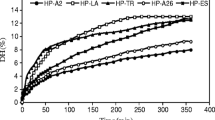
Abstract
Aiming to explore the use of Ionic liquids (ILs) not yet described in the literature, this work evaluated the hydrolysis of proteins from chicken viscera using the protease Alcalase modified and unmodified by the compound tetramethylammonium bromide. The protein hydrolysates produced in the presence of the IL presented values of antioxidant activities 40% higher than the hydrolysates obtained without IL. For DPPH-radical scavenging, the enzymatic hydrolysis performed using an amount of enzyme adjusted to 120 U/mL, pH 7.0 and 40 °C resulted in protein hydrolysates with maximum antioxidant activity of 188.11 µmol TE/g. For FRAP assay, the protein hydrolysates produced with 60 U/mL, pH 9.0 and 40 °C showed the highest antioxidant potential (1126.33 µmol TE/g). In the presence of the IL, it was possible to obtain protein hydrolysates from chicken viscera with similar antioxidant activities, compared to the protein hydrolysates produced without IL, using 1/3 of the amount of enzyme.
Graphical Abstract





Similar content being viewed by others
References
Neto, B.A.D., Spencer, J.: The impressive chemistry, applications and features of ionic liquids: properties, catalysis & catalysts and trends. J. Braz. Chem. 23, 987–1007 (2012)
Santiago, R.S., Santos, G.R., Aznar, M.: UNIQUAC correlation of liquid-liquid equilibrium in systems involving ionic liquids: the DFT-PCM approach. Fluid Phase Equilib. 293, 66–72 (2010)
Zhang, Q., Zhang, S., Deng, Y.: Recent advances in ionic liquid catalysis. Green Chem. 13, 2619–2637 (2011)
Armand, M., Macfarlane, D.R., Scrosati, B., Endres, F., Ohno, H.: Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 8, 621–629 (2009)
Kragl, U., Eckstein, M., Kaftzik, N.: Enzyme catalysis in ionic liquids. Curr. Opin. Biotechnol. 13, 565–571 (2002)
Moniruzzaman, M., Kamiya, N., Goto, M.: Activation and stabilization of enzymes in ionic liquids. Org. Biomol. Chem. 8, 2887–2899 (2010)
Naushad, M., Othman, Z.A.A.L., Kahn, A.B., Ali, M.: Effect of ionic liquid on acitivty, stability, and structure of enzymes: a review. Int. J. Biol. Macromol. 51, 555–560 (2012)
Sáez, F., Ballesteros, M., Ballesteros, I., Manzanares, P., Oliva, J.M., Negro, M.J.: Enzymatic hydrolysis from carbohydrates of barley straw pretreated by ionic liquids. J. Chem. Technol. Biotechnol. 88, 937–941 (2013)
Silva, V.G., De Castro, R.J.S.: Biocatalytic action of proteases in ionic liquids: improvements on their enzymatic activity, thermal stability and kinetic parameters. Int. J. Biol. Macromol. 114, 124–129 (2018)
Zhang, G., Holler, T., Napper, A.: Protease Assays. In: Holler, T., Napper, A. (eds.) Assay Guidance Manual. Eli Lilly & Company, Indianapolis (2012)
Vijayaraghavan, P., Lazarus, S., Vincent, S.G.P.: De-hairing protease production by an isolated Bacillus cereus strain at under solid-state fermentation using cow dung: biosynthesis and properties. Saudi J. Biol. Sci. 21, 27–34 (2014)
De Castro, R.J.S., Ohara, A., Nishide, T.G., Albernaz, J.R.M., Soares, M.H., Sato, H.H.: A new approach for proteases production by Aspergillus niger based on the kinetic and thermodynamic parameters of the enzymes obtained. Biocatal. Agric. Biotechnol. 4, 199–207 (2015)
Jamdar, S.N., Harikumar, P.: Autolytic degradation of chicken intestinal proteins. Bioresour. Technol. 96, 1276–1284 (2005)
Grazziotin, A., Pimentel, F.A., Sangali, S., Dejong, E.V., Brandelli, A.: Production of feather protein hydrolysate by keratinolytic bacterium. Vibrio sp. kr2. Bioresour. Technol. 98, 3172–3175 (2007)
Lasekan, A., Bakar, F.A., Hashim, D.: Potential of chicken by-produtcs as sources of useful biological resources. Waste Manage. 33, 552–565 (2013)
Bueno-Solano, C., López-Cervantes, J., Campas-Baypoli, O.N., Lauterio-García, R., Adan-Bante, N.P., Sánchez-Machado, D.I.: Chemical and biological characteristics of protein hydrolysates from fermented shrimp by-products. Food Chem. 112, 671–675 (2009)
Pagán, J., Ibarz, A., Falguera, V., Benítez, R.: Enzymatic hydrolysis kinetics and nitrogen recovery in the protein hydrolysate production from pig bones. J. Food Eng. 119, 655–659 (2013)
De Castro, R.J.S., Sato, H.H.: Biologically active peptides: processes for their generation, purification and identification and applications as natural additives in the food and pharmaceutical industries. Food Res. Int. 74, 185–198 (2015)
Huang, D., Ou, B., Prior, R.: The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 53, 1841–1856 (2005)
Charney, J., Tomarelli, R.M.: A colorimetric method for the determination of the proteolytic activity of duodenal juice. J. Biol. Chem. 171, 501–505 (1947)
Hartree, E.F.: Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal. Biochem. 48, 422–427 (1972)
Bougatef, A., Hajji, M., Balti, R., Lassoued, I., Triki-Ellouz, Y., Nasri, M.: Antioxidant and free radical-scavenging activities of smooth hound (Mustelus mustelus) muscle protein hydrolysates obtained by gastrointestinal proteases. Food Chem. 114, 1198–1205 (2009)
EMBRAPA, Comunicado técnico 125, Metodologia científica: determinação da atividade antioxidante total em frutas pelo método de redução do ferro (FRAP). https://ainfo.cnptia.embrapa.br/digital/bitstream/CNPAT-2010/11964/1/cot-125.pdf 10 Feb 2018
Zhao, H.: Methods for stabilizing and activating enzymes in ionic liquids—a review. J. Chem. Technol. Biotechnol. 85, 891–907 (2010)
Gao, W., Zhang, F., Zhang, G., Zhou, C.: Key factors affecting the activity and stability of enzymes in ionic liquids and novel applications in biocatalysis. Biochem. Eng. J. 99, 67–84 (2015)
Moniruzzaman, M., Nakashima, K., Kamiya, N., Goto, M.: Recent advances of enzymatic reactions in ionic liquids. Biochem. Eng. J. 48, 295–314 (2010)
Rodriguez, O., Cristovao, R.O., Tavares, A.P.M., Macedo, E.A.: Study of the alkyl chain length on laccase stability and enzymatic kinetic with imidazolium ionic liquids. Appl. Biochem. Biotechnol. 164, 524–533 (2011)
Smirnova, N.A., Safonova, E.A.: Ionic liquids as surfactants. Russ. J. Phys. Chem. 84, 1695–1704 (2010)
Rantwijk, F., Sheldon, R.A.: Biocatalysis in ionic liquids. Chem. Rev. 107, 2757–2785 (2007)
Dong, X., Fan, Y., Zhang, H., Zhong, Y., Yang, Y., Miao, J., Hua, S.: Inhibitory effects of ionic liquids on the latic dehydrogenase activity. Int. J. Biol. Macromol. 86, 155–161 (2016)
Fan, Y., Dong, X., Yan, L., Li, D., Hua, S., Hu, C., Pan, C.: Evaluation of the toxicity of ionic liquids on trypsin: a mechanism study. Chemosphere 148, 241–247 (2016)
Fan, Y., Dong, X., Li, X., Zhong, Y., Kong, J., Hua, S., Miao, J., Li, Y.: Spectroscopic studies on the inhibitory effects of ionic liquids on lipase activity. Spectrochim. Acta A. 159, 128–133 (2016)
Fan, Y., Wang, X., Li, J., Zhang, L., Yang, L., Gao, P., Zhou, Z.: Kinectic study of the inhibition of ionic liquids on the trypsin activity. J. Mol. Liq. 252, 392–398 (2018)
Klahn, M., Lim, G.S., Seduraman, A., Wu, P.: On the different roles of anions and cations in the solvation of enzymes in ionic liquids. Phys. Chem. Chem. 13, 1649–1662 (2011)
Kumari, M., Dohare, N., Maurya, N., Dohare, R., Patel, R.: Effect of 1-methyl-3- octyleimmidazolium chloride on the stability and activity of lysozyme: a spectroscopic and molecular dynamics studies. J. Biomol. Struct. Dyn. 35, 2016–2030 (2017)
Fan, Y., Dong, X., Zhong, Y., Li, J., Miao, J., Hua, S., Li, Y., Cheng, B., Chen, W.: Effects of ionic liquids on the hydrolysis of casein by lumbrokinase. Biochem. Eng. J. 109, 35–42 (2016)
Siemensma, A.D., Weijer, W.J., Bak, H.J.: The importance of peptide lengths in hypoallergenic infant formulas. Trends Food Sci. Technol. 4, 16–21 (1993)
Boza, J.J., Moennoz, D., Vuichoud, J., Jarret, A.R., Gaudard-De-Weck, D., Ballevre, O.: Protein hydrolysate vs free amino acid- based diets on the nutritional recovery of the starved rat. Eur. J. Nutr. 39, 237–243 (2000)
Althouse, P.J., Dinakar, P., Kilara, A.: Screening of proteolytic enzymes to enhance foaming of whey protein isolates. J. Food Sci. 60, 1110–1112 (1995)
Bernadini, R., Harnedy, P., Bolton, D., Kerry, J., O’Neil, E., Mullen, A.M.: Antioxidant and antimicrobial peptidic hydrolysates from muscle protein sources and by products. Food Chem. 124, 1296–1307 (2011)
Doucet, D., Otter, D.E., Gauthier, S.F., Foegeding, E.A.: Enzyme-induced gelation of extensively hydrolyzed whey proteins by Alcalase: peptide identification and determination of enzyme specificity. J. Agric. Food Chem. 51, 6300–6308 (2003)
Sarmadi, H., Ismail, A.: Antioxidative peptides from food proteins: a review. Peptides 31, 1949–1956 (2010)
Guo, H., Kouzuma, Y., Yonekura, M.: Structures and properties of antioxidative peptides derived from royal jelly protein. Food Chem. 113, 238–245 (2009)
Luna-Vital, D.A., Mojica, L., De Mejía, E.G., Mendoza, S., Loarca-Piña, G.: Biological potential of protein hydrolysates and peptides from common bean (Phaseolus vulgaris L.): a review. Food Res. Int. 76, 39–50 (2015)
Archer, M.C., Ragnarsson, J.O., Tannenbaum, S.R., Wang, D.I.C.: Enzymatic solubilization of an insoluble substrate fish protein concentrate: process and kinetic considerations. Biotechnol. Bioeng. 15, 181–196 (1973)
Montoya, C.A., Leterme, P., Victoria, N.F., Toro, O., Soufrant, W.B., Beebe, S.: Susceptibility of phaseolin to in vitro proteolysis is highly variable across common bean varieties (Phaseolus vulgaris). J. Agric. Food Chem. 56, 2183–2191 (2008)
Cornish-Bowden, A.: Fundamentals of Enzyme Kinetics, 3rd edn. Portland Press, London (2014)
Sukan, G., Andrews, A.T.: Application of the plastein reaction to caseins and to skim milk powder. I. Protein hydrolysis and plastein formation. J. Dairy Res. 49, 265–278 (1982)
Betancur-Ancona, D., Sosa-Espinoza, T., Ruiz-Ruiz, J., Segura-Campos, M., Chel-Guerrero, L.: Enzymatic hydrolysis of hard-to-cook bean (Phaseolus vulgaris L.) protein concentrates and its effects on biological and functional properties. Int. J. Food Sci. Technol. 49, 2–8 (2014)
Ortega, N., Diego, S., Perez-Mateos, M., Busto, M.D.: Kinetic properties and thermal behaviour of polygalacturonase used in fruit juice clarification. Food Chem. 88, 209–217 (2004)
Silvestre, M.P.C., Hamon, M., Yvon, M.: Analysis of protein hydrolysates. 2. Characterization of casein hydrolysates by a rapid peptide quantification method. J. Agric. Food Chem. 42, 2783–2789 (1994)
Irvine, G.B., Shaw, C.: High-performance gel permeation chromatography of proteins and peptides on columns of TSK- G2000-SW and TSK-G3000-SWsA volatile solvent giving separation based on charge and size of polypeptides. Anal. Biochem. 155, 141–148 (1986)
Spreti, N., Bartoletti, A., Di Profrio, P., Germani, R., Savelli, G.: Effects of ionic and zwitterionic surfactants on the stabilization of bovine catalase. Biotechnol. Prog. 11, 107–111 (1995)
Savelli, G., Spreti, N., Di Profrio, P.: Enzyme activity and stability control by amphiphlic self-organizing systems in aqueous solution. Curr. Opin. Colloid Interface Sci. 5, 111–117 (2000)
Yang, Z., Deng, J., Chen, L.F.: Effect of ionic and non-ionic surfactants of the activity and stability of mushroom tyrosinase. J. Mol. Catal. B Enzym. 47, 79–85 (2007)
Acknowledgements
The work described in this paper was substantially supported by the Department of Food Science, School of Food Engineering, University of Campinas, which is gratefully acknowledged. Acknowledgment to the Scientific Initiation Program of UNICAMP (PIBIC) for the opportunity to develop the research project and grant funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, V.G., de Castro, R.J.S. Enzymatic Hydrolysis of Proteins from Chicken Viscera in the Presence of an Ionic Liquid Enhanced Their Antioxidant Properties. Waste Biomass Valor 11, 3183–3193 (2020). https://doi.org/10.1007/s12649-019-00693-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-019-00693-y