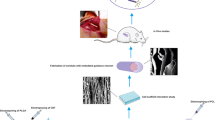
Abstract
Peripheral nerve injuries (PNIs) are one of the common causes of morbidity and disability worldwide. Autograft is considered the gold standard treatment for PNIs. However, due to the complications associated with autografts, other sources are considered as alternatives. Recently, electrospun nanofibrous scaffolds have received wide attention in the field of tissue engineering. Exogenous tubular constructs with uniaxially aligned topographical cues to enhance the axonal re-growth are needed to bridge large nerve gaps between proximal and distal ends. Although several studies have used PLGA/PCL, but few studies have been conducted on developing a two-layer scaffold with aligned fibers properly orientated along the axis direction of the sciatic nerve to meet the physical properties required for suturing, transplantation, and nerve regeneration. In this study, we sought to design and develop PLGA-PCL-aligned nanofibers. Following the conventional examinations, we implanted the scaffolds into 7-mm sciatic nerve gaps in a rat model of nerve injury. Our in vivo evaluations did not show any adverse effects, and after eight weeks, an acceptable improvement was noted in the electrophysiological, functional, and histological analyses. Thus, it can be concluded that nanofiber scaffolds can be used as a reliable approach for repairing PNIs. However, further research is warranted.







Similar content being viewed by others
Data availability
The data available to editors and peer reviewers if requested.
References
Bryan DJ, Holway AH, Wang KK, Silva AE, Trantolo DJ, Wise D, Summerhayes IC (2000) Influence of glial growth factor and Schwann cells in a bioresorbable guidance channel on peripheral nerve regeneration. Tissue Eng 6(2):129–138
Cao H, Liu T, Chew SY (2009) The application of nanofibrous scaffolds in neural tissue engineering. Adv Drug Deliv Rev 61(12):1055–1064
Chew SY, Mi R, Hoke A, Leong KW (2008) The effect of the alignment of electrospun fibrous scaffolds on Schwann cell maturation. Biomaterials 29(6):653–661
De Luca AC, Lacour SP, Raffoul W, Di Summa PG (2014) Extracellular matrix components in peripheral nerve repair: how to affect neural cellular response and nerve regeneration? Neural Regen Res 9(22):1943
Doyle LM, Roberts BL (2006) Exercise enhances axonal growth and functional recovery in the regenerating spinal cord. Neuroscience 141(1):321–327. https://doi.org/10.1016/j.neuroscience.2006.03.044
English AW, Schwartz G, Meador W, Sabatier MJ, Mulligan A (2007) Electrical stimulation promotes peripheral axon regeneration by enhanced neuronal neurotrophin signaling. Dev Neurobiol 67(2):158–172
Evans GRD, Brandt K, Katz S, Chauvin P, Otto L, Bogle M, Mikos AG (2002) Bioactive poly (L-lactic acid) conduits seeded with Schwann cells for peripheral nerve regeneration. Biomaterials 23(3):841–848
Ezra M, Bushman J, Shreiber D, Schachner M, Kohn J (2013) Enhanced femoral nerve regeneration after tubulization with a tyrosine-derived polycarbonate terpolymer: effects of protein adsorption and independence of conduit porosity. Tissue Eng A 20(3–4):518–528
Faroni A, Mobasseri SA, Kingham PJ, Reid AJ (2015) Peripheral nerve regeneration: experimental strategies and future perspectives. Adv Drug Deliv Rev 82–83:160–167. https://doi.org/10.1016/j.addr.2014.11.010
Frantz C, Stewart KM, Weaver VM (2010) The extracellular matrix at a glance. J Cell Sci 123(24):4195–4200
Fu SY, Gordon T (1997) The cellular and molecular basis of peripheral nerve regeneration. Mol Neurobiol 14(1–2):67–116
Guenard V, Kleitman N, Morrissey TK, Bunge RP, Aebischer P (1992) Syngeneic Schwann cells derived from adult nerves seeded in semipermeable guidance channels enhance peripheral nerve regeneration. J Neurosci 12(9):3310–3320
Hadlock T, Sundback C (2006) Biologically inspired approaches to drug delivery for nerve regeneration. Expert Opin Biol Ther 6(11):1105–1111
Hadlock T, Elisseeff J, Langer R, Vacanti J, Cheney M (1998) A tissue-engineered conduit for peripheral nerve repair. Arch Otolaryngol Head Neck Surg 124(10):1081–1086
Hall S (2005) The response to injury in the peripheral nervous system. J Bone Joint Surg Br 87(10):1309–1319. https://doi.org/10.1302/0301-620x.87b10.16700
Ichihara S, Inada Y, Nakamura T (2008) Artificial nerve tubes and their application for repair of peripheral nerve injury: an update of current concepts. Injury 39(Suppl 4):29–39. https://doi.org/10.1016/j.injury.2008.08.029
Jahromi Z, Mohammadghasemi F, Moharrami Kasmaie F, Zaminy A (2020) Cinnamaldehyde enhanced functional recovery after sciatic nerve crush injury in rats. Cells Tissues Organs 209(1):43–53. https://doi.org/10.1159/000507016
Kasemo B (2002) Biological surface science. Surf Sci 500(1–3):656–677
King VR, Henseler M, Brown RA, Priestley JV (2003) Mats made from fibronectin support oriented growth of axons in the damaged spinal cord of the adult rat. Exp Neurol 182(2):383–398
Konofaos P, Ver Halen JP (2013) Nerve repair by means of tubulization: past, present, future. J Reconstr Microsurg 29(03):149–164
Kou Y, Peng J, Wu Z, Yin X, Zhang P, Zhang Y, Jiang B (2013) Small gap sleeve bridging can improve the accuracy of peripheral nerve selective regeneration. Artif Cells Nanomed Biotechnol 41(6):402–407
Liu BS (2008) Fabrication and evaluation of a biodegradable proanthocyanidin-crosslinked gelatin conduit in peripheral nerve repair. J Biomed Mater Res A 87(4):1092–1102
Mohamadi F, Ebrahimi-Barough S, Reza Nourani M, Ali Derakhshan M, Goodarzi V, Sadegh Nazockdast M, Ai J (2017) Electrospun nerve guide scaffold of poly (ε-caprolactone)/collagen/nanobioglass: an in vitro study in peripheral nerve tissue engineering. J Biomedl Mater Res A 105(7):1960–1972
Moharrami Kasmaie F, Jahromi Z, Gazor R, Zaminy A (2019) Comparison of melatonin and curcumin effect at the light and dark periods on regeneration of sciatic nerve crush injury in rats. EXCLI J 18:653–665. https://doi.org/10.17179/excli2019-1369
Paino CL, Fernandez-Valle C, Bates ML, Bunge MB (1994) Regrowth of axons in lesioned adult rat spinal cord: promotion by implants of cultured Schwann cells. J Neurocytol 23(7):433–452
Panseri S, Cunha C, Lowery J, Del Carro U, Taraballi F, Amadio S, Gelain F (2008) Electrospun micro-and nanofiber tubes for functional nervous regeneration in sciatic nerve transections. BMC Biotechnol 8(1):39
Rodriguez FJ, Verdu E, Ceballos D, Navarro X (2000) Nerve guides seeded with autologous Schwann cells improve nerve regeneration. Exp Neurol 161(2):571–584
Rutkowski GE, Miller CA, Jeftinija S, Mallapragada SK (2004) Synergistic effects of micropatterned biodegradable conduits and Schwann cells on sciatic nerve regeneration. J Neural Eng 1(3):151
Sangsanoh P, Waleetorncheepsawat S, Suwantong O, Wutticharoenmongkol P, Weeranantanapan O, Chuenjitbuntaworn B, Supaphol P (2007) In vitro biocompatibility of schwann cells on surfaces of biocompatible polymeric electrospun fibrous and solution-cast film scaffolds. Biomacromol 8(5):1587–1594
Santos D, Gonzalez-Perez F, Giudetti G, Micera S, Udina E, Del Valle J, Navarro X (2016) Preferential enhancement of sensory and motor axon regeneration by combining extracellular matrix components with neurotrophic factors. Int J Mol Sci. https://doi.org/10.3390/ijms18010065
Schnell E, Klinkhammer K, Balzer S, Brook G, Klee D, Dalton P, Mey J (2007) Guidance of glial cell migration and axonal growth on electrospun nanofibers of poly-ε-caprolactone and a collagen/poly-ε-caprolactone blend. Biomaterials 28(19):3012–3025
Siemionow M, Sonmez E (2007) Nerve allograft transplantation: a review. J Reconstr Microsurg 23(8):511–520. https://doi.org/10.1055/s-2007-1022694
Subramanian A, Krishnan UM, Sethuraman S (2012) Fabrication, characterization and in vitro evaluation of aligned PLGA–PCL nanofibers for neural regeneration. Ann Biomed Eng 40(10):2098–2110
Walsh S, Midha R (2009) Practical considerations concerning the use of stem cells for peripheral nerve repair. Neurosurg Focus 26(2):E2. https://doi.org/10.3171/foc.2009.26.2.e2
Wang W, Itoh S, Konno K, Kikkawa T, Ichinose S, Sakai K, Watabe K (2009) Effects of Schwann cell alignment along the oriented electrospun chitosan nanofibers on nerve regeneration. J Biomed Mater Res A 91(4):994–1005. https://doi.org/10.1002/jbm.a.32329
Xie J, MacEwan MR, Willerth SM, Li X, Moran DW, Sakiyama-Elbert SE, Xia Y (2009) Conductive core–sheath nanofibers and their potential application in neural tissue engineering. Adv Funct Mater 19(14):2312–2318
Yang A, Huang Z, Yin G, Pu X (2015) Fabrication of aligned, porous and conductive fibers and their effects on cell adhesion and guidance. Colloids Surf B Biointerfaces 134:469–474
Yannas IV, Hill BJ (2004) Selection of biomaterials for peripheral nerve regeneration using data from the nerve chamber model. Biomaterials 25(9):1593–1600
Yu W, Zhao W, Zhu C, Zhang X, Ye D, Zhang W, Zhang Z (2011) Sciatic nerve regeneration in rats by a promising electrospun collagen/poly (ε-caprolactone) nerve conduit with tailored degradation rate. BMC Neurosci 12(1):68
Zamani F, Latifi M, Amani-Tehran M, Shokrgozar MA (2013) Effects of PLGA nanofibrous scaffolds structure on nerve cell directional proliferation and morphology. Fiber Polym 14(5):698–702
Zamani F, Amani-Tehran M, Latifi M, Shokrgozar MA, Zaminy A (2014) Promotion of spinal cord axon regeneration by 3D nanofibrous core–sheath scaffolds. J Biomed Mater Res A 102(2):506–513
Zamani F, Amani-Tehran M, Zaminy A, Shokrgozar MA (2017) Conductive 3D structure nanofibrous scaffolds for spinal cord regeneration. Fiber Polym 18(10):1874–1881
Zaminy A, Sayad-Fathi S, Kasmaie FM, Jahromi Z, Zendedel A (2021) Decellularized peripheral nerve grafts by a modified protocol for repair of rat sciatic nerve injury. Neural Regen Res 16:1086–1092
Zilic L, Wilshaw SP, Haycock JW (2016) Decellularisation and histological characterisation of porcine peripheral nerves. Biotechnol Bioeng 113(9):2041–2053. https://doi.org/10.1002/bit.25964
Funding
This research was fully supported by Vice-Chancellor for Research and Technology of Guilan University of Medical Sciences (Grant number: 97021503).
Author information
Authors and Affiliations
Contributions
Investigation and writing the draft: AZ, FZ; Conceptualization, formal analysis, and writing the draft: AZ, FZ; Data curation, methodology: FM-K and SS-F Writing–review and editing: AZ, FZ, SS-F, FM-K; supervision, project administration, funding acquisition: AZ.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest in this paper.
Ethical considerations
All procedures were approved by the Ethics Committee of Guilan University of Medical Sciences, Iran (IR.GUMS.REC.1397.52).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moharrami Kasmaie, F., Zamani, F., Sayad-Fathi, S. et al. Promotion of nerve regeneration by biodegradable nanofibrous scaffold following sciatic nerve transection in rats. Prog Biomater 10, 53–64 (2021). https://doi.org/10.1007/s40204-021-00151-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40204-021-00151-w