Abstract
Industrial strains of Rhizopus oryzae is known for its strong ability to produce L-( +)-lactic acid, ethanol, and fumaric acid at high yields. To better understand the underlying mechanism behind the physiology of R. oryzae, we conducted the proteome changes between two different morphologies using two-dimensional polyacrylamide gel electrophoresis and mass spectrometry. R. oryzae exhibited pellet morphology and filamentous morphology when the initial pH of the culture medium was 3.0 and 5.0, respectively. The concentration of lactic acid reached 63.5 g L−1 in the samples containing the pellet morphology, compared to 41.5 g L−1 produced by filamentous R. oryzae. Proteomic analysis indicated that expression levels of 128 proteins changed significantly. Of these, 17 protein spots were successfully identified by mass spectrometry and were deemed to be mainly involved in carbohydrate metabolism, genetic information processing, chitin metabolism, protein catabolism, protein folding, and antioxidative pathway. L-lactate dehydrogenase (RO3G_06188), enolase (RO3G_05466) and 2, 3-bisphosphoglycerate-independent phosphoglycerate mutase (RO3G_02462) were found to be upregulated, while isocitrate dehydrogenase (RO3G_13820) was downregulated in the samples with pellet morphology compared to the filamentous hyphae. These results suggested that more carbon flow was directed towards lactic acid biosynthesis in R. oryzae hyphae with pellet morphology.


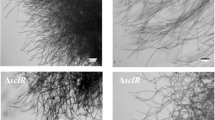
taken from a 12 cm Petri-dish at the end of the cultivations by digital camera showed the different morphologies of R. oryzae



Similar content being viewed by others
References
Bhadauria V, Wang LX, Peng YL (2010) Proteomic changes associated with deletion of the Magnaporthe oryzae conidial morphology-regulating gene COM1. Biol Direct 5:61. https://doi.org/10.1186/1745-6150-5-61
Byrne GS, Ward OP (1989) Effect of nutrition on pellet formation by Rhizopus arrhizus. Biotechnol Bioeng 33:912–914. https://doi.org/10.1002/bit.260330715
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The Carbohydrate-Active EnZymes database (CAZy): an expert resource for Glycogenomics. Nucleic Acids Res 37:D233–238. https://doi.org/10.1093/nar/gkn663
Chen CC, Cho YC, Lai CC, Hsu WH (2009) Purification and characterization of a new Rhizopuspepsin from Rhizopus oryzae NBRC 4749. J Agric Food Chem 57:6742–6747. https://doi.org/10.1021/jf8040337
Das RK, Brar SK (2014) Enhanced fumaric acid production from brewery wastewater and insight into the morphology of Rhizopus oryzae 1526. Appl Biochem Biotechnol 172:2974–2988. https://doi.org/10.1007/s12010-014-0739-z
Fu YQ, Xu Q, Li S, Huang H, Chen Y (2009) A novel multi-stage preculture strategy of Rhizopus oryzae ME-F12 for fumaric acid production in a stirred-tank reactor. World J Microbiol Biotechnol 25:1871–1876. https://doi.org/10.1007/s11274-009-0076-5
Fu YQ, Yin LF, Zhu HY, Jiang R, Li S, Xu Q (2014) Effects of pellet characteristics on L-lactic acid fermentation by R. oryzae: pellet morphology, diameter, density, and interior structure. Appl Biochem Biotechnol 174:2019–2030. https://doi.org/10.1007/s12010-014-1146-1
Isaacson T, Damasceno CMB, Saravanan RS, He Y, Catalá C, Saladié M, Rose JKC (2006) Sample extraction techniques for enhanced proteomic analysis of plant tissues. Nat Protoc 1:769–774. https://doi.org/10.1038/nprot.2006.102
Kossen NW (2000) The morphology of filamentous fungi. Adv Biochem Eng Biotechnol 70:1–33. https://doi.org/10.1007/3-540-44965-5_1
Langner T, Gohre V (2016) Fungal chitinases: function, regulation, and potential roles in plant/pathogen interactions. Curr Genet 62:243–254. https://doi.org/10.1007/s00294-015-0530-x
Liao W, Liu Y, Chen S (2007) Studying pellet formation of a filamentous fungus Rhizopus oryzae to enhance organic acid production. Appl Biochem Biotechnol 137–140:689–701. https://doi.org/10.1007/s12010-007-9089-4
Liu XH, Gao HM, Xu F, Lu JP, Devenish RJ, Lin FC (2012) Autophagy vitalizes the pathogenicity of pathogenic fungi. Autophagy 8:1415–1425. https://doi.org/10.4161/auto.21274
Liu Y, Liao W, Liu C, Chen S (2006) Optimization of L-(+)-lactic acid production using pelletized filamentous Rhizopus oryzae NRRL 395. Appl Biochem Biotechnol 131:844–853. https://doi.org/10.1385/ABAB:131:1-3:844
Ma LJ et al (2009) Genomic analysis of the basal lineage fungus Rhizopus oryzae reveals a whole-genome duplication. PLoS Genet 5:e1000549. https://doi.org/10.1371/journal.pgen.1000549
Meussen BJ, de Graaff LH, Sanders JP, Weusthuis RA (2012) Metabolic engineering of Rhizopus oryzae for the production of platform chemicals. Appl Microbiol Biotechnol 94:875–886. https://doi.org/10.1007/s00253-012-4033-0
Miura S, Arimura T, Hoshino M, Kojima M, Dwiarti L, Okabe M (2003) Optimization and scale-up of L-lactic acid fermentation by mutant strain Rhizopus sp. MK-96-1196 in airlift bioreactors. J Biosci Bioeng 96:65–69. https://doi.org/10.1016/s1389-1723(03)90098-3
Pera LM, Callieri DA (1997) Influence of calcium on fungal growth, hyphal morphology and citric acid production in Aspergillus niger. Folia Microbiol (Praha) 42:551–556. https://doi.org/10.1007/BF02815463
Xu J, Wang L, Ridgway D, Gu T, Moo-Young M (2000) Increased heterologous protein production in Aspergillus niger fermentation through extracellular proteases inhibition by pelleted growth. Biotechnol Prog 16:222–227. https://doi.org/10.1021/bp000006s
Xu Q, Liu Y, Li S, Jiang L, Huang H, Wen J (2016) Transcriptome analysis of Rhizopus oryzae in response to xylose during fumaric acid production. Bioprocess Biosyst Eng 39:1267–1280. https://doi.org/10.1007/s00449-016-1605-x
Yang J, Zhao X, Sun J, Kang Z, Ding S, Xu JR, Peng YL (2010) A novel protein Com1 is required for normal conidium morphology and full virulence in Magnaporthe oryzae. Mol Plant Microbe Interact 23:112–123. https://doi.org/10.1094/MPMI-23-1-0112
Yin LF, Ruan QC, Fu YQ (2013) Strain improvement of Rhizopus oryzae for over-production of lactic acid by random mutations. Afr J Microbiol Res 7:2970–2975. https://doi.org/10.5897/AJMR2013.5425
Yin P, Nishina N, Kosakai Y, Yahiro K, Pakr Y, Okabe M (1997) Enhanced production of l (+)-lactic acid from corn starch in a culture of Rhizopus oryzae using an air-lift bioreactor. J Ferment Bioeng 84:249–253. https://doi.org/10.1016/s0922-338x(97)82063-6
Zhou Y, Du J, Tsao GT (2000) Mycelial pellet formation by Rhizopus oryzae ATCC 20344. Appl Biochem Biotechnol 84–86:779–789. https://doi.org/10.1385/ABAB:84-86:1-9:779
Acknowledgements
This work was supported by a grant from the National Natural Science Foundation of China (21106091), a grant from Taizhou Science and Technology Plan Project (1701hb01) and a grant from Zhejiang Provincial Natural Science Foundation of China (LGG20B060001).
Author information
Authors and Affiliations
Contributions
LY and YF conceived and designed research. LY, XL, YZ, WY and WZ conducted research and collected data. LY was responsible for data analysis and wrote the manuscript. YF revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Yin, L., Luo, X., Zhang, Y. et al. Comparative proteomic analysis of Rhizopus oryzae hyphae displaying filamentous and pellet morphology. 3 Biotech 10, 469 (2020). https://doi.org/10.1007/s13205-020-02458-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02458-0