Abstract
Introduction
Atrial fibrillation (AF) is a major cause of stroke. Anticoagulants substantially reduce risk of stroke but are also associated with an increased risk of bleeding. Because of that, many patients do not receive anticoagulants, particularly patients at risk of falls. This systematic review and meta-analysis aims to compare anticoagulant treatment options for the management of atrial fibrillation patients at risk of falls or with a history of falls.
Methods
We conducted a PRISMA systematic review (until March 2022), including studies evaluating safety and efficacy of different anticoagulants (vitamin K antagonist [VKA] versus non-vitamin K antagonist oral anticoagulant [NOAC]). Outcomes were ischemic stroke, major bleeding, intracranial hemorrhage, hemorrhagic stroke, myocardial infarction, gastrointestinal bleeding, cardiovascular and all-cause mortality. A multilevel meta-analysis was conducted adjusting for clustering effects within studies examining more than one effect size.
Results
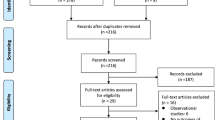
A total of 919 articles were identified, 848 after removing duplicates. The full text of 155 were screened and 10 articles were retained for final quantitative synthesis. Risk of bias was moderate to serious for the included studies. In meta-analysis, NOACs were associated with superior effectiveness compared with VKA for ischemic stroke/systemic embolism (hazard ratio [HR] 0.82, 95% confidence interval [CI] 0.69–0.98; p < 0.05) and safety (HR 0.53, 95% CI 0.40–0.71; p < 0.05) for intracranial hemorrhage. There were no differences in other outcomes.
Conclusion
NOACs were associated with less intracranial hemorrhages and ischemic strokes/systemic embolisms than VKAs in AF patients at risk of falls. These findings suggesting preferred use of NOACs over VKAs have clinical implications for physicians, patients and policy makers.



Similar content being viewed by others
References
Markides V, Schilling RJ. Atrial fibrillation: classification, pathophysiology, mechanisms and drug treatment. Heart. 2003;89:939–43.
Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, Benjamin EJ, et al. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation. 2014;129:837–47.
Burdett P, Lip GYH. Atrial fibrillation in the UK: predicting costs of an emerging epidemic recognizing and forecasting the cost drivers of atrial fibrillation-related costs. Eur Heart J Qual Care Clin Outcomes. 2022;8:187–94.
Katan M, Luft A. Global burden of stroke. Semin Neurol. 2018;38:208–11.
Adamson J, Beswick A, Ebrahim S. Is stroke the most common cause of disability? J Stroke Cerebrovasc Dis. 2004;13:171–7.
Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized controlled trials. Arch Intern Med. 1994;154:1449.
Staerk L, Sherer JA, Ko D, Benjamin EJ, Helm RH. Atrial fibrillation: epidemiology, pathophysiology, and clinical outcomes. Circ Res. 2017;120:1501–17.
Katsanos AH, Schellinger PD, Köhrmann M, Filippatou A, Gurol ME, Caso V, et al. Fatal oral anticoagulant-related intracranial hemorrhage: a systematic review and meta-analysis. Eur J Neurol. 2018;25:1299–302.
Kakkar AK, Mueller I, Bassand J-P, Fitzmaurice DA, Goldhaber SZ, Goto S, et al. Risk profiles and antithrombotic treatment of patients newly diagnosed with atrial fibrillation at risk of stroke: perspectives from the international, observational, prospective GARFIELD registry. PLoS ONE. 2013;8: e63479.
Bahri O, Roca F, Lechani T, Druesne L, Jouanny P, Serot J-M, et al. Underuse of oral anticoagulation for individuals with atrial fibrillation in a nursing home setting in France: comparisons of resident characteristics and physician attitude. J Am Geriatr Soc. 2015;63:71–6.
Sussman M, Barnes GD, Guo JD, Tao CY, Gillespie JA, Ferri M, et al. The burden of undertreatment and non-treatment among patients with non-valvular atrial fibrillation and elevated stroke risk: a systematic review. Curr Med Res Opin. 2022;38:7–18.
Garwood CL, Corbett TL. Use of anticoagulation in elderly patients with atrial fibrillation who are at risk for falls. Ann Pharmacother. 2008;42:523–32.
Donzé J, Clair C, Hug B, Rodondi N, Waeber G, Cornuz J, et al. Risk of falls and major bleeds in patients on oral anticoagulation therapy. Am J Med. 2012;125:773–8.
Man-Son-Hing M, Nichol G, Lau A, Laupacis A. Choosing antithrombotic therapy for elderly patients with atrial fibrillation who are at risk for falls. Arch Intern Med. 1999;159:677–85.
Lip GYH, Frison L, Halperin JL, Lane DA. Comparative validation of a novel risk score for predicting bleeding risk in anticoagulated patients with atrial fibrillation. J Am Coll Cardiol. 2011;57:173–80.
Gorog DA, Gue YX, Chao T-F, Fauchier L, Ferreiro JL, Huber K, et al. Assessment and mitigation of bleeding risk in atrial fibrillation and venous thromboembolism: a position paper from the ESC Working Group on Thrombosis, in collaboration with the European Heart Rhythm Association, the Association for Acute CardioVascular Care and the Asia-Pacific Heart Rhythm Society [published online ahead of print, 2022 Mar 22]. EP Europace. 2022; p. euac020.
Caldeira D, Barra M, Pinto FJ, Ferreira JJ, Costa J. Intracranial hemorrhage risk with the new oral anticoagulants: a systematic review and meta-analysis. J Neurol. 2015;262:516–22.
Hicks T, Stewart F, Eisinga A. NOACs versus warfarin for stroke prevention in patients with AF: a systematic review and meta-analysis. Open Heart. 2016;3: e000279.
Chao T-F, Joung B, Takahashi Y, Lim TW, Choi E-K, Chan Y-H, et al. 2021 Focused update consensus guidelines of the Asia Pacific heart rhythm society on stroke prevention in atrial fibrillation: executive summary. Thromb Haemost. 2022;122:020–47.
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2021;42:373–498.
Grymonprez M, Steurbaut S, De Backer TL, Petrovic M, Lahousse L. Effectiveness and safety of oral anticoagulants in older patients with atrial fibrillation: a systematic review and meta-analysis. Front Pharmacol. 2020;11: 583311.
Galvain T, Hill R, Donegan S, Lisboa P, Lip GYH, Czanner G. The management of anticoagulants in patients with atrial fibrillation and history of falls or risk of falls: protocol for a systematic review and meta-analysis. Syst Rev. 2022;11:63.
Higgins J, Thomas J, Chandler J, Cumpston M, Li T, Page M, et al. Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021) [Internet]. Cochrane, 2021. www.training.cochrane.org/handbook. Accessed 28 Jan 2022.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, The PRISMA, et al. statement: an updated guideline for reporting systematic reviews. BMJ. 2020;2021: n71.
Steffel J, Giugliano RP, Braunwald E, Murphy SA, Mercuri M, Choi Y, et al. Edoxaban versus warfarin in atrial fibrillation patients at risk of falling: ENGAGE AF–TIMI 48 analysis. J Am Coll Cardiol. 2016;68:1169–78.
Schulman S, Kearon C, the SUBCOMMITTEE ON CONTROL OF ANTICOAGULATION OF THE SCIENTIFIC AND STANDARDIZATION COMMITTEE OF THE INTERNATIONAL SOCIETY ON THROMBOSIS AND HAEMOSTASIS. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients: Definitions of major bleeding in clinical studies. J Thromb Haemost. 2005;3:692–4.
Kaatz S, Ahmad D, Spyropoulos AC, Schulman S, the Subcommittee on Control of Anticoagulation. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost. 2015;13:2119–26.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919.
Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Assink M, Wibbelink CJM. Fitting three-level meta-analytic models in R: a step-by-step tutorial. TQMP. 2016;12:154–74.
Higgins JP, Eldridge S, Li T, et al. Including variants on randomized trials. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al., editors. Cochrane handbook for systematic reviews of interventions [Internet]. 1st ed. Wiley; 2019. https://doi.org/10.1002/9781119536604.ch23.
Pustejovsky J, Tipton E. Meta-analysis with robust variance estimation: expanding the range of working models. Prev Sci. 2022;23(3):425–38.
Pustejovsky JE, Tipton E. Small-sample methods for cluster-robust variance estimation and hypothesis testing in fixed effects models. J Bus Econ Stat. 2018;36:672–83.
Rodgers MA, Pustejovsky JE. Evaluating meta-analytic methods to detect selective reporting in the presence of dependent effect sizes [published online ahead of print, 2020 Jul 16]. Psychol Methods. 2020.
Harrer M, Cuijpers P, Ebert D. Doing Meta-Analysis in R. 2019. https://bookdown.org/MathiasHarrer/Doing_Meta_Analysis_in_R/. Accessed 23 May 2022.
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–6.
Schünemann HJ, Cuello C, Akl EA, Mustafa RA, Meerpohl JJ, Thayer K, et al. GRADE Guidelines: 18. How ROBINS-I and other tools to assess risk of bias in non-randomized studies should be used to rate the certainty of a body of evidence. J Clin Epidemiol. 2019;111:105–14.
GRADEpro GDT: GRADEpro Guideline Development Tool [Software]. McMaster University and Evidence Prime, 2021. https://www.gradepro.org/. Accessed 10 May 2022.
Rao MP, Vinereanu D, Wojdyla DM, Alexander JH, Atar D, Hylek EM, et al. Clinical outcomes and history of fall in patients with atrial fibrillation treated with oral anticoagulation: insights from the ARISTOTLE Trial. Am J Med. 2018;131:269-275.e2.
Miao B, Alberts MJ, Bunz TJ, Coleman CI. Safety and effectiveness of oral factor Xa inhibitors versus warfarin in nonvalvular atrial fibrillation patients at high-risk for falls. J Thromb Thrombolysis. 2019;48:366–72.
Jaspers Focks J, Brouwer MA, Wojdyla DM, Thomas L, Lopes RD, Washam JB, et al. Polypharmacy and effects of apixaban versus warfarin in patients with atrial fibrillation: post hoc analysis of the ARISTOTLE trial. BMJ. 2016;353:i2868.
Alexander KP, Brouwer MA, Mulder H, Vinereanu D, Lopes RD, Proietti M, et al. Outcomes of apixaban versus warfarin in patients with atrial fibrillation and multi-morbidity: Insights from the ARISTOTLE trial. Am Heart J. 2019;208:123–31.
Fanning L, Lau WCY, Mongkhon P, Man KKC, Bell JS, Ilomäki J, et al. Safety and effectiveness of direct oral anticoagulants vs warfarin in people with atrial fibrillation and dementia. J Am Med Dir Assoc. 2020;21:1058-1064.e6.
Piccini JP, Hellkamp AS, Washam JB, Becker RC, Breithardt G, Berkowitz SD, et al. Polypharmacy and the efficacy and safety of rivaroxaban versus warfarin in the prevention of stroke in patients with nonvalvular atrial fibrillation. Circulation. 2016;133:352–60.
Lip GYH, Keshishian AV, Kang AL, Dhamane AD, Luo X, Li X, et al. Oral anticoagulants for nonvalvular atrial fibrillation in frail elderly patients: insights from the ARISTOPHANES study. J Intern Med. 2021;289:42–52.
Martinez BK, Sood NA, Bunz TJ, Coleman CI. Effectiveness and safety of apixaban, dabigatran, and rivaroxaban versus warfarin in frail patients with nonvalvular atrial fibrillation. JAHA. 2018;7: e008643.
Hohmann C, Hohnloser SH, Jacob J, Walker J, Baldus S, Pfister R. Non-vitamin K oral anticoagulants in comparison to phenprocoumon in geriatric and non-geriatric patients with non-valvular atrial fibrillation. Thromb Haemost. 2019;119:971–80.
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;369:2093–104.
Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981–92.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883–91.
January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64:e1-76.
January CT, Wann LS, Calkins H, Chen LY, Cigarroa JE, Cleveland JC, et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society in Collaboration With the Society of Thoracic Surgeons. Circulation [Internet]. 2019. https://doi.org/10.1161/CIR.0000000000000665.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139–51.
López-López JA, Sterne JAC, Thom HHZ, Higgins JPT, Hingorani AD, Okoli GN, et al. Oral anticoagulants for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis, and cost effectiveness analysis. BMJ. 2017;359: j5058.
Ruff CT, Giugliano RP, Braunwald E, Hoffman EB, Deenadayalu N, Ezekowitz MD, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383:955–62.
Ntaios G, Papavasileiou V, Diener H-C, Makaritsis K, Michel P. Nonvitamin-K-antagonist oral anticoagulants versus warfarin in patients with atrial fibrillation and previous stroke or transient ischemic attack: An updated systematic review and meta-analysis of randomized controlled trials. Int J Stroke. 2017;12:589–96.
Makam RCP, Hoaglin DC, McManus DD, Wang V, Gore JM, Spencer FA, et al. Efficacy and safety of direct oral anticoagulants approved for cardiovascular indications: systematic review and meta-analysis. PLoS ONE. 2018;13: e0197583.
Bai Y, Guo S-D, Deng H, Shantsila A, Fauchier L, Ma C-S, et al. Effectiveness and safety of oral anticoagulants in older patients with atrial fibrillation: a systematic review and meta-regression analysis. Age Ageing. 2018;47:9–17.
Bai Y, Deng H, Shantsila A, Lip GYH. Rivaroxaban versus dabigatran or warfarin in real-world studies of stroke prevention in atrial fibrillation. Stroke. 2017;48:970–6.
Almutairi AR, Zhou L, Gellad WF, Lee JK, Slack MK, Martin JR, et al. Effectiveness and safety of non-vitamin K antagonist oral anticoagulants for atrial fibrillation and venous thromboembolism: a systematic review and meta-analyses. Clin Ther. 2017;39:1456-1478.e36.
Mitchell A, Watson MC, Welsh T, McGrogan A. Effectiveness and safety of direct oral anticoagulants versus vitamin K antagonists for people aged 75 years and over with atrial fibrillation: a systematic review and meta-analyses of observational studies. JCM. 2019;8:554.
Douros A, Renoux C, Coulombe J, Suissa S. Patterns of long-term use of non-vitamin K antagonist oral anticoagulants for non-valvular atrial fibrillation: Quebec observational study. Pharmacoepidemiol Drug Saf. 2017;26:1546–54.
Acknowledgements
We would like to acknowledge Donna Burgess and Carolyn Benny, both librarians, for their useful reviews and expert advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
TG’s PhD education program is funded by Johnson and Johnson Medical. The funder was not involved in the research, nor in any decisions taken by the authors.
Competing interests
TG is an employee of Johnson and Johnson Medical. RH declares a financial, non-personal, non-specific interest, having delivered educational workshops on health economics, medicines management and HTA for cancer specialists supported by unrestricted sponsorship by the pharmaceutical industry and an industry association (March 2019). No fees received personally. Not specific to the topic of the review. GYHL has served as a consultant for Bayer/Janssen, Bristol Myers Squibb (BMS)/Pfizer, Biotronik, Medtronic, Boehringer Ingelheim, Novartis, Verseon and Daiichi-Sankyo and as a speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim and Daiichi-Sankyo. The other authors report no competing interests (SD, PL and GC).
Ethics approval and consent to participate
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Template data collection form, data extracted, analytical code and any other study documents will be available from the corresponding author on reasonable request.
Code availability
Analytical code will be available from the corresponding author on reasonable request.
Authors' contributions
Conceptualization: GYHL, PL, GC, SD, RH and TG; methodology: GYHL, PL, GC, SD, RH and TG; formal analysis: GC and TG; investigation: GYHL, PL, GC, SD, RH and TG; data curation: GC and TG; writing original draft: TG; writing review and editing: GYHL, PL, GC, SD, RH and TG; visualization: TG; final approval: GYHL, PL, GC, SD, RH and TG. All authors read and approved the final version of the manuscript.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galvain, T., Hill, R., Donegan, S. et al. Efficacy and Safety of Anticoagulants in Patients with Atrial Fibrillation and History of Falls or Risk of Falls: A Systematic Review and Multilevel Meta-Analysis. Drug Saf 45, 1349–1362 (2022). https://doi.org/10.1007/s40264-022-01231-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-022-01231-x