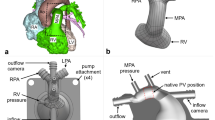
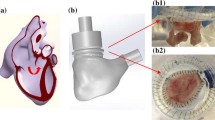
Abstract
Purpose
The congenital heart defect Tetralogy of Fallot (ToF) affects 1 in 2500 newborns annually in the US and typically requires surgical repair of the right ventricular outflow tract (RVOT) early in life, with variations in surgical technique leading to large disparities in RVOT anatomy among patients. Subsequently, often in adolescence or early adulthood, patients usually require surgical placement of a xenograft or allograft pulmonary valve prosthesis. Valve longevity is highly variable for reasons that remain poorly understood.
Methods
This work aims to assess the performance of bioprosthetic pulmonary valves in vitro using two 3D printed geometries: an idealized case based on healthy subjects aged 11 to 13 years and a diseased case with a 150% dilation in vessel diameter downstream of the valve. Each geometry was studied with two valve orientations: one with a valve leaflet opening posterior, which is the native pulmonary valve position, and one with a valve leaflet opening anterior.
Results
Full three-dimensional, three-component, phase-averaged velocity fields were obtained in the physiological models using 4D flow MRI. Flow features, particularly vortex formation and reversed flow regions, differed significantly between the RVOT geometries and valve orientations. Pronounced asymmetry in streamwise velocity was present in all cases, while the diseased geometry produced additional asymmetry in radial flows. Quantitative integral metrics demonstrated increased secondary flow strength and recirculation in the rotated orientation for the diseased geometry.
Conclusions
The compound effects of geometry and orientation on bioprosthetic valve hemodynamics illustrated in this study could have a crucial impact on long-term valve performance.











Similar content being viewed by others
References
Babu-Narayan, S. V., G. P. Diller, R. R. Gheta, A. J. Bastin, T. Karonis, W. Li, D. J. Pennell, H. Uemura, B. Sethia, M. A. Gatzoulis, and D. F. Shore. Clinical outcomes of surgical pulmonary valve replacement after repair of tetralogy of fallot and potential prognostic value of preoperative cardiopulmonary exercise testing. Circulation 129(1):18–27, 2014.
Bächler, P., N. Pinochet, J. Sotelo, G. Crelier, P. Irarrazaval, C. Tejos, and S. Uribe. Assessment of normal flow patterns in the pulmonary circulation by using 4D magnetic resonance velocity mapping. Magn. Reson. Imaging 31(2):178–188, 2013.
Bangalore, S., and D. L. Bhatt. Right heart catheterization, coronary angiography, and percutaneous coronary intervention. Circulation 124(17):e428–e433, 2011.
Banko, A. J., F. Coletti, C. J. Elkins, and J. K. Eaton. Oscillatory flow in the human airways from the mouth through several bronchial generations. Int. J. Heat Fluid Flow 61:45–57, 2016.
Barker, A. J., A. Roldán-Alzate, P. Entezari, S. J. Shah, N. C. Chesler, O. Wieben, M. Markl, and C. J. François. Four-dimensional flow assessment of pulmonary artery flow and wall shear stress in adult pulmonary arterial hypertension: results from two institutions. Magn. Reson. Med. 73(5):1904–1913, 2015.
Baskett, R. J. F., M. A. Nanton, A. E. Warren, and D. B. Ross. Human leukocyte antigen-DR and ABO mismatch are associated with accelerated homograft valve failure in children: implications for therapeutic interventions. J. Thorac. Cardiovasc. Surg. 126(1):232–238, 2003.
Batlivala, S. P., S. Emani, J. E. Mayer, and D. B. McElhinney. Pulmonary valve replacement function in adolescents: a comparison of bioprosthetic valves and homograft conduits. Ann. Thorac. Surg. 93(6):2007–2016, 2012.
Biglino, G., A. Giardini, T. Y. Hsia, R. Figliola, A. M. Taylor, S. Schievano S, and MOCHA Collaborative Group. Modeling single ventricle physiology: review of engineering tools to study first stage palliation of hypoplastic left heart syndrome. Front. Pediatr. 1:31, 2013.
Biglino, G., P. Verschueren, R. Zegels, A. M. Taylor, and S. Schievano. Rapid prototyping compliant arterial phantoms for in-vitro studies and device testing. J. Cardiovasc. Magn. Reson. 15(1):2, 2013.
Camp, T. A., K. C. Stewart, R. S. Figliola, and T. McQuinn. In vitro study of flow regulation for pulmonary insufficiency. J. Biomech. Eng. 129(2):284–288, 2007.
Chen, P. C., M. S. Sager, D. Zurakowski, F. A. Pigula, C. W. Baird, J. E. Mayer, P. J. del Nido, and S. M. Emani. Younger age and valve oversizing are predictors of structural valve deterioration after pulmonary valve replacement in patients with tetralogy of Fallot. J. Thorac. Cardiovasc. Surg. 143(2):352–360, 2012.
Cheng, C. P., R. J. Herfkens, C.A. Taylor, and J. A. Feinstein. Proximal pulmonary artery blood flow characteristics in healthy subjects measured in an upright posture using MRI: the effects of exercise and age. J. Magn. Reson. Imaging 21(6):752–758, 2005.
Christenson, J. T., D. Vala, J. Sierra, M. Beghetti, and A. Kalangos. Blood group incompatibility and accelerated homograft fibrocalcifications. J. Thorac. Cardiovasc. Surg. 127(1):242–250, 2004.
Das, A., W. Gottliebson, M. Karve, and R. K. Banerjee. Assessment of energy loss due to pulmonary valve insufficiency in tetralogy of fallot physiology using patient specific geometry. In ASME 2010 Summer Bioengineering Conference, Parts A and B, Naples, FL, USA, June 2010. American Society of Mechanical Engineers, pp. 245–246, 2010.
Dasi, L. P., H. A. Simon, P. Sucosky, and A. P. Yoganathan. Fluid mechanics of artificial heart valves. Clin. Exp. Pharmacol. Physiol. 36(2):225–237, 2009.
DiLorenzo, M. P., O. U. Elci, Y. Wang, A. Banerjee, T. Sato, B. Ky, E. Goldmuntz, and L. Mercer-Rosa. Longitudinal changes in right ventricular function in tetralogy of fallot in the initial years after surgical repair. J. Am. Soc. Echocardiogr. 31(7):816–821, 2018.
Elkins, C. J., and M. T. Alley. Magnetic resonance velocimetry: applications of magnetic resonance imaging in the measurement of fluid motion. Exp. Fluids 43(6):823–858, 2007.
Elkins, C. J., M. Markl, N. Pelc, and J. K. Eaton. 4D Magnetic resonance velocimetry for mean velocity measurements in complex turbulent flows. Exp. Fluids 34(4):494–503, 2003.
Figliola, R. S., A. Giardini, T. Conover, T. A. Camp, G. Biglino, J. Chiulli, and T. Y. Hsia. In vitro simulation and validation of the circulation with congenital heart defects. Prog. Pediatr. Cardiol. 30(1–2):71–80, 2010.
François, C. J., S. Srinivasan, M. L. Schiebler, S. B. Reeder, E. Niespodzany, B. R. Landgraf, O. Wieben, and A. Frydrychowicz. 4D cardiovascular magnetic resonance velocity mapping of alterations of right heart flow patterns and main pulmonary artery hemodynamics in tetralogy of Fallot. J. Cardiovasc. Magn. Reson. 14:16, 2012.
Gohean, J., R. Figliola, T. Camp, and T. McQuinn. Comparative in vitro study of bileaflet and tilting disk valve behavior in the pulmonary position. J. Biomech. Eng. 128(4):631–635, 2006.
Hang, T., A. Giardini, G. Biglino, T. Conover, R. S. Figliola, and MOCHA Collaborative Group. In vitro validation of a multiscale patient-specific Norwood palliation model. ASAIO J. 62(3):317–324, 2016.
Hess, A. T., M. M. Bissell, S. J. Glaze, A. Pitcher, S. Myerson, S. Neubauer, and M. D. Robson. Evaluation of circulation as a quantifying metric in 4D flow MRI. J. Cardiovasc. Magn. Reson. 15(Suppl 1):E36, 2013.
Jeong, D., P. V. Anagnostopoulos, A. Roldan-Alzate, S. Srinivasan, M. L. Schiebler, O. Wieben, and C. J. François. Ventricular kinetic energy may provide a novel noninvasive way to assess ventricular performance in patients with repaired tetralogy of Fallot. J. Thorac. Cardiovasc. Surg. 149(5):1339–1347, 2015.
Karamlou, T., R. M. Ungerleider, B. Alsoufi, G. Burch, M. Silberbach, M. Reller, and I. Shen. Oversizing pulmonary homograft conduits does not significantly decrease allograft failure in children. Eur. J. Cardiothorac. Surg. 27(4):548–553, 2005.
Khanna, A. D., K. D. Hill, S. K. Pasquali, A. S. Wallace, F. A. Masoudi, M. L. Jacobs, J. P. Jacobs, and T. Karamlou. Benchmark outcomes for pulmonary valve replacement using the Society of Thoracic Surgeons Databases. Ann. Thorac. Surg. 100(1):138–146, 2015.
Kwak, J. G., C. Lee, M. Lee, C. H. Lee, S. I. Jang, S. Y. Lee, S. J. Park, M. K. Song, and S. H. Kim. Does implantation of larger bioprosthetic pulmonary valves in young patients guarantee durability in adults? Durability analysis of stented bioprosthetic valves in the pulmonary position in patients with Tetralogy of Fallot. Eur. J. Cardiothorac. Surg. 49(4):1207–1212, 2016.
Lawley, C., K. Broadhouse, F. Callaghan, D. Winlaw, G. Figtree, and S. Grieve. 4D flow magnetic resonance imaging: role in pediatric congenital heart disease. Asian Cardiovasc. Thorac. Ann. 26:1–10, 2017.
Lee, C., Y. M. Kim, C. H. Lee, J. G. Kwak, C. S. Park, J. Y. Song, W. S. Shim, E. Y. Choi, S. Y. Lee, and J. S. Baek. Outcomes of pulmonary valve replacement in 170 patients with chronic pulmonary regurgitation after relief of right ventricular outflow tract obstruction: implications for optimal timing of pulmonary valve replacement. J. Am. Coll. Cardiol. 60(11):1005–1014, 2012.
Lee, C., C. H. Lee, and J. G. Kwak. Outcomes of redo pulmonary valve replacement for bioprosthetic pulmonary valve failure in 61 patients with congenital heart disease. Eur. J. Cardiothorac. Surg. 50(3):470–475, 2016.
Lee, N., A. Das, R. K. Banerjee, and W. M. Gottliebson. Comparison of stroke work between repaired tetralogy of Fallot and normal right ventricular physiologies. Heart Vessels 28(1):76–85, 2013.
Lim, W. L., Y. T. Chew, T. C. Chew, and H. T. Low. Pulsatile flow studies of a porcine bioprosthetic aortic valve in vitro: PIV measurements and shear-induced blood damage. J. Biomech. 34(11):1417–1427, 2001.
López-Zazueta, A., R. Ledesma-Alonso, J. E. V. Guzman, and R. Zenit. Study of the velocity and strain fields in the flow through prosthetic heart valves. J. Biomech. Eng. 133(12):121003, 2011.
Louvelle, L. M., M. G. Doyle, G. S. Van Arsdell, and C. H. Amon. A methodology to assess subregional geometric complexity for tetralogy of fallot patients. ASME J. Med. Diagn. 2(4):041006, 2019.
Markl, M., F. P. Chan, M. T. Alley, K. L. Wedding, M. T. Draney, C. J. Elkins, D. W. Parker, R. Wicker, C. A. Taylor, R. J. Herfkens, and N. J. Pelc. Time-resolved three-dimensional phase-contrast. MRI J. Magn. Reson. Imaging 17(4):499–506, 2003.
Markl, M., A. Frydrychowicz, S. Kozerke, M. Hope, and O. Wieben. 4D flow MRI. J. Magn. Reson. Imaging 36(5):1015–1036, 2012.
McElhinney, D. B., J. P. Cheatham, T. K. Jones, J. E. Lock, J. A. Vincent, E. M. Zahn, W. E. Hellenbrand. Stent fracture, valve dysfunction, and right ventricular outflow tract reintervention after transcatheter pulmonary valve implantation. Circ. Cardiovasc. Interv. 4(6):602–614, 2011.
Medero, R., C. Hoffman, and A. Roldán-Alzate. Comparison of 4D flow MRI and particle image velocimetry using an in vitro carotid bifurcation model. Ann. Biomed. Eng. 46(12):2112–2122, 2018.
Medero, R., K. Ruedinger, D. Rutkowski, K. Johnson, and A. Roldán-Alzate. In vitro assessment of flow variability in an intracranial aneurysm model using 4D flow MRI and tomographic PIV. Ann. Biomed. Eng. 48(10):2484–2493, 2020.
Nguyen, Y. N., M. Ismail, F. Kabinejadian, C. W. Ong, E. L. W. Tay, and H. L. Leo. Experimental study of right ventricular hemodynamics after tricuspid valve replacement therapies to treat tricuspid regurgitation. Cardiovasc. Eng. Technol. 8(4):401–418, 2017.
Nordmeyer, J., V. Tsang, R. Gaudin, P. Lurz, A. Frigiola, A. Jones, S. Schievano, C. van Doorn, P. Bonhoeffer, and A. M. Taylor. Quantitative assessment of homograft function 1 year after insertion into the pulmonary position: impact of in situ homograft geometry on valve competence. Eur. Heart J. 30(17):2147–2154. 2009.
Oliver, J. M., D. Garcia-Hamilton, A. E. Gonzalez, J. Ruiz-Cantador, A. Sanchez-Recalde, M. L.Polo, and A. Aroca. Risk factors for prosthetic pulmonary valve failure in patients with congenital heart disease. Am. J. Cardiol. 116(8):1252–1256, 2015.
Palacios-Morales, C. A., J. E. V. Guzmán, A. Beltrán, L. Ruiz-Huerta, A. Caballero-Ruiz, and R. Zenit. On the maximum operating frequency of prosthetic heart valves. Biomed. Phys. Eng. Express. 4(4):1–6, 2018.
Pelc, N. J., F. G. Sommer, K. C. Li, T. J. Brosnan, R. J. Herfkens, and D. R. Enzmann. Quantitative magnetic resonance flow imaging. Magn. Reson. Q. 10(3):125–147, 1994.
Raghav, V., C. Clifford, P. Midha, I. Okafor, B. Thurow, and A. Yoganathan. Three-dimensional extent of flow stagnation in transcatheter heart valves. J. R. Soc. Interface 16(154):1–9, 2019.
Raghav, V., S. Sastry, and N. Saikrishnan. Experimental assessment of flow fields associated with heart valve prostheses using particle image velocimetry (PIV): recommendations for best practices. Cardiovasc. Eng. Technol. 9(3):273–287, 2018.
Reiter, G., U. Reiter, G. Kovacs, B. Kainz, K. Schmidt, R. Maier, H. Olschewski, and R. Rienmueller. Magnetic resonance-derived 3-dimensional blood flow patterns in the main pulmonary artery as a marker of pulmonary hypertension and a measure of elevated mean pulmonary arterial pressure. Circ. Cardiovasc. Imaging 1(1):23–30, 2008.
Sacks, M. S., W. D. Merryman, and D. E. Schmidt. On the biomechanics of heart valve function. J. Biomech. 42(12):1804–1824, 2009.
Sacks, M. S., and A. P. Yoganathan. Heart valve function: a biomechanical perspective. Philos. Trans. R. Soc. Lond. B Biol. Sci. 362(1484):1369–1391, 2007.
Schäfer, M., S. Humphries, K. R. Stenmark, V. O. Kheyfets, J. K. Buckner, K. S. Hunter, and B. E. Fenster. 4D-flow cardiac magnetic resonance-derived vorticity is sensitive marker of left ventricular diastolic dysfunction in patients with mild-to-moderate chronic obstructive pulmonary disease. Eur. Heart J. Cardiovasc. Imaging 19(4):415–424, 2018.
Schievano, S., C. Capelli, C. Young, P. Lurz, J. Nordmeyer, C. Owens, P. Bonhoeffer, and A. M. Taylor. Four-dimensional computed tomography: a method of assessing right ventricular outflow tract and pulmonary artery deformations throughout the cardiac cycle. Eur. Radiol. 21(1):36–45, 2011.
Schievano, S., L. Coats, F. Migliavacca, W. Norman, A. Frigiola, J. Deanfield, P. Bonhoeffer, and A. Taylor. Variations in right ventricular outflow tract morphology following repair of congenital heart disease: implications for percutaneous pulmonary valve implantation. J. Cardiovasc. Magn. Reson. 9:687–695, 2007.
Siddiqui, R. F., J. R. Abraham, and J. Butany. Bioprosthetic heart valves: modes of failure. Histopathology 55(2):135–144, 2009.
Sotiropoulos, F., T. B. Le, and A. Gilmanov. Fluid mechanics of heart valves and their replacements. Annu. Rev. Fluid Mech. 48(1):259–283, 2016.
Stankovic, Z., B. D. Allen, J. Garcia, K. B. Jarvis, and M. Markl. 4D flow imaging with MRI. Cardiovasc. Diagn. Ther. 4(2):173–192, 2014.
van Straten, A., H. W. Vliegen, M. G. Hazekamp, J. J. Bax, P. H. Schoof, J. Ottenkamp, E. E. van der Wall, and A. de Roos. Right ventricular function after pulmonary valve replacement in patients with tetralogy of fallot. Radiology 233(3):824–829, 2004.
Vismara, R., K. Laganà, F. Migliavacca, S. Schievano, L. Coats, A. Taylor, and P. Bonhoeffer. Experimental setup to evaluate the performance of percutaneous pulmonary valved stent in different outflow tract morphologies. Artif. Organs. 33(1):46–53, 2009.
Vongpatanasin, W., L. D. Hillis, and R. A. Lange. Prosthetic heart valves. N. Engl. J. Med. 335(6):407–416, 1996.
Vukicevic, M., T. Conover, M. Jaeggli, J. Zhou, G. Pennati, T. Y. Hsia, R. S. Figliola. Control of respiration-driven retrograde flow in the subdiaphragmatic venous return of the Fontan circulation. ASAIO J. 60(4):391–399, 2014.
Wells, W. J., H. Arroyo, R. M. Bremner, J. Wood, and V. A. Starnes. Homograft conduit failure in infants is not due to somatic outgrowth. J. Thorac. Cardiovasc. Surg. 124(1):88–96, 2002.
Yoganathan, A. P., K. B. Chandran, and F. Sotiropoulos. Flow in prosthetic heart valves: state-of-the-art and future directions. Ann. Biomed. Eng. 33(12):1689–1694, 2005.
Yoganathan, A. P., Z. He, and S. Casey Jones. Fluid mechanics of heart valves. Annu. Rev. Biomed. Eng. 6(1):331–362, 2004.
Zubairi, R., S. Malik, R. D. B. Jaquiss, M. Imamura, J. Gossett, and W. R. Morrow. Risk factors for prosthesis failure in pulmonary valve replacement. Ann. Thorac. Surg. 91(2):561–565, 2011.
Acknowledgements
We gratefully acknowledge Professor Richard Figliola for his assistance in adjusting the experimental design in order to produce physiological results. In addition, we thank Dr. Katsuhide Maeda for the opportunity to personally observe pulmonary valve replacement surgeries. The authors also thank their funding sources: The Stanford Maternal and Child Health Research Institute (Marsden, Eaton, and McElhinney), the American Heart Association (Marsden, Eaton, and McElhinney), the Stanford Bio-X Bowes Fellowship (Schiavone), the Vera Moulton Wall Center (Marsden), and the Benchmark Capital Fellowship (Marsden).
Funding Information
This work is funded by The Stanford Maternal and Child Health Research Institute, the American Heart Association, the Stanford Bio-X Bowes Fellowship, the Vera Moulton Wall Center, and the Benchmark Capital Fellowship.
Conflict of interest
N.K. Schiavone, C.J. Elkins, J.K. Eaton, and A.L. Marsden declare they have no conflicts of interest. D.B. McElhinney is a proctor and consultant for Medtronic, which is not directly relevant to this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Keefe B. Manning oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (mp4 3970 KB)
Supplementary material 2 (mp4 4077 KB)
Rights and permissions
About this article
Cite this article
Schiavone, N.K., Elkins, C.J., McElhinney, D.B. et al. In Vitro Assessment of Right Ventricular Outflow Tract Anatomy and Valve Orientation Effects on Bioprosthetic Pulmonary Valve Hemodynamics. Cardiovasc Eng Tech 12, 215–231 (2021). https://doi.org/10.1007/s13239-020-00507-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-020-00507-6