Abstract
Background
Based on reassuring short-term foetal and maternal safety data, there is an increasing trend to administer chemotherapy during the second and third trimesters of pregnancy. The pharmacokinetics (PK) of drugs might change as a result of several physiological changes that occur during pregnancy, potentially affecting the efficacy and safety of chemotherapy.
Objective
With this analysis, we aimed to quantitatively describe the changes in the PK of docetaxel, paclitaxel, doxorubicin and epirubicin in pregnant women compared with non-pregnant women.
Methods
PK data from 9, 20, 22 and 16 pregnant cancer patients from the International Network of Cancer, Infertility and Pregnancy (INCIP) were available for docetaxel, paclitaxel, doxorubicin and epirubicin, respectively. These samples were combined with available PK data from non-pregnant patients. Empirical non-linear mixed-effects models were developed, evaluating fixed pregnancy effects and gestational age as covariates.
Results
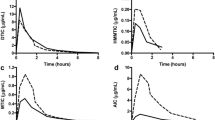
Overall, 82, 189, 271, and 227 plasma samples were collected from pregnant patients treated with docetaxel, paclitaxel, doxorubicin and epirubicin, respectively. The plasma PK data were adequately described by the respective models for all cytotoxic drugs. Typical increases in central and peripheral volumes of distribution of pregnant women were identified for docetaxel, paclitaxel, doxorubicin and epirubicin. Additionally, docetaxel, doxorubicin and paclitaxel clearance were increased in pregnant patients, resulting in lower exposure in pregnant women compared with non-pregnant patients.
Conclusion
Given the interpatient variability, the identified pregnancy-induced changes in PK do not directly warrant dose adjustments for the studied drugs. Nevertheless, these results underscore the need to investigate the efficacy of chemotherapy, when administered during pregnancy.


Similar content being viewed by others
References
FDA Guidance for Industry – Pharmaockinetics in Pregnancy – Study Design, Data Analysis, and Impact on Dosing and Labeling. FDA Guidance. US FDA; 2004.
FDA Guidance for Industry – Establishing Pregnancy Exposure Registries. FDA Guidance. US FDA; 2002.
European Medicines Agency (EMA). Guideline on the exposure to medicinal products during pregnancy: need for post-authorization data. London: European Medicines Agency; 2006.
de Haan J, Verheecke M, Van Calsteren K, Van CB, Shmakov RG, Gziri MM, et al. Oncological management and obstetric and neonatal outcomes for women diagnosed with cancer during pregnancy: a 20-year international cohort study of 1170 patients. Lancet Oncol. 2018;19:337–46.
Amant F, Vandenbroucke T, Verheecke M, Fumagalli M, Halaska MJ, Boere I, et al. Pediatric outcome after maternal cancer diagnosed during pregnancy. N Engl J Med. 2015;373:1824–34.
Abduljalil K, Furness P, Johnson TN, Rostami-Hodjegan A, Soltani H. Anatomical, physiological and metabolic changes with gestational age during normal pregnancy. Clin Pharmacokinet. 2012;51:365–96.
Van Calsteren K, Verbesselt R, Ottevanger N, Halaska M, Heyns L, Van Bree R, et al. Pharmacokinetics of chemotherapeutic agents in pregnancy: a preclinical and clinical study. Acta Obstet Gynecol Scand. 2010;89:1338–45.
van Hasselt JGC, Van Calsteren K, Heyns L, Han S, Mhallem Gziri M, Schellens JHM, et al. Optimizing anticancer drug treatment in pregnant cancer patients: pharmacokinetic analysis of gestation-induced changes for doxorubicin, epirubicin, docetaxel and paclitaxel. Ann Oncol. 2014;25:2059–65.
Hendrikx JJMA, Hillebrand MJX, Thijssen B, Rosing H, Schinkel AH, Schellens JHM, et al. A sensitive combined assay for the quantification of paclitaxel, docetaxel and ritonavir in human plasma using liquid chromatography coupled with tandem mass spectrometry. J Chromatogr B. 2011;879:2984–90.
Beijnen JH, Meenhorst PL, Van Gijn R, Fromme M, Rosing H, Underberg WJM. HPLC determination of doxorubicin, doxorubicinol and four aglycone metabolites in plasma of AIDS patients. J Pharm Biomed Anal. 1991;9:995–1002.
Crombag M-RBS, de Vries Schultink AHM, Koolen SLW, Wijngaard S, Joerger M, Schellens JHM, et al. Impact of older age on the exposure of paclitaxel: a population pharmacokinetic study. Pharm Res. 2019;36:33.
West GB, Brown JH, Enquist BJ. A general model for the origin of allometric scaling laws in biology. Science. 1997;276:122–6.
Nguyen T, Mouksassi M-S, Holford N, Al-Huniti N, Freedman I, Hooker A, et al. Model evaluation of continuous data pharmacometric models: metrics and graphics. CPT Pharmacomet Syst Pharmacol. 2017;6:87–109.
Dosne A-G, Bergstrand M, Karlsson MO. An automated sampling importance resampling procedure for estimating parameter uncertainty. J Pharmacokinet Pharmacodyn. 2017;44:509–20.
Lindbom L, Ribbing J, Jonsson EN. Perl-speaks-NONMEM (PsN)—a Perl module for NONMEM related programming. Comput Methods Programs Biomed. 2004;75:85–94.
Keizer RJ, van Benten M, Beijnen JH, Schellens JHM, Huitema ADR. Pirana and PCluster: a modeling environment and cluster infrastructure for NONMEM. Comput Methods Programs Biomed. 2011;101:72–9.
Beal S, Boeckmann A, Sheiner L. NONMEM user guides. San Francisco: University of California; 1988.
RC Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2009.
Vandenbroucke T, Verheecke M, Fumagalli M, Lok C, Amant F. Effects of cancer treatment during pregnancy on fetal and child development. Lancet Child Adolesc Health. 2017;1:302–10.
Joerger M, Huitema ADR, Richel DJ, Dittrich C, Pavlidis N, Briasoulis E, et al. Population pharmacokinetics and pharmacodynamics of doxorubicin and cyclophosphamide in breast cancer patients: a study by the EORTC-PAMM-NDDG. Clin Pharmacokinet. 2007;46:1051–68.
Kontny NE, Würthwein G, Joachim B, Boddy AV, Krischke M, Fuhr U, et al. Population pharmacokinetics of doxorubicin: establishment of a NONMEM model for adults and children older than 3 years. Cancer Chemother Pharmacol. 2013;71:749–63.
Bruno R, Vivier N, Veyrat-Follet C, Montay G, Rhodes GR. Population pharmacokinetics and pharmacokinetic-pharmacodynamic relationships for docetaxel. Invest New Drugs. 2001;19:163–9.
Sandstrom M, Lindman H, Nygren P, Johansson M, Bergh J, Karlsson M. Population analysis of the pharmacokinetics and the haematological toxicity of the fluorouracil-epirubicin-cyclophosphamide regimen in breast cancer patients. Cancer Chemother Pharmacol. 2006;58:143–56.
Graham JW. Missing data analysis: making it work in the real world. Annu Rev Psychol. 2009;60:549–76.
Schafer JL, Graham JW. Missing data: our view of the state of the art. Psychol Methods. 2002;7:147–77.
Ince DI, Eissing T, Hempel G. gestation-specific changes in the anatomy and physiology of healthy pregnant women : an extended repository of model parameters for physiologically based pharmacokinetic modeling in pregnancy. Clin Pharmacokinet. 2017;56:1303–30.
Amant F, Von Minckwitz G, Han SN, Bontenbal M, Ring AE, Giermek J, et al. Prognosis of women with primary breast cancer diagnosed during pregnancy: results from an international collaborative study. J Clin Oncol. 2013;31:2532–9.
Ploquin A, Pistilli B, Tresch E, Frenel JS, Lerebours F, Lesur A, et al. 5-year overall survival after early breast cancer diagnosed during pregnancy: a retrospective case-control multicentre French study. Eur J Cancer. 2018;95:30–7.
Maggen C, Dierickx D, Lugtenburg P, Laenen A, Cardonick E, Smakov R, et al. Obstetric and maternal outcomes in patients diagnosed with Hodgkin lymphoma during pregnancy: a multicentre, retrospective, cohort study. Lancet Haematol. 2019;6:e551–61.
Koolen SLW, Oostendorp RL, Beijnen JH, Schellens JHM, Huitema ADR. Population pharmacokinetics of intravenously and orally administered docetaxel with or without co-administration of ritonavir in patients with advanced cancer. Br J Clin Pharmacol. 2010;69:465–74.
Acknowledgements
The authors thank Liesbeth Leemans and Katrien Van Tornout for sample collections, and the Research HPC Facility of the Netherlands Cancer Institute for support in the use of computational resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Funding for the INCIP registry, sample bioanalysis and sample logistics was provided by the European Research Council under the European Union’s Horizon 2020 research and innovation programme (grant agreement number 647047), the Research Foundation-Flanders and the Belgian Cancer Plan, Ministry of Health, Belgium.
Conflicts of Interest
Frédéric Amant is a senior clinical investigator for the Research Fund-Flanders; Kristel Van Calsteren received a clinical research fund from the University Hospitals Leuven; Thomas Dorlo was personally supported by a Dutch Research Council (NWO)/ZonMw Veni grant; Michael J. Halaska was supported by the Charles University research project Progres Q28 and Q34; and Jos Beijnen is a part-time employee, patent holder (partly) and stock holder (indirectly) of Modra Pharmaceuticals BV, a spin-out company developing oral taxane formulations and therapies, which is not related to the submitted work. Julie M. Janssen, Robert Fruscio, Petronella Ottevanger, Carolien P. Schröder, Ingrid Boere, Petronella O. Witteveen, Rebecca C. Painter, Ruud Bekkers, Vit Drochytek and Alwin D.R. Huitema have no conflicts of interest to declare.
Ethics approval
The data used in this study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by institutional review boards and independent ethics committees at participating 137 institutions.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
All individual participants signed informed consent regarding publishing their data.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Author contributions
JJ, TD and AH performed the analysis, data interpretation and writing of the manuscript. KVC and FA designed the study, included patients, contributed to the writing of the manuscript, and provided financial support. MH, RF, PO, CS, IB, PW, RP, RB and VD included patients and contributed to the writing of the manuscript. JB contributed to drug analysis and writing of the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Janssen, J.M., Van Calsteren, K., Dorlo, T.P.C. et al. Population Pharmacokinetics of Docetaxel, Paclitaxel, Doxorubicin and Epirubicin in Pregnant Women with Cancer: A Study from the International Network of Cancer, Infertility and Pregnancy (INCIP). Clin Pharmacokinet 60, 775–784 (2021). https://doi.org/10.1007/s40262-020-00961-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-020-00961-4