 (13) (2013) 2158–2164.
Ø. Evju, K. Valen-Sendstad and K. A. Mardal, A study of wall shear stress in 12 aneurysms with respect to different viscosity models and flow conditions, J. Biomech., 46 (16) (2013) 2802–2808.
S. De Gruttola, K. Boomsma and D. Poulikakos. Computational simulation of a non-Newtonian model of the blood separation process, Artif. Organs, 29 (12) (2005) 949–959.
T. Fukushima, T. Matsuzawa and T. Azuma, Numerical analysis of blood flow in the vertebral artery, J. Biomech. Eng., 104 (2) (1982) 143–147.
S. A. Berger and L. D. Jou, Flows in stenotic vessels, Annu. Rev. Fluid Mech., 32 (1) (2000) 347–382.
J. Cebral, M. Castro, O. Soto, R. Löhner and N. Alperin, Blood-flow models of the circle of Willis from magnetic resonance data, J. Eng. Math., 47 (3–4) (2003) 369–386.
A. G. Kanaris, A. D. Anastasiou and S. V. Paras, Numerical study of pulsatile blood flow in micro channels, Proc. of the 7 th European Congress of Chemical Engineering, Prague, Czech Republic (2010).
S. Shin and D. Y. Keum, Measurement of blood viscosity using mass-detecting sensor, Biosens. Bioelectron, 17 (5) (2002) 383–388.
D. M. Eckmann, S. Bowers, M. Stecker and A. T. Cheung, Hematocrit, volume expander, temperature and shear rate effects on blood viscosity, Anesth. Analg., 91 (3) (2000) 539–545.
J. Jung, R. W. Lyczkowski, C. B. Panchal and A. Hassanein, Multiphase hemodynamic simulation of pulsatile flow in a coronary artery. J. Biomech. 39 (11) (2006) 2064–2073.
N. M. P. Kakalis, A. P. Mitsos, J. V. Byrne and Y. Ventikos, The haemodynamics of endovascular aneurysm treatment: A computational modelling approach for estimating the influence of multiple coil deployment, IEEE Trans. Med. Imaging, 27 (6) (2008) 814–824.
G. Arbia, C. Corsini, M. E. Moghadam, A. L. Marsden, F. Migliavacca, G. Pennati, T. Y. Hsia and I. E. Vignon-Clementel, Numerical blood flow simulation in surgical corrections: What do we need for an accurate analysis?, J. Surg. Res., 186 (1) (2014) 44–55.
V. M. Pereira, O. Brina, A. M. Gonzales, A. P. Narata, P. Bijlenga, K. Schaller, K. O. Lovblad and R. Ouared, Evaluation of the influence of inlet boundary conditions on computational fluid dynamics for intracranial aneurysms: A virtual experiment, J. Biomech., 46 (9) (2013) 1531–1539.
T. M. Buckenham and I. A. Wright, Ultrasound of the extracranial vertebral artery, British J. Radiol., 77 (913) (2004) 15–20.
J. J. Bishop, A. S. Popel. M. Intaglietta and P. C. Johnson, Rheological effects of red blood cell aggregation in the venous network: A review of recent studies, Biorheology, 38 (2) (2001) 263–274.
H. Schmid-Schönbein, P. Gaehtgens and H. Hirsch, On the shear rate dependence of red cell aggregation in vitro, J. Clin. Invest., 47 (6) (1968) 1447–1454.
S. Chien, Shear dependence of effective cell volume as a determinant of blood viscosity, Science, 168 (3934) (1970) 977–979.
B. Chen, F. Guo and H. Xiang, Visualization study of motion and deformation of red blood cells in a microchan nel with straight, divergent and convergent sections, J. Biol. Phys., 37 (4) (2011) 429–440.
C. G. Caro, T. J. Pedley, R. C. Schroter and W. A. Seed, The mechanics of the Circulation, Second Ed., Cambridge University Press, Cambridge, UK (2012).
Y. H. Kim, P. J. VandeVord and J. S. Lee, Multiphase non-Newtonian effects on pulsatile hemodynamics in a coronary artery, Int. J. Numer. Methods Fluids, 58 (7) (2008) 803–825.
S. Chen, G. Barshtein, B. Gavish, Y. Mahler and S. Yedgar, Monitoring of red blood cell aggregability in a flowchamber by computerized image analysis, Clin. Hemorheol. Microcirc, 14 (4) (1994) 497–508.
A. L. Copley, R. G. King, C. R. Huang and S. Oka, Erythrocyte sedimentation of human blood at varying shear rates, Biorheology, 13 (5) (1976) 281–286.
M. Fenech, D. Garcia, H. Meiselman and G. Cloutier, A particle dynamic model of red blood cell aggregation kinetics, Ann. Biomed. Eng., 37 (11) (2009) 2299–2309.
R. S. Ajmani, Hypertension and hemorheology, Clin, Hemorheol, Microcirc, 17 (6) (1997) 397–420.
B. Almog, R. Gamzu, R. Almog, J. B. Lessing, I. Shapira, S. Berliner, D. Pauzner, S. Maslovitz and I. Levin, Enhanced erythrocyte aggregation in clinically diagnosed pelvic inflammatory disease, Sex. Transm. Dis., 32 (8) (2005) 484–486.
Author information
Authors and Affiliations
Corresponding author
Additional information
Recommended by Associate Editor Sung Yang
Myungjoon Kim received his Bachelor of Science degree in Mechanical Engineering from Sungkyunkwan University of Technology, Korea in 2009. Currently, he is a candidate in the combined master’s and doctorate program in the School of Mechanical Engineering at Sungkyunkwan University. His research is focused on the numerical analysis of particles suspended in fluid.
Taesung Kim received his Bachelor of Science degree in Mechanical Engineering from Seoul National University of Technology, Korea in 1994. He received his Master of Science and Doctor of Philosophy degrees in Mechanical Engineering from the University of Minnesota, Minneapolis, MN, USA in 1998 and 2002, respectively. Dr. Kim currently works as a professor in the School of Mechanical Engineering and as an adjunct professor at the SKKU Advanced Institute of Nano Technology at Sungkyunkwan University in Suwon, Korea. His research interests include nanoparticle synthesis, development of applications related to bio aerosols, Chemical Mechanical Polishing and thin film synthesis.
Rights and permissions
About this article
Cite this article
Kim, M., Min, T., Kwon, OK. et al. Numerical study on proximal ischemia. J Mech Sci Technol 29, 5523–5529 (2015). https://doi.org/10.1007/s12206-015-1153-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12206-015-1153-3