Abstract
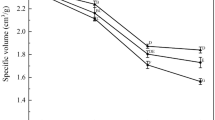
The cryoprotective effects of trehalose (w = 0–10%) on washed chicken meat (WCM) were investigated. WCM was produced from broiler, frozen and stored for 360 days on −30 °C. Myofibrillar protein functional stability was monitored by salt extractable protein (SEP) and differential scanning calorimetry (DSC). Salt extractable protein (SEP) showed that the addition of trehalose caused smaller decrease in protein solubility during frozen storage. Peak thermal transition (T p) and denaturation enthalpy (ΔH) of myofibrillar proteins were evaluated. Differential scanning calorimetry (DSC) revealed a shift in peak thermal transition temperature (T p) of myosin and actin to higher temperature as the mass fraction of trehalose increases. The transitions enthalpies of myosin and actin of WCM samples showed higher increase with the increase of mass fraction of trehalose. Since the value of denaturation enthalpy is directly related to amount of native proteins, higher values of ΔH indicates to the higher cryoprotective effects of trehalose.


Similar content being viewed by others
References
AOAC (2007) Official methods of analysis 18th edn, Association of Official Analytical Chemists. Gaithersburg, MD
Auh JH, Lee HG, Kim JW, Kim JC, Yoon HS, Park KH (1999) Highly concentrated branched oligosaccharides as cryoprotectant for surimi. J Food Sci 64:418–422. doi:10.1111/j.1365-2621.1999.tb15055.x
Barbut S, Findlay CJ (1991) Influence of sodium, potassium and magnesium-chloride on thermal-properties of beef muscle. J Food Sci 56:180–182. doi:10.1111/j.1365-2621.1991.tb08006.x
Bircan C, Barringer SA (2002) Determination of protein denaturation of muscle foods using the dielectric properties. J Food Sci 67:202–205. doi:10.1111/j.1365-2621.2002.tb11384.x
Colaco CALS, Smith CJS, Sen S, Roser DH, Newman Y, Ring S, Roser BJ (1994) Chemistry of Protein Stabilization by Trehalose. In: Cleland JL and Langer R (ed) Formulation and Delivery of Proteins and Peptides 567th edn., American Chemical Society, pp 222–240 doi: 10.1021/bk-1994-0567.ch014
Dawson PL, Sheldon BW, Ball HR (1988) Extraction of lipid and pigment components from mechanically deboned chicken meat. J Food Sci 53:1615–1617. doi:10.1111/j.1365-2621.1988.tb07798.x
Day SS, Dora KC (2010) Effect of sodium lactate as cryostabilizer on physico-chemical attributes of croaker (Johnius gangeticus) muscle protein. J Food Sci Tech Mys 47:432–436. doi:10.1007/s13197-010-0071-8
Fernandez-Martin F (2007) Bird muscles under hydrostatic high-pressure/temperature combinations - A DSC evaluation. J Therm Anal 87:285–290. doi:10.1007/s10973-006-7809-6
Findlay CJ, Barbut S (1990) Thermal Analysis of Food Proteins in Relation to Processing Data. In: Harwalkar VR, Ma CY (eds) Thermal Analysis of Food. Elsevier Science Publishers, Barking, pp 92–125
Herrera JJ, Pastoriza L, Sampedro G (2001) A DSC study on the effects of various maltodextrins and sucrose on protein changes in frozen-stored minced blue whiting muscle. J Sci Food Aric 81:377–384. doi:10.1002/1097-0010(200103)81:4<377::AID-JSFA820>3.0.CO;2-0
Kijowski J, Richardson RI (1996) The effect of cryoprotectants during freezing or freeze drying upon properties of washed mechanically recovered broiler meat. Int J Food Sci Technol 31:45–54. doi:10.1111/j.1365-2621.1996.00310.x
Kijowski JM, Mast MG (1988) Thermal-properties of proteins in chicken broiler tissues. J Food Sci 53:363–366. doi:10.1111/j.1365-2621.1988.tb07706.x
Lesiow T, Xiong YLL (2001) Mechanism of rheological changes in poultry myofibrillar proteins during gelation. Avian Poultry Biol Rev 12:137–149. doi:10.3184/147020601783698486
Leslie SB, Israeli E, Lighthart B, Crowe JH, Crowe LM (1995) Trehalose and sucrose protect both membranes and proteins in intact bacteria during drying. Appl Environ Microbiol 61:3592–3597
Li CT, Wick M (2001) Improvement of the physicochemical properties of pale soft and exudative (PSE) pork meat products with an extract from mechanically deboned turkey meat (MDTM). Meat Sci 58:189–195. doi:10.1016/S0309-1740(00)00151-0
MacDonald GA, Lanier T (1991) Carbohydrates as cryoprotectants for meats and surimi. Food Technol 45:150–159
Miller DP, Anderson RE, de Pablo JJ (1998) Stabilization of lactate dehydrogenase following freeze-thawing and vacuum-drying in the presence of trehalose and borate. Pharmacol Res 15:1215–1221. doi:10.1023/A:1011987707515
Murphy RY, Marks BP, Marcy JA (1998) Apparent specific heat of chicken breast patties and their constituent proteins by differential scanning calorimetry. J Food Sci 63:88–91. doi:10.1111/j.1365-2621.1998.tb15682.x
Nopianti R, Huda N, Ismail N, Ariffin F, Easa AM (2011) Effect of polydextrose on physicochemical properties of threadfin bream. J Food Sci Tech Mys. doi:10.1007/s13197-011-0394-0
Osako K, Hossain MA, Kuwahara K, Nozaki Y (2005) Effect of trehalose on the gel-forming ability, state of water and myofibril denaturation of horse mackerel Trachurus japonicus surimi during frozen storage. Fisheries Sci 71:367–373. doi:10.1111/j.1444-2906.2005.00973.x
Park JW (1994) Functional protein additives in surimi gels. J Food Sci 59:525–527. doi:10.1111/j.1365-2621.1994.tb05554.x
Park JW, Lanier TC, Green DP (1988) Cryoprotective effects of sugar, polyols, and or phosphates on Alaska pollack surimi. J Food Sci 53:1–3. doi:10.1111/j.1365-2621.1988.tb10163.x
Patist A, Zoerb H (2005) Preservation mechanisms of trehalose in food and biosystems. Colloid Surface B 40:107–113. doi:10.1016/j.colsurfb.2004.05.003
Stangierski J, Kijowski J (2003) Effect of selected commercial substances with cryoprotective activity on the quality of mechanically recovered, washed and frozen stored poultry meat. Nahrung 47:49–53. doi:10.1002/food.200390011
Stangierski J, Kijowski J (2008) Effect of selected substances on the properties of frozen myofibril preparation obtained from mechanically recovered poultry meat. Eur Food Res Technol 226:1415–1429. doi:10.1007/s00217-007-0672-2
Sych J, Lacroix C, Adambounou LT, Castaigne F (1990) Cryoprotective effects of lactitol, palatinit and polydextrose on cod surimi proteins during frozen storage. J Food Sci 55:356–360. doi:10.1111/j.1365-2621.1990.tb06762.x
Sych J, Lacroix C, Carrier M (1991) Determination of optimal level of lactitol for surimi. J Food Sci 56:285–290. doi:10.1111/j.1365-2621.1991.tb05263.x
Wright DJ, Wilding P (1984) Differential scanning calorimetric study of muscle and its proteins - myosin and its subfragments. J Sci Food Agric 35:357–372. doi:10.1002/jsfa.2740350317
Xiong YL, Brekke CJ, Leung HK (1987) Thermal-denaturation of muscle proteins from different species and muscle types as studied by differential scanning calorimetry. Can I Food Sc Tech J 20:357–362
Yang TS, Froning GW (1992) Changes in myofibrillar protein and collagen content of mechanically deboned chicken meat due to washing and screening. Poultry Sci 71:1221–1227. doi:10.3382/ps.0711221
Zhou AM, Benjakul S, Pan K, Gong H, Liu X (2006) Cryoprotective effects of trehalose and sodium lactate on tilapia (Sarotherodon nilotica) surimi during frozen storage. Food Chem 96:96–103. doi:10.1016/j.foodchem.2005.02.013
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kovačević, D., Mastanjević, K. Cryoprotective effect of trehalose on washed chicken meat. J Food Sci Technol 51, 1006–1010 (2014). https://doi.org/10.1007/s13197-011-0553-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-011-0553-3