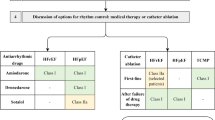
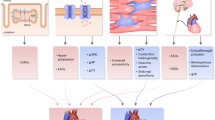
Abstract
Introduction
Rivaroxaban reduces the risk of thromboembolism in atrial fibrillation (AF) patients, who often also receive antiarrhythmic drugs (AADs) to maintain sinus rhythm. Current guidelines contraindicate concomitant use of rivaroxaban with the popular AAD dronedarone, despite little data demonstrating interactions with AADs. This study investigates the outcomes of concomitant rivaroxaban and AAD drug use in a real-world cohort.
Methods
This retrospective study included 1777 non-permanent AF patients taking rivaroxaban for ≥ 1 month between 2011 and 2016 from a multicenter cohort in Taiwan, and compared concomitant AAD use against clinical outcome endpoints for safety, effectiveness, and major adverse cardiac events (MACE). Multivariate Cox proportional hazard analyses were used to evaluate the association between concomitant AAD use and outcomes.
Results
Patients were divided into rivaroxaban alone (n = 1205) and with concomitant amiodarone (n = 177), dronedarone (n = 231), or propafenone (n = 164) groups. The proportion of patients using rivaroxaban 10 mg was highest in the concomitant dronedarone group: rivaroxaban alone, 53.6%; with amiodarone, 57.6%; with dronedarone, 77.1%; and with propafenone, 46.3% (p < 0.001). The cumulative incidences of safety (p = 0.892), effectiveness (p = 0.336), and MACE (p = 0.674) were similar between the four groups; however, there were significantly fewer new systemic thromboembolisms in the dronedarone group: rivaroxaban alone, 2.5%; with amiodarone, 0.6%; with dronedarone, 0%; and with propafenone, 1.2% (p = 0.029). The all-cause death rate was also lowest in the dronedarone group: rivaroxaban alone, 9.0%; with amiodarone, 9.6%; with dronedarone, 3.0%; and with propafenone: 6.1% (p = 0.013). After covariate adjustment, there were no differences in the safety, effectiveness, and MACE endpoints between patients receiving or not receiving AADs.
Conclusion
Concomitant use of rivaroxaban with AADs appears to be well tolerated, warranting further investigation into the apparent benefits of a reduced dose of rivaroxaban combined with dronedarone.



Similar content being viewed by others
References
Friberg L. Safety of apixaban in combination with dronedarone in patients with atrial fibrillation. Int J Cardiol. 2018;264:85–90. https://doi.org/10.1016/j.ijcard.2018.02.055.
Lin PL, Huang CC, Wu YJ, Su MI, Chiou WR, Liu LY, et al. Relations between baseline burden, maximum duration, and relative reduction of atrial fibrillation: Insights from continuous monitoring in rhythm control. J Cardiovasc Electrophysiol. 2019;30(2):178–82. https://doi.org/10.1111/jce.13824.
Hohnloser SH, Crijns HJ, van Eickels M, Gaudin C, Page RL, Torp-Pedersen C, et al. Effect of dronedarone on cardiovascular events in atrial fibrillation. N Engl J Med. 2009;360(7):668–78. https://doi.org/10.1056/NEJMoa0803778.
Trujillo TC, Nolan PE. Antiarrhythmic agents: drug interactions of clinical significance. Drug Saf. 2000;23(6):509–32. https://doi.org/10.2165/00002018-200023060-00003.
Szczerba E. Summary of the article: Patel MR, Mahaffey KW, Garg J et al Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:1557–9 ((Kardiol Pol. 2012;70(1):102-3)).
Steffel J, Verhamme P, Potpara TS, Albaladejo P, Antz M, Desteghe L, et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur Heart J. 2018;39(16):1330–93.
Escobar C, Arceluz M, de Oca MR, Mori R, López-Sendón JL, Merino JL. Concomitant rivaroxaban and dronedarone administration in patients with nonvalvular atrial fibrillation. Rev Esp Cardiol. 2017;70(2):121–2.
Schulman S, Kearon C, Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. 2005;3(4):692–4.
Kaatz S, Ahmad D, Spyropoulos A, Schulman S, Subcommittee on Control of Anticoagulation. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost. 2015;13(11):2119–26.
Celikyurt I, Meier CR, Kuhne M, Schaer B. Safety and interactions of direct oral anticoagulants with antiarrhythmic drugs. Drug Saf. 2017;40(11):1091–8. https://doi.org/10.1007/s40264-017-0567-5.
Abrams PJ, Emerson CR. Rivaroxaban: a novel, oral, direct factor Xa inhibitor. Pharmacotherapy. 2009;29(2):167–81. https://doi.org/10.1592/phco.29.2.167.
Heidbuchel H, Verhamme P, Alings M, Antz M, Hacke W, Oldgren J, et al. European Heart Rhythm Association Practical Guide on the use of new oral anticoagulants in patients with non-valvular atrial fibrillation. Europace. 2013;15(5):625–51.
Heidbuchel H, Verhamme P, Alings M, Antz M, Diener H-C, Hacke W, et al. Updated European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. Ep Europace. 2015;17(10):1467–507. https://doi.org/10.1093/europace/euv309.
Gnoth MJ, Buetehorn U, Muenster U, Schwarz T, Sandmann S. In vitro and in vivo P-glycoprotein transport characteristics of rivaroxaban. J Pharmacol Exp Ther. 2011;338(1):372–80.
Chang S-H, Chou I-J, Yeh Y-H, Chiou M-J, Wen M-S, Kuo C-T, et al. Association between use of non–vitamin K oral anticoagulants with and without concurrent medications and risk of major bleeding in nonvalvular atrial fibrillation. JAMA. 2017;318(13):1250–9.
Heist EK, Mansour M, Ruskin JN. Rate control in atrial fibrillation: targets, methods, resynchronization considerations. Circulation. 2011;124(24):2746–55. https://doi.org/10.1161/circulationaha.111.019919.
Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, Rosenberg Y, Schron EB, et al. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002;347(23):1825–33. https://doi.org/10.1056/NEJMoa021328.
Santangeli P, Di Biase L, Pelargonio G, Burkhardt JD, Natale A. The pharmaceutical pipeline for atrial fibrillation. Ann Med. 2011;43(1):13–32. https://doi.org/10.3109/07853890.2010.538431.
Freemantle N, Lafuente-Lafuente C, Mitchell S, Eckert L, Reynolds M. Mixed treatment comparison of dronedarone, amiodarone, sotalol, flecainide, and propafenone, for the management of atrial fibrillation. Europace. 2011;13(3):329–45. https://doi.org/10.1093/europace/euq450.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365(10):883–91.
Kirchhof P, Camm AJ, Goette A, Brandes A, Eckardt L, Elvan A, et al. Early rhythm-control therapy in patients with atrial fibrillation. N Engl J Med. 2020;383(14):1305–16.
Chiang C-E, Wu T-J, Ueng K-C, Chao T-F, Chang K-C, Wang C-C, et al. 2016 Guidelines of the Taiwan Heart Rhythm Society and the Taiwan Society of Cardiology for the management of atrial fibrillation. J Formos Med Assoc. 2016;115(11):893–952. https://doi.org/10.1016/j.jfma.2016.10.005.
Chan YH, See LC, Tu HT, Yeh YH, Chang SH, Wu LS, et al. Efficacy and safety of apixaban, dabigatran, rivaroxaban, and warfarin in asians with nonvalvular atrial fibrillation. J AmHeart Assoc. 2018;7(8):e008150. https://doi.org/10.1161/jaha.117.008150.
Lee HF, Chan YH, Tu HT, Kuo CT, Yeh YH, Chang SH, et al. The effectiveness and safety of low-dose rivaroxaban in Asians with non-valvular atrial fibrillation. Int J Cardiol. 2018;261:78–83. https://doi.org/10.1016/j.ijcard.2018.03.063.
Ikeda T, Ogawa S, Kitazono T, Nakagawara J, Minematsu K, Miyamoto S, et al. Real-world outcomes of the Xarelto Post-Authorization Safety & Effectiveness Study in Japanese Patients with Atrial Fibrillation (XAPASS). J Cardiol. 2019;74(1):60–6. https://doi.org/10.1016/j.jjcc.2019.01.001.
Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Eur Pacing Arrhyth Cardiac Electrophysiology. 2016;18(11):1609–78. https://doi.org/10.1093/europace/euw295.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflicts of interest/Competing interests
Wei-Ru Chiou, Chun-Che Huang, Po-Lin Lin, Jen-Yu Chuang, Lawrence Yu-Min Liu, Min-I Su, Feng-Ching Liao, Chun-Yen Chen, Jen-Yuan Kuo, Cheng-Ting Tsai, Yih-Jer Wu, and Ying-Hsiang Lee declare they have no potential conflicts of interest that might be relevant to the contents of this manuscript.
Ethics approval
The Institutional Review Board of MacKay Memorial Hospital approved the study protocol (IRB No. 16MMHIS009), which waived the requirement for informed consent in this retrospective study.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Author contributions
W-RC: Conceptualization, primary author, review and editing, data collection, data interpretation, data resources. C-CH: Data analysis, data interpretation, writing – review and editing. P-LL: Conceptualization, data collection, data resources. J-YC: Writing – original draft. Lawrence Y-ML: Data collection, data resources. M-IS: Conceptualization, data resources. F-CL: Data collection, data resources. C-YC: Data resources. J-YK: Data resources. C-TT: Data resources. Y-JW: Supervision, data resources. Y-HL: Conceptualization, project administration, writing – review and editing, methodology, data collection, data resources.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chiou, WR., Huang, CC., Lin, PL. et al. Safety and Effectiveness of Rivaroxaban in Combination with Various Antiarrhythmic Drugs in Patients with Non-Permanent Atrial Fibrillation. Am J Cardiovasc Drugs 21, 459–469 (2021). https://doi.org/10.1007/s40256-020-00454-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40256-020-00454-6