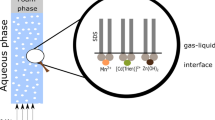
Abstract
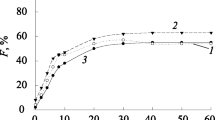
Ion flotation is an efficient separation technique for the recovery of metal ions from dilute aqueous solutions. Provided that both the collected metal and the surfactant are recovered from the foam phase, it could be considered as an alternative to ion exchange, adsorption or solvent extraction in hydrometallurgy. However, most studies are focusing only on the metal recovery by ion flotation and neglect the development of a closed-loop ion flotation flow sheet with regeneration of the collector. As a consequence, the industrial implementation of this technique to hydrometallurgical operations is limited. The objective of this paper was to develop a simple solvometallurgical process for the decomposition of copper(II) tetraamine dodecyl sulfate sublates from foams generated after the ion flotation of dilute copper–zinc ammoniacal solutions with sodium dodecyl sulfate (SDS). It was possible to recover copper almost quantitatively as a copper-bearing precipitate, after mixing the foam phase with a 8 g L−1 NaOH in methanol solution. The collector was regenerated from the solution by removal of methanol by distillation and its reusability was tested in five subsequent ion flotation cycles.The efficiency of the regenerated SDS for the recovery of copper from dilute copper–zinc ammoniacal solutions was similar (ca. 80%) to that of fresh SDS solution without make-up. Based on the experimental results, a conceptual closed-loop flow sheet is proposed for the recovery of copper from dilute ammoniacal leachates by ion flotation.
Graphical Abstract









Similar content being viewed by others
References
Lottermoser B (2010) Mine wastes: characterization, treatment and environmental impacts. Springer, Berlin. https://doi.org/10.1007/978-3-642-12419-8
Kefeni KK, Titus AMM, Mamba BB (2017) Acid mine drainage: prevention, treatment options, and resource recovery: a review. J Clean Prod 151:475–493. https://doi.org/10.1016/j.jclepro.2017.03.082
Kossoff D, Dubbin WE, Alfredsson M, Edwards SJ, Macklin MG, Hudson-Edwards KA (2014) Mine tailings dams: characteristics, failure, environmental impacts, and remediation. Appl Geochem 51:229–245. https://doi.org/10.1016/j.apgeochem.2014.09.010
Jamieson HE, Walker SR, Parsons MB (2015) Mineralogical characterization of mine waste. Appl Geochem 57:85–105. https://doi.org/10.3390/min9050303
Spooren J, Binnemans K, Björkmalm J, Breemersch K, Dams Y, Folens K, González-Moya M, Horckmans L, Komnitsas K, Kurylak W, Lopez M, Mäkinen J, Onisei S, Oorts K, Peys A, Pietek G, Pontikes Y, Snellings R, Tripiana M, Varia J, Willquist K, Yurramendi L, Kinnunen P (2020) Near-zero-waste processing of low-grade, complex primary ores and secondary raw materials in Europe: technology development trends. Resour Conserv Recycl 160(May):104919. https://doi.org/10.1016/j.resconrec.2020.104919
Sebba F (1959) Concentration by ion flotation. Nature 184:1062–1063. https://doi.org/10.1038/1841062a0
Xanthopoulos P, Binnemans K (2021) Removal of cadmium, zinc, and manganese from dilute aqueous solutions by foam separation. J Sustain Metall 7:78–86. https://doi.org/10.1007/s40831-020-00322-2
Chang L, Cao Y, Fan G, Li C, Peng WA (2019) Review of the applications of ion floatation: wastewater treatment, mineral beneficiation and hydrometallurgy. RSC Adv 9(35):20226–20239. https://doi.org/10.1039/c9ra02905b
Somasundaran P (1975) Separation using foaming techniques. Sep Sci 10:93–109. https://doi.org/10.1080/00372367508057071
Pinfold T (1972) Ion flotation. In: Lemlich R (ed) Adsorptive bubble separation techniques. Academic Press, New York, pp 53–73. https://doi.org/10.1016/B978-0-12-443350-2.50009-5
Lemlich R (1972) Ion flotation. Adsorptive bubble separation techniques. Academic Press, New York, pp 53–68. https://doi.org/10.1016/b978-0-12-443350-2.x5001-1
Grieves RB (1975) Foam separations: a review. Chem Eng J 9:93–106. https://doi.org/10.1016/0300-9467(75)80001-3
Lu S, Pugh RJ, Forssberg E (2005) Gas/liquid interfacial separation. Studies in interface science. Elsevier, Amsterdam, pp 559–645. https://doi.org/10.1016/s1383-7303(05)80012-2
Doyle FM (2003) Ion flotation—its potential for hydrometallurgical operations. Int J Miner Process 72(1–4):387–399. https://doi.org/10.1016/S0301-7516(03)00113-3
Nicol S, Galvin K, Engel M (1992) Ion flotation- potential applications to mineral processing. Min Eng 5(10–12):1259–1275. https://doi.org/10.1016/0892-6875(92)90163-4
Crini G, Lichtfouse E (2019) Advantages and disadvantages of techniques used for wastewater treatment. Environ Chem Lett 17:145–155. https://doi.org/10.1007/s10311-018-0785-9
Polat H, Erdogan D (2007) Heavy metal removal from waste waters by ion flotation. J Hazard Mater 148(1–2):267–273. https://doi.org/10.1016/j.jhazmat.2007.02.013
Mcdonald C, Suleiman A (1979) Ion flotation of copper using ethylhexadecyldimethyl-ammonium bromide. Sep Sci Technol 14(3):219–225. https://doi.org/10.1080/01496397908066960
Stoica L, Lacatusu I (2012) Cu(II) separation from diluted aqueous solutions by flotation with atypical collectors anti and syn 2-hydroxy-3,5-di-tert-butyl-benzaldoxime. Int J Environ Waste Manage 9(3–4):293–312. https://doi.org/10.1504/IJEWM.2012.046394
Strel’tsov KA, Abryutin DV (2010) Investigation of regularities of ion flotation of copper with the use of sodium diethyldithiocarbamate. Russ. J. Non-Ferrous Met. 51(2):85–88. https://doi.org/10.3103/S106782121002001X
Hoseinian FS, Rezai B, Kowsari E, Safari M (2018) Kinetic study of Ni(II) removal using ion flotation: effect of chemical interactions. Miner Eng 119(February):212–221. https://doi.org/10.1016/j.mineng.2018.01.028
McDonald CW, Ogunkeye OA (1981) Ion flotation of zinc using ethylhexadecyldimethylammonium bromide. Microchem J 26(1):80–85. https://doi.org/10.1016/0026-265X(81)90013-8
Ulewicz M, Walkowiak W, Jang Y, Kim JS, Bartsch RA (2003) Ion flotation of cadmium (II) and zinc (II) in the presence of proton-ionizable lariat ethers. Anal Chem 75(10):2276–2279. https://doi.org/10.1021/ac026322y
Lusher JA, Sebba F (1965) The separation of aluminium from beryllium by ion flotation of an oxalato-aluminate complex. J Appl Chem 15(12):577–580. https://doi.org/10.1002/jctb.5010151205
Thanh LH, Liu JC (2021) Ion flotation of palladium by using cationic surfactants–effects of chloride ions. Colloids Surf A 616:126326. https://doi.org/10.1016/j.colsurfa.2021.126326
Matis KA, Mavros P (1991) Recovery of metals by ion flotation from dilute aqueous solutions. Sep Purif Rev 20:1–48. https://doi.org/10.1080/03602549108021407
Yenial Ü, Bulut G (2017) Examination of flotation behavior of metal ions for process water remediation. J Mol Liq 241:130–135. https://doi.org/10.1016/j.molliq.2017.06.011
He XC (1991) Ion flotation of rhodium (III) and palladium (II) with anionic surfactants. Talanta 38(3):319–323. https://doi.org/10.1016/0039-9140(91)80054-4
Berg EW, Downey DM (1980) Ion flotation studies of the chlorocomplexes of some platinum group metals. Anal Chim Acta 120:237–248. https://doi.org/10.1016/S0003-2670(01)84367-1
Chirkst DE, Lobacheva OL, Dzhevaga NV (2012) Ion flotation of lanthanum (III) and holmium (III) from nitrate and nitrate-chloride media. Russ J Appl Chem 85(1):25–28. https://doi.org/10.1134/S1070427212010053
Micheau C, Diat O, Bauduin P (2018) Ion foam flotation of neodymium: from speciation to extraction. J Mol Liq 253:217–227. https://doi.org/10.1016/j.molliq.2018.01.022
Eivazihollagh A, Svanedal I, Edlund H, Norgren M (2019) On chelating surfactants: molecular perspectives and application prospects. J Mol Liq 278:688–705. https://doi.org/10.1016/j.molliq.2019.01.076
Liu Z, Doyle FM (2009) Ion flotation of Co2+, Ni2+, and Cu2+ using dodecyldiethylenetriamine (Ddien). Langmuir 25(16):8927–8934. https://doi.org/10.1021/la900098g
Xanthopoulos P, Kalebić D, Kamariah N, Bussé J, Dehaen W, Spooren J, Binnemans K (2021) Recovery of copper from ammoniacal leachates by ion flotation. J Sustain Metall. https://doi.org/10.1007/s40831-021-00363-1
Lazaridis NK, Peleka EN, Karapantsios TD, Matis KA (2004) Copper removal from effluents by various separation techniques. Hydrometallurgy 74(1–2):149–156. https://doi.org/10.1016/j.hydromet.2004.03.003
Jafari M, Abdollahzadeh AA, Aghababaei F (2017) Copper ion recovery from mine water by ion flotation. Mine Water Environ 36:323–327. https://doi.org/10.1007/s10230-016-0408-2
Peng W, Chang L, Li P, Han G, Huang Y, Cao Y (2019) An overview on the surfactants used in ion flotation. J Mol Liq. https://doi.org/10.1039/c9ra02905b
Masuyama A, Okano T, Okahara M, Section E (1990) Application of surface-active amide oximes to the collectors for gallium ion in an ion-flotation system. Ind Eng Chem Res 29:290–294. https://doi.org/10.1021/ie00098a02
Doyle FM, Duyvesteyn S, Sreenivasarao K (1995) The use of ion flotation for detoxification of metal-contaminated waters and process effluents. In: Proceedings of the XIX international mineral processing congress. San Francisco, CA (United States), pp 22–27
Eivazihollagh A, Bäckström J, Norgren M, Edlund H (2018) Electrochemical treatment of copper complexed by chelating agent and chelating surfactant in alkaline solution using a membrane cell. J Chem Technol Biotechnol 93(5):1421–1431. https://doi.org/10.1002/jctb.5510
Kammel R (1984) Metal recovery from dilute aqueous solutions by various electrochemical reactors. In: Bautista RG (eds) Hydrometallurgical process fundamentals. NATO conference series (VI materials science), vol 10, Springer, Boston. https://doi.org/10.1007/978-1-4899-2274-8_24
Binnemans K, Jones PT (2017) Solvometallurgy: an emerging branch of extractive metallurgy. J Sustain Metall 3:570–600. https://doi.org/10.1007/s40831-017-0128-2
Peeters N, Binnemans K, Riaño S (2020) Solvometallurgical recovery of cobalt from lithium-ion battery cathode materials using deep-eutectic solvents. Green Chem 22(13):4210–4221. https://doi.org/10.1039/D0GC00940G
Li Z, Li X, Raiguel S, Binnemans K (2018) Separation of transition metals from rare earths by non-aqueous solvent extraction from ethylene glycol solutions using Aliquat 336. Sep Purif Technol 201:318–326. https://doi.org/10.1016/j.seppur.2018.03.022
Li X, Monnens W, Li Z, Fransaer J, Binnemans K (2020) Solvometallurgical process for extraction of copper from chalcopyrite and other sulfidic ore minerals. Green Chem 22(2):417–426. https://doi.org/10.1039/C9GC02983D
Bi PY, Dong HR, Dong J (2010) The recent progress of solvent sublation. J Chrom A 1217(16):2716–2725. https://doi.org/10.1016/j.chroma.2009.11.020
Tünay O, Kabdaşli NI (1994) Hydroxide precipitation of complexed metals. Water Res 28(10):2117–2124. https://doi.org/10.1016/0043-1354(94)90022-1
Dean JA (1999) Lange’s handbook of chemistry, 15th edn. McGraw-Hill, New York
Bevandić S, Blannin R, Vander Auwera J, Delmelle N, Caterina D, Nguyen F, Muchez P (2021) Geochemical and mineralogical characterisation of historic Zn–Pb mine waste, plombières. East Belg Miner 11(1):28. https://doi.org/10.3390/min11010028
Prasad MS, Kenyen VP, Assar DN (1992) Development of SX-EW process for copper recovery—an overview. Miner Process Extr Metall Rev 8(1–4):95–118. https://doi.org/10.1080/08827509208952680
Niraula TP, Chatterjee SK, Bhattarai A (2018) Micellization of sodium dodecyl sulphate in presence and absence of alkali metal halides at different temperatures in water and methanol-water mixtures. J Mol Liq 250:287–294. https://doi.org/10.1016/j.molliq.2017.12.014
Viana RB, da Silva AB, Pimentel AS (2012) Infrared spectroscopy of anionic, cationic, and zwitterionic surfactants. Adv Phys Chem. https://doi.org/10.1155/2012/903272
Zhao P, Lu L, Liu X, De la Torre AG, Cheng X (2018) Error analysis and correction for quantitative phase analysis based on rietveld-internal standard method: whether the minor phases can be ignored? Curr Comput-Aided Drug Des 8(3):110. https://doi.org/10.3390/cryst8030110
Acknowledgements
The research leading to these results has received funding from the European Community’s Horizon 2020 Programme under Grant Agreement No. 812580 SULTAN) (MSCA- ETN SULTAN). This publication reflects only the authors’ view, exempting the Community from any liability. The authors thank Bart van Huffel (KU Leuven) for helping with the LC/MS analysis. Thomas Abo Atia (KU Leuven), Srećko Bevandić (KU Leuven) and Demian Kalebić (KU Leuven) are gratefully acknowledged for the fruitful scientific discussions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
The contributing editor for this article was Yongxiang Yang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xanthopoulos, P., Binnemans, K. Closing the Loop in Ion Flotation: Recovery of Copper, Regeneration and Reuse of Collector from the Foam Phase by a Solvometallurgical Process. J. Sustain. Metall. 7, 1565–1574 (2021). https://doi.org/10.1007/s40831-021-00463-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40831-021-00463-y