Abstract
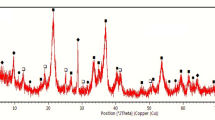
The mineralogical characterization and occurrence of target elements in limonitic laterite from Africa was comprehensively studied by X-ray diffraction (XRD), scanning electron microscopy energy spectrum (SEM–EDS), and optical microscope (OM) measurements. The results suggest that laterite ore has a complex internal structure. In the limonitic laterite, the contents of Ni, Co, and Fe are 1.14%, 0.16%, and 45.91%, respectively. The limonitic laterite mainly contains 72.2% iron oxide/oxyhydroxide, 9.37% silicate, 6.22% chlorite. Limonite, chlorite, and manganese oxide are the main nickel carrier with distribution rates of 60.98%, 22.65%, and 13.94%, respectively. Cobalt is hosted in manganese oxides and iron oxides, respectively. Additionally, Au, Pt, Pd, and Ag are also detected in the limonitic laterite. Gold, platinum, and silver are mainly found in their native metals. Palladium is mainly dispersed in the iron minerals. Furthermore, the route of rotary kiln-electric furnace (RKEF) was selected to this limonitic laterite; the results demonstrate that comprehensive recovery efficiency of Ni, Co, Fe is high with recovery rate of 98.72%, 98.38%, 54.89%, respectively. Notably, the majority of noble metals such as Au, Pt, Pd are enriched in crude ferronickel.
Graphical Abstract














Similar content being viewed by others
References
Johnson J, Reck B, Wang T, Graedel T (2008) The energy benefit of stainless steel recycling. Energy policy 36(1):181–192. https://doi.org/10.1016/j.enpol.2007.08.028
Kim J, Dodbiba G, Tanno H, Okaya K, Matsuo S, Fujita T (2010) Calcination of low-grade laterite for concentration of Ni by magnetic separation. Miner Eng 23(4):282–288. https://doi.org/10.1016/j.mineng.2010.01.005
Innocenzi V, Ippolito NM, De Michelis I, Prisciandaro M, Medici F, Vegliò F (2017) A review of the processes and lab-scale techniques for the treatment of spent rechargeable NiMH batteries. J Power Sourc 362:202–218. https://doi.org/10.1016/j.jpowsour.2017.07.034
Schipper F, Erickson EM, Erk C, Shin J-Y, Chesneau FF, Aurbach D (2016) Review—recent advances and remaining challenges for lithium ion battery cathodes. J Electrochem Soc 164(1):A6220–A6228. https://doi.org/10.1149/2.0351701jes
Ma B, Yang W, Yang B, Wang C, Chen Y, Zhang Y (2015) Pilot-scale plant study on the innovative nitric acid pressure leaching technology for laterite ores. Hydrometallurgy 155:88–94. https://doi.org/10.1016/j.hydromet.2015.04.016
Ober JA (2018) Mineral commodity summaries 2018. Mineral Commodity Summaries, Reston. https://doi.org/10.3133/70194932-6
Chen P, Chen T, Xie Q, Xu L, Liu H, Zhou Y (2018) Mineralogy and geochemistry of limonite as a weathering product of ilvaite in the yeshan iron deposit, Tongling, China. Clays Clay Miner 66(2):190–207. https://doi.org/10.1346/CCMN.2018.064102a
Pariser HH, Backeberg NR, Masson OCM, Bedder JCM (2018) Changing nickel and chromium stainless steel markets—a review. J South Afr Inst Min Metall 118:563–568. https://doi.org/10.17159/2411-9717/2018/v118n6a1
Zeng X, Xu M, Li J (2018) Examining the sustainability of China’s nickel supply: 1950–2050. Resourc Conserv Recycl 139:188–193. https://doi.org/10.1016/j.resconrec.2018.08.011
Chen P, Chen T, Xu L, Liu H, Xie Q (2017) Mn-rich limonite from the yeshan iron deposit, Tongling District, China: a natural nanocomposite. J Nanosci Nanotechnol 17(9):6931–6935. https://doi.org/10.1166/jnn.2017.14410
Jiang Y, Hou M-X (2008) The study of global nickel supply and demand. Non-ferr Min Metall 24(2):55–57
Zevgolis E, Zografidis C, Halikia I (2010) The reducibility of the Greek nickeliferous laterites: a review. Miner Process Extract Metall 119(1):9–17. https://doi.org/10.1179/174328509X431472
Fan R, Gerson AR (2011) Nickel geochemistry of a Philippine laterite examined by bulk and microprobe synchrotron analyses. Geochim et Cosmochim Acta 75(21):6400–6415. https://doi.org/10.1016/j.gca.2011.08.003
Dalvi AD, Bacon WG, Osborne RC (2004) The past and the future of nickel laterites. In: PDAC 2004 International Convention, Trade Show & Investors Exchange. The prospectors and Developers Association of Canada, Toronto, pp 1–27
Queneau P, Boldt JR (1967) The winning of nickel: its geology, mining and extractive metallurgy. Longmans, Canada
Survey USG (2015) Mineral commodity summaries 2015. United States Geological Survey, Reston
Luo W, Feng Q, Ou L, Zhang G, Chen Y (2010) Kinetics of saprolitic laterite leaching by sulphuric acid at atmospheric pressure. Miner Eng 23(6):458–462. https://doi.org/10.1016/j.mineng.2009.10.006
Loveday BK (2008) The use of oxygen in high pressure acid leaching of nickel laterites. Miner Eng 21(7):533–538. https://doi.org/10.1016/j.mineng.2007.11.002
Ma B, Wang C, Yang W, Yin F, Chen Y (2013) Screening and reduction roasting of limonitic laterite and ammonia-carbonate leaching of nickel–cobalt to produce a high-grade iron concentrate. Miner Eng 50–51:106–113. https://doi.org/10.1016/j.mineng.2013.06.014
Zeng X, Zheng H, Gong R, Eheliyagoda D, Zeng X (2018) Uncovering the evolution of substance flow analysis of nickel in China. Resourc Conserv Recycl 135:210–215. https://doi.org/10.1016/j.resconrec.2017.10.014
Chen Y, Mariba ERM, Van Dyk L, Potgieter JH (2011) A review of non-conventional metals extracting technologies from ore and waste. Int J Miner Process 98(1):1–7. https://doi.org/10.1016/j.minpro.2010.10.001
Kempthorne D, Myers D (2007) Mineral commodity summaries 2007: US geological survey. US Government Printing Office, Washington
Whittington BI, Muir D (2000) Pressure acid leaching of nickel laterites: a review. Miner Process Extract Metall Rev 21(6):527–599. https://doi.org/10.1080/08827500008914177
Harris B, Magee J, Valls R (2003) Beyond pressure acid leaching/the Chesbar option. In: Proceedings of 9th ALTA Nickel/Cobalt Forum. Jo-Al Enterprises, Melbourne, 2003. pp 1–15
McDonald R, Whittington B (2008) Atmospheric acid leaching of nickel laterites review. Part II. Chloride and bio-technologies. Hydrometallurgy 91(1–4):56–69. https://doi.org/10.1016/j.hydromet.2007.11.010
Kotzé IJ (2002) Pilot plant production of ferronickel from nickel oxide ores and dusts in a DC arc furnace. Miner Eng 15(11 Supplement 1):1017–1022. https://doi.org/10.1016/S0892-6875(02)00127-9
Nayak J (1985) Production of ferro-nickel from Sukinda laterites in rotary kiln-electric furnace. Trans Indian Inst Metals 38(3):241–247
Ma B, Wang C, Yang W, Chen Y, Yang B (2013) Comprehensive utilization of Philippine laterite ore, part 1: design of technical route and classification of the initial ore based on mineralogical analysis. Int J Miner Process 124:42–49. https://doi.org/10.1016/j.minpro.2013.08.003
Zhou S, Wei Y, Li B, Wang H, Ma B, Wang C, Luo X (2017) Mineralogical characterization and design of a treatment process for Yunnan nickel laterite ore, China. Int J Miner Process 159:51–59. https://doi.org/10.1016/j.minpro.2017.01.002
Watanabe T, Ono S, Arai H, Matsumori T (1987) Direct reduction of garnierite ore for production of ferro-nickel with a rotary kiln at Nippon Yakin Kogyo Co., Ltd., Oheyama Works. Int J Miner Process 19(1):173–187. https://doi.org/10.1016/0301-7516(87)90039-1
Wang XL, Guo JD, Wang YM, Wu XY, Wang BQ (2006) Segregation of lead in Cu–Zn alloy under electric current pulses. Appl Phys Lett 89(6):061910. https://doi.org/10.1063/1.2266034
Liu SJ (1983) Appraisal meeting of new technology for extracting precious metals from copper-nickel alloys. Trans Nonferr Metals Soc China 05(63):1007–7545
Acknowledgements
This work was supported by the National Natural Science Foundation of China (51974025, U1802253, 51674026), the State Key Laboratory of Nickel and Cobalt Resources Comprehensive Utilization Research Project (JKY2019-09), the Fundamental Research Funds for the Central Universities (FRF-TT-19-001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
The contributing editor for this article was D. Panias.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, D., Ma, B., Shi, B. et al. Mineralogical Characterization of Limonitic Laterite from Africa and Its Proposed Processing Route. J. Sustain. Metall. 6, 491–503 (2020). https://doi.org/10.1007/s40831-020-00290-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40831-020-00290-7