Abstract
Purpose
Invasive pneumococcal disease (IPD) is responsible for substantial mortality and morbidity worldwide. We aimed to identify host and bacterial factors associated with 30-day mortality in 18-year-old patients hospitalized with IPD in France from 2013 to 2015.
Methods
This study analyzed data collected from consecutives IPD cases included in two parallel multi-center cohort studies: COMBAT study (280 patients with pneumococcal community-acquired bacterial meningitis) and SIIP study (491 patients with non-meningitis IPD). Factors associated with 30-day mortality were identified using logistic regression.
Results
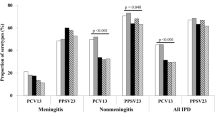
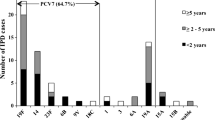
Among the 771 enrolled patients (median age 66 years, IQR [52.0–79.7]), 592/767 (77.2%) had at least one chronic disease. Patients with meningitis were younger (60.2 vs 70.9 years; p < 0.001) and had fewer chronic diseases than those with non-meningitis IPD (73.3% vs 79.4%; p = 0.05). Non-vaccine serotypes were more frequent in meningitis patients than in those with other IPD (36.1% vs 23.1%; p < 0.001). The overall 30-day mortality was 16.7% and patients with concurrent meningitis and extra-cerebral IPD had the highest 30-day mortality rate (26.5%). On multivariate analyses, older age, history of malignant solid tumor, meningeal IPD and serotypes previously identified with high mortality potential were independently associated with 30-day mortality. Of the serotypes with high mortality potential, 80% were included in licensed (PCV13 or PPV23) vaccines.
Conclusion
We observed an effect of both host factors and pneumococcal serotypes on 30-day mortality in IPD. This highlights the need for a focused strategy to vaccinate at-risk patients.
Clinical trial
ClinicalTrial. Gov identification number: NCT01730690

Similar content being viewed by others
References
Muhammad RD, Oza-Frank R, Zell E, Link-Gelles R, Narayan KMV, Schaffner W, et al. Epidemiology of invasive pneumococcal disease among high-risk adults since the introduction of pneumococcal conjugate vaccine for children. Clin Infect Dis. 2013;56:e59-67.
Drijkoningen JJC, Rohde GGU. Pneumococcal infection in adults: burden of disease. Clin Microbiol Infect. 2014;20:45–51.
Amaro R, Liapikou A, Cilloniz C, Gabarrus A, Marco F, Sellares J, et al. Predictive and prognostic factors in patients with blood-culture-positive community-acquired pneumococcal pneumonia. Eur Respir J. 2016;48:797–807.
Fine MJ, Smith MA, Carson CA, Mutha SS, Sankey SS, Weissfeld LA, et al. Prognosis and outcomes of patients with community-acquired pneumonia. A meta-analysis. JAMA. 1996;275:134–41.
Naucler P, Darenberg J, Morfeldt E, Ortqvist A, Henriques NB. Contribution of host, bacterial factors and antibiotic treatment to mortality in adult patients with bacteraemic pneumococcal pneumonia. Thorax. 2013;68:571–9.
Kalin M, Ortqvist A, Almela M, Aufwerber E, Dwyer R, Henriques B, et al. Prospective study of prognostic factors in community-acquired bacteremic pneumococcal disease in 5 countries. J Infect Dis. 2000;182:840–7.
Waterer GW, Quasney MW, Cantor RM, Wunderink RG. Septic shock and respiratory failure in community-acquired pneumonia have different TNF polymorphism associations. Am J Respir Crit Care Med. 2001;163:1599–604.
Meehan TP, Fine MJ, Krumholz HM, Scinto JD, Galusha DH, Mockalis JT, et al. Quality of care, process, and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–4.
Baddour LM, Yu VL, Klugman KP, Feldman C, Ortqvist A, Rello J, et al. Combination antibiotic therapy lowers mortality among severely ill patients with pneumococcal bacteremia. Am J Respir Crit Care Med. 2004;170:440–4.
Aspa J, Rajas O, de Castro FR, Blanquer J, Zalacain R, Fenoll A, et al. Drug-resistant pneumococcal pneumonia: clinical relevance and related factors. Clin Infect Dis. 2004;38:787–98.
Feikin DR, Schuchat A, Kolczak M, Barrett NL, Harrison LH, Lefkowitz L, et al. Mortality from invasive pneumococcal pneumonia in the era of antibiotic resistance, 1995–1997. Am J Public Health. 2000;90:223–9.
Rello J, Lisboa T, Lujan M, Gallego M, Kee C, Kay I, et al. Severity of pneumococcal pneumonia associated with genomic bacterial load. Chest. 2009;136:832–40.
Hughes GJ, Wright LB, Chapman KE, Wilson D, Gorton R. Serotype-specific differences in short- and longer-term mortality following invasive pneumococcal disease. Epidemiol Infect. 2016;144:2654–69.
Geno KA, Saad JS, Nahm MH. Discovery of novel pneumococcal serotype 35D, a natural WciG-deficient variant of serotype 35B. J Clin Microbiol. 2017;55:1416–25.
Calix JJ, Nahm MH. A new pneumococcal serotype, 11E, has a variably inactivated wcjE gene. J Infect Dis. 2010;202:29–38.
Ganaie F, Saad JS, McGee L, van Tonder AJ, Bentley SD, Lo SW, et al. (2020) A new pneumococcal capsule type, 10D, is the 100th serotype and has a large cps fragment from an oral Streptococcus. mBio. Cited 8 Mar 2021. https://mbio.asm.org/content/11/3/e00937-20
Brueggemann AB, Peto TEA, Crook DW, Butler JC, Kristinsson KG, Spratt BG. Temporal and geographic stability of the serogroup-specific invasive disease potential of Streptococcus pneumoniae in children. J Infect Dis. 2004;190:1203–11.
Jansen AGSC, Rodenburg GD, van der Ende A, van Alphen L, Veenhoven RH, Spanjaard L, et al. Invasive pneumococcal disease among adults: associations among serotypes, disease characteristics, and outcome. Clin Infect Dis. 2009;49:e23-29.
Sjöström K, Spindler C, Ortqvist A, Kalin M, Sandgren A, Kühlmann-Berenzon S, et al. Clonal and capsular types decide whether pneumococci will act as a primary or opportunistic pathogen. Clin Infect Dis. 2006;42:451–9.
Harboe ZB, Thomsen RW, Riis A, Valentiner-Branth P, Christensen JJ, Lambertsen L, et al. Pneumococcal serotypes and mortality following invasive pneumococcal disease: a population-based cohort study. PLoS Med. 2009;6: e1000081.
Ouldali N, Varon E, Levy C, Angoulvant F, Georges S, Ploy M-C, et al. Invasive pneumococcal disease incidence in children and adults in France during the pneumococcal conjugate vaccine era: an interrupted time-series analysis of data from a 17-year national prospective surveillance study. Lancet Infect Dis. 2021;21:137–47.
Ministères des Affaires Sociales et de la Santé (2017) Calendrier des vaccinations et recommandations vaccinales 2017. https://www.mesvaccins.net/textes/calendrier_vaccinations_2017.pdf
Tubiana S, Varon E, Biron C, Ploy M-C, Mourvillier B, Taha M-K, et al. Community-acquired bacterial meningitis in adults: in-hospital prognosis, long-term disability and determinants of outcome in a multicentre prospective cohort. Clin Microbiol Infect. 2020;26:1192–200.
Danis K, Varon E, Lepoutre A, Janssen C, Forestier E, Epaulard O, et al. Factors associated with severe nonmeningitis invasive pneumococcal disease in adults in France. Open Forum Infect Dis. 2019;6:ofz510.
European Commission. Commission Decision 2018/945/EC of 22 June 2018 on the communicable diseases and related special health issues to be covered by epidemiological surveillance as well as relevant case definitions. Official Journal of European Union 2018, pp L170/33–L170/34.
Brueggemann AB, Griffiths DT, Meats E, Peto T, Crook DW, Spratt BG. Clonal relationships between invasive and carriage Streptococcus pneumoniae and serotype- and clone-specific differences in invasive disease potential. J Infect Dis. 2003;187:1424–32.
Hanage WP, Kaijalainen TH, Syrjänen RK, Auranen K, Leinonen M, Mäkelä PH, et al. Invasiveness of serotypes and clones of Streptococcus pneumoniae among children in Finland. Infect Immun. 2005;73:431–5.
Balsells E, Dagan R, Yildirim I, Gounder PP, Steens A, Muñoz-Almagro C, et al. The relative invasive disease potential of Streptococcus pneumoniae among children after PCV introduction: a systematic review and meta-analysis. J Infect. 2018;77:368–78.
Varon E, Cohen R, Béchet S, Doit C, Levy C. Invasive disease potential of pneumococci before and after the 13-valent pneumococcal conjugate vaccine implementation in children. Vaccine. 2015;33:6178–85.
Sá-Leão R, Pinto F, Aguiar S, Nunes S, Carriço JA, Frazão N, et al. Analysis of invasiveness of pneumococcal serotypes and clones circulating in Portugal before widespread use of conjugate vaccines reveals heterogeneous behavior of clones expressing the same serotype. J Clin Microbiol. 2011;49:1369–75.
Weinberger DM, Harboe ZB, Sanders EAM, Ndiritu M, Klugman KP, Rückinger S, et al. Association of serotype with risk of death due to pneumococcal pneumonia: a meta-analysis. Clin Infect Dis. 2010;51:692–9.
Hanada S, Iwata S, Kishi K, Morozumi M, Chiba N, Wajima T, et al. Host factors and biomarkers associated with poor outcomes in adults with invasive pneumococcal disease. PLoS ONE. 2016;11: e0147877.
Martens P, Worm SW, Lundgren B, Konradsen HB, Benfield T. Serotype-specific mortality from invasive Streptococcus pneumoniae disease revisited. BMC Infect Dis. 2004;4:21.
Cohen C, Naidoo N, Meiring S, de Gouveia L, von Mollendorf C, Walaza S, et al. Streptococcus pneumoniae serotypes and mortality in adults and adolescents in South Africa: analysis of national surveillance data, 2003–2008. PLoS ONE. 2015;10:e0140185.
Alanee SRJ, McGee L, Jackson D, Chiou CC, Feldman C, Morris AJ, et al. Association of serotypes of Streptococcus pneumoniae with disease severity and outcome in adults: an international study. Clin Infect Dis. 2007;45:46–51.
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS international sepsis definitions conference. Intensive Care Med. 2003;29:530–8.
Rubin DB. Multiple imputation for nonresponse in surveys. New York: Wiley; 1987.
Lepoutre A, Varon E, Georges S, Dorléans F, Janoir C, Gutmann L, et al. Impact of the pneumococcal conjugate vaccines on invasive pneumococcal disease in France, 2001–2012. Vaccine. 2015;33:359–66.
Delelis-Fanien A-S, Séité F, Priner M, Paccalin M. Couverture vaccinale antigrippale et antipneumococcique à partir de 65ans: étude sur 299 patients ambulatoires. Rev Méd Interne. 2009;30:656–60.
Wyplosz B, Fernandes J, Raguideau F, Leboucher C, Goussiaume G, Moïsi J. COVARISQ (estimation de la COuverture VAccinale des adultes à RISQues): taux de vaccination des malades atteints de comorbidités en France en 2017. Méd Mal Infect. 2020;50:S179–80.
Vietri J, Harnett J, Emir B, Chilson E. Uptake of 13-valent pneumococcal conjugate vaccine among US adults aged 19 to 64 years with immunocompromising conditions. Hum Vaccines Immunother. 2020;16:161–8.
Vila-Córcoles A, Ochoa-Gondar O, de Diego C, Satué E, Vila-Rovira A, Aragón M. Pneumococcal vaccination coverages by age, sex and specific underlying risk conditions among middle-aged and older adults in Catalonia, Spain, 2017. Euro Surveill. 2019;24:1800446.
Falkenhorst G, Remschmidt C, Harder T, Hummers-Pradier E, Wichmann O, Bogdan C (2017) Effectiveness of the 23-valent pneumococcal polysaccharide vaccine (PPV23) against pneumococcal disease in the elderly: systematic review and meta-analysis. PLoS One. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5218810/. Cited 10 Dec 2020
Djennad A, Ramsay ME, Pebody R, Fry NK, Sheppard C, Ladhani SN, et al. Effectiveness of 23-valent polysaccharide pneumococcal vaccine and changes in invasive pneumococcal disease incidence from 2000 to 2017 in those aged 65 and over in England and Wales. EClinicalMedicine. 2018;6:42–50.
Tomczyk S, Bennett NM, Stoecker C, Gierke R, Moore MR, Whitney CG, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among adults aged ≥65 years: recommendations of the advisory committee on immunization practices (ACIP). MMWR Morb Mortal Wkly Rep. 2014;63:822–5.
Chen H, Matsumoto H, Horita N, Hara Y, Kobayashi N, Kaneko T. Prognostic factors for mortality in invasive pneumococcal disease in adult: a system review and meta-analysis. Sci Rep. 2021;11:11865.
Song J-H, Oh WS, Kang C-I, Chung DR, Peck KR, Ko KS, et al. Epidemiology and clinical outcomes of community-acquired pneumonia in adult patients in Asian countries: a prospective study by the Asian network for surveillance of resistant pathogens. Int J Antimicrob Agents. 2008;31:107–14.
Wagenvoort GHJ, Knol MJ, de Melker HE, Vlaminckx BJ, van der Ende A, Rozenbaum MH, et al. Risk and outcomes of invasive pneumococcal disease in adults with underlying conditions in the post-PCV7 era, The Netherlands. Vaccine. 2016;34:334–40.
Houseman C, Chapman KE, Manley P, Gorton R, Wilson D, Hughes GJ. Decreasing case fatality rate following invasive pneumococcal disease, North East England, 2006–2016. Epidemiol Infect. 2019;147:e175.
Acknowledgements
Combat Study Group. Principal Investigator: Xavier Duval. Steering Committee: Bruno Hoen, Bruno Mourvillier, Marie-Cécile Ploy, Sarah Tubiana, Emmanuelle Varon. Scientific committee: steering committee and the following members François Caron, Pierre-Edouard Bollaert, Olivier Gaillot, Muhamed-Kheir Taha, Claire Poyart, Stephane Bonacorsi, François Vandenesch, Emmanuelle Cambau, Marc Lecuit, Alain Gravet, Bruno Frachet, Thomas Debroucker, Daniel Levy Bruhl, François Raffi, Marie Preau. Combat Clinical Centers: Nadia Anguel, Laurent Argaud, Sophie Arista, Laurence Armand-Lefevre, Stéphanie Balavoine, Régine Baraduc, Guilène Barnaud, Guillaume Beraud, Louis Bernard, Georges Bernars, Dominique Bertei, Emilie Bessede, Typhaine Billard Pomares, Charlotte Biron, Stéphane Bland, Julien Boileau, Patrice Boubeau, Sandra Bourdon, Aurore Bousquet, Sophie Boyer, Alexis Bozorg-Grayeli, Laurent Bret, Cédric Bretonniere, François Bricaire, Elsa Brocas, Michel Brun, Jennifer Buret, Christophe Burucoa, Jean Cabalion, Mathieu Cabon, Emmanuelle Cambau, Guillaume Camuset, Christophe Canevet, François Caron, Anne Carricajo, Bernard Castan, Eric Caumes, Charles Cazanave, Amélie Chabrol, Thibaut Challan-Belval, Vanessa Chanteperdrix-Marillier, Chantal Chaplain, Caroline Charlier-Woerther, Hélène Chaussade, Catherine Chirouze, Bernard Clair, Julien Colot, Jean-Marie Conil, Hugues Cordel, Philippe Cormier, Joël Cousson, Pierrick Cronier, Eric Cua, Anne Dao-Dubremetz, Sylvie Dargere, Nicolas Degand, Sophie Dekeyser, Deborah Delaune, Eric Denes, Pierre-Francois Dequin, Diane Descamps, Elodie Descloux, Jean-Luc Desmaretz, Jean-Luc Diehl, Jérôme Dimet, Aurélien Dinh, Xavier Duval, Lelia Escaut, Claude Fabe, Frédéric Faibis, Clara Flateau, Nathalie Fonsale, Emmanuel Forestier, Nicolas Fortineau, Amandine Gagneux-Brunon, Caroline Garandeau, Magali Garcia, Denis Garot, Stéphane Gaudry, François Goehringer, Alain Gravet, Valérie Gregoire-Faucher, Marine Grosset, Camélia Gubavu, Isabelle Gueit, Dominique Guelon, Thomas Guimard, Jérôme Guinard, Tahar Hadou, Jean-Pierre Helene, Sandrine Henard, Benoit Henry, Anne-Cécile Hochart, Bruno Hoen, Gabriela Illes, Sylvain Jaffuel, Irène Jarrin, Françoise Jaureguy, Cédric Joseph, Marie-Emmanuelle Juvin, Samer Kayal, Solen Kerneis, Isabelle Lamaury, Philippe Lanotte, Etienne Laurens, Henri Laurichesse, Cécile Le Brun, Vincent Le Moing, Paul Le Turnier, Hervé Lecuyer, Sylvie Ledru, Céline Legrix, Adrien Lemaignen, Chantal Lemble, Ludovic Lemee, Olivier Lesens, Marion Levast, Claire Lhommet, Silvija Males, Edith Malpote, Guillaume Martin-Blondel, Matthieu Marx, Raphael Masson, Olivier Matray, Aurore Mbadi, Frédéric Mechai, Guillaume Mellon, Audrey Merens, Marie Caroline Meyohas, Adrien Michon, Joy Mootien Yoganaden, David Morquin, Stéphane Mouly, Natacha Mrozek, Sophie Nguyen, Yohan Nguyen, Maja Ogielska, Eric Oziol, Bernard Page, Solène Patrat-Delon, Isabelle Patry, André Pechinot, Sandrine Picot, Denys Pierrejean, Lionel Piroth, Claire Plassart, Patrice Plessis, Marie-Cécile Ploy, Laurent Portel, Patrice Poubeau, Marie Poupard, Claire Poyart, Thierry Prazuck, Luc Quaesaet, François Raffi, Adriatsiferana Ramanantsoa, Christophe Rapp, Laurent Raskine, Josette Raymond, Matthieu Revest, Agnès Riche, Stéphanie Robaday-Voisin, Frédéric Robin, Jean-Pierre Romaszko, Florence Rousseau, Anne-Laure Roux, Cécile Royer, Matthieu Saada, Dominique Salmon, Carlo Saroufim, Jean Luc Schmit, Manuela Sebire, Christine Segonds, Valérie Sivadon-Tardy, Nathalie Soismier, Olivia Son, Simon Sunder, Florence Suy, Didier Tande, Jacques Tankovic, Nadia Valin, Nicolas Van Grunderbeeck, François Vandenesch, Emmanuelle Varon, Renaud Verdon, Michel Vergnaud, Véronique Vernet-Garnier, Magali Vidal, Virginie Vitrat, Daniel Vittecoq, Fanny Vuotto. Coordination and statistical analyses (Clinical trial unit, Hôpitaux Universitaires Paris Nord Val de Seine, AP-HP, Paris) : Isabelle Gorenne, Cédric Laouenan, Estelle Marcault, France Mentre, Blandine Pasquet, Carine Roy, Sarah Tubiana. Scientific partnership: SPLIF, CMIT, SRLF, SFM, REIVAC, SFORL, APNET, SPF. Partners: ORP (Marie-Cécile PLOY), GPIP/ACTIV (Corinne Levy). Sponsor: DRCI APHP, Flore Lacassin. SIIP Study Group. Philippe Arsac, Martha Benoit, Louis Bernard, François Bissuel, Laurent Bret, Natalie Brieu, Jennifer Burret, Claudia Carvalhoschneider, Hélène Champagne, Joséphine Chapalain, Hubert Chardon, Pascal Chavanet, Judith Ducruet, Olivier Epaulard, Marc Fabre, Dominique Fasquelle, Emmanuel Forestier, Jean-Marc Galempoix, Guillaume Gautier, Claire Goulard De Curraize, Alain Gracet, Camélia Gubavu, Jerome Guinard, Tarik Habet, Aurélie Haudour, Caroline Henry, Cécile Hombrouckalet, Céline Janssen, Jean-Paul Kisteman, Philippe Lanotte, Marie-Frédérique Lartigue, Claire Launois, Cécile Lebrun, Laurence Legout, Marion Levast, Yannick Madoux, Laurence Maulin, Stéphanie Mestrallet, Abdo Mohareb, Joy Mootien, Yohan Nguyen, Franck Noel, Maja Ogielska, Agathe Ogier Desserrey, Anne Paleau, Simona Pavel, André Pechinot, Isabelle Pelloux, Camille Petillon, Hélène Petitprez, Bianca Podac, Jerome Poirot, Bénédicte Ponceau, Nathalie Prieur, Christine Recule, Orlando Saraceni, Jacques Sartre, Farid Sifaoui, Catherine Simonin, Christophe Strady, Charlotte Tellini, Anthony Texier, Maxime Thouvenin, Anne Tixier, Pauline Tremeaux, Pascale Verger, Véronique Vernet-Garnier, Jean-Pierre Verquin, Virginie Vitrat, Isabelle Vray, Oana Zamfir, Laure Zucchini.
Funding
The COMBAT study was supported by the Assistance Publique Hopitaux de Paris, French Ministry of Health, Inserm, SPILF, and the Pfizer Pharmaceutical company, which also funded ORP. The study was endorsed by the following professional organizations: Association Pour l’enseignement de la Thérapeutique, Société de Pathologie Infectieuse de Langue Française, Société Française de Microbiologie, Société Nationale Française de Médecine Interne, Société de Réanimation de Langue Française, Société Française de Gérontologie, Société Française d’Anesthésie-Réanimation. The SIIP study was supported by (i) the Société de Pathologie Infectieuse de Langue Fran.aise (SPILF; the French Infectious Diseases Society), (ii) Santé Publique France (SpFrance; the French National Public Health Agency), (iii) the European Centre for Disease Prevention and Control (ECDC), and (iv) Pfizer. The Regional Observatories of Pneumococci (Observatoires Régionaux du Pneumocoque) were supported by Pfizer, BioMerieux, and Sanofi.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors have no commercial or other associations that might pose a conflict of interest.
Additional information
The members of “COMBAT and SIIP Study Groups” are presented in the Acknowledgements section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Benadji, A., Duval, X., Danis, K. et al. Relationship between serotypes, disease characteristics and 30-day mortality in adults with invasive pneumococcal disease. Infection 50, 223–233 (2022). https://doi.org/10.1007/s15010-021-01688-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-021-01688-5