Abstract
Purpose
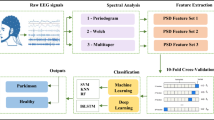
Diagnosis of Parkinson’s disease (PD) is generally based on family medical history, physical examination, and response to medication. Objective tools using machine learning algorithms have been developed to aid in PD diagnosis; however, feature extraction is time consuming, computationally intensive, and difficult to implement in clinical settings. This study compared the performance of two methods, namely a support vector machine (SVM) and convolutional neural network (CNN), in the classification of patients with PD based on resting-state electroencephalography (EEG).
Methods
In total, 39 patients with PD and 40 healthy controls participated in the experiment. Mean frequency, relative power, coherence, sample entropy, and multiscale entropy were calculated as features.
Results
The accuracies of the SVM using 548 selected features and that of the CNN using 2992 extracted features were 88.88% and 98.66%, respectively. The accuracy of the CNN using raw data was 97.54%. Furthermore, the CNN model using features required 1272 s for training and 0.07 s for testing, whereas the CNN model using raw data required 994 s for training and 0.32 s for testing.
Conclusion
Our results imply that a CNN model taking raw data as inputs can automatically select the salient features, thereby reducing the required training time and achieving high classification performance.



Similar content being viewed by others
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
References
Amin, S. U., Alsulaiman, M., Muhammad, G., Bencherif, M. A., & Hossain, M. S. (2019). Multilevel weighted feature fusion using convolutional neural networks for EEG motor imagery classification. in IEEE Access, 7, 18940–18950. https://doi.org/10.1109/ACCESS.2019.2895688
Belardinelli, P., Azodi-Avval, R., Ortiz, E., Naros, G., Grimm, F., Weiss, D., & Gharabaghi, A. (2019). Intraoperative localization of spatially and spectrally distinct resting-state networks in Parkinson’s disease. Journal of Neurosurgery, 132(4), 1234–1242. https://doi.org/10.3171/2018.11.JNS181684
Betrouni, N., Delval, A., Chaton, L., Defebvre, L., Duits, A., Moonen, A. … Dujardin, K. (2019). Electroencephalography-based machine learning for cognitive profiling in Parkinson’s disease: Preliminary results. Movement Disorders, 34(2), 210–217. https://doi.org/10.1002/mds.27528
Blesa, J., Trigo-Damas, I., Dileone, M., Del Rey, N. L., Hernandez, L. F., & Obeso, J. A. (2017). Compensatory mechanisms in Parkinson’s disease: Circuits adaptations and role in disease modification. Experimental Neurology, 298(Pt B), 148–161. https://doi.org/10.1016/j.expneurol.2017.10.002
Boonstra, T. W., Nikolin, S., Meisener, A. C., Martin, D. M., & Loo, C. K. (2016). Change in mean frequency of resting-state electroencephalography after transcranial direct current stimulation. Frontiers in Human Neuroscience, 10, 270. https://doi.org/10.3389/fnhum.2016.00270
Chaturvedi, M., Hatz, F., Gschwandtner, U., Bogaarts, J. G., Meyer, A., Fuhr, P., & Roth, V. (2017). Quantitative EEG (QEEG) measures differentiate Parkinson’s disease (PD) patients from healthy controls (HC). Frontiers in Aging Neuroscience 9:3, 2017. https://doi.org/10.3389/fnagi.2017.00003
Chaudhuri, K. R., & Schapira, A. H. (2009). Non-motor symptoms of Parkinson’s disease: dopaminergic pathophysiology and treatment. The Lancet Neurology, 8(5), 464–474. https://doi.org/10.1016/S1474-4422(09)70068-7
Chen, J. X., Zhang, P. W., Mao, Z. J., Huang, Y. F., Jiang, D. M., & Zhang, Y. N. (2019). Accurate EEG-based emotion recognition on combined features using deep convolutional neural networks. in IEEE Access, 7, 44317–44328. https://doi.org/10.1109/ACCESS.2019.2908285
Cho, K. O., & Jang, H. J. (2020). Comparison of different input modalities and network structures for deep learning-based seizure detection. Scientific Reports, 10, 122. https://doi.org/10.1038/s41598-019-56958-y
Christensen, J. A., Zoetmulder, M., Koch, H., Frandsen, R., Arvastson, L., Christensen, S. R. … Sorensen, H. B. (2014). Data-driven modeling of sleep EEG and EOG reveals characteristics indicative of pre-Parkinson’s and Parkinson’s disease. Journal of Neuroscience Methods, 235, 262–276. https://doi.org/10.1016/j.jneumeth.2014.07.014
Fatima, M., & Pasha, M. (2017). Survey of machine learning algorithms for disease diagnostic. Journal of Intelligent Learning Systems and Applications, 9, 1–16. https://doi.org/10.4236/jilsa.2017.91001
Folstein, M. F., Folstein, S. E., & McHugh, P. R. (1975). Mini-mental state: A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research, 12(3), 189–198. doi: https://doi.org/10.1016/0022-3956(75)90026-6
Gongora, M., Velasques, B., Cagy, M., Teixeira, S., & Ribeiro, P. (2019). EEG coherence as a diagnostic tool to measure the initial stages of Parkinson Disease. Medical Hypotheses, 123, 74–78. https://doi.org/10.1016/j.mehy.2018.12.014
Grinberg, L. T., Rueb, U., Alho, A. T., & Heinsen, H. (2010). Brainstem pathology and non-motor symptoms in Parkinson’s disease. Journal of the Neurological Sciences, 289(1), 81–88. https://doi.org/10.1016/j.jns.2009.08.021
Han, C. X., Wang, J., Yi, G., & Che, Y. Q. (2013). Investigation of EEG abnormalities in the early stage of Parkinson’s disease. Cognitive Neurodynamics, 7, 351–359. https://doi.org/10.1007/s11571-013-9247-z
Heida, T., Poppe, N. R., de Vos, C. C., van Putten, M. J., & van Vugt, J. P. (2014). Event-related mu-rhythm desynchronization during movement observation is impaired in Parkinson’s disease. Clinical Neurophysiology, 125(9), 1819–1825. https://doi.org/10.1016/j.clinph.2014.01.016
Jaramillo-Jimenez, A., Suarez-Revelo, J. X., Ochoa-Gomez, J. F., Carmona Arroyave, J. A., Bocanegra, Y., Lopera, F. … Aarsland, D. (2021). Resting-state EEG alpha/theta ratio related to neuropsychological test performance in Parkinson’s Disease. Clinical Neurophysiology, 132(3), 756–764. https://doi.org/10.1016/j.clinph.2021.01.001
Liu, Q., Cai, J. F., Fan, S. Z., Abbod, M., Shieh, J. S., Kung, Y. C., & Lin, L. (2019). Spectrum analysis of EEG signals using CNN to model patient’s consciousness level based on anesthesiologists’ experience. in IEEE Access, 7, 53731–53742. https://doi.org/10.1109/ACCESS.2019.2912273
Maitín, A. M., García-Tejedor, A. J., & Muñoz, J. P. R. (2020). Machine learning approaches for detecting Parkinson’s disease from EEG analysis: A systematic review. Applied Sciences, 10(23), 8662. https://doi.org/10.3390/app10238662
Marsden, C. D., & Obeso, J. A. (1994). The functions of the basal ganglia and the paradox of stereotaxic surgery in Parkinson’s disease. Brain, 117(4), 877–897. https://doi.org/10.1093/brain/117.4.877
Moazami-Goudarzi, M., Sarnthein, J., Michels, L., Moukhtieva, R., & Jeanmonod, D. (2008). Enhanced frontal low and high frequency power and synchronization in the resting EEG of parkinsonian patients. Neuroimage, 41(3), 985–997. https://doi.org/10.1016/j.neuroimage.2008.03.032
Olde Dubbelink, K. T., Stoffers, D., Deijen, J. B., Twisk, J. W., Stam, C. J., & Berendse, H. W. (2013). Cognitive decline in Parkinson’s disease is associated with slowing of resting-state brain activity: a longitudinal study. Neurobiology of Aging, 34(2), 408–418. https://doi.org/10.1016/j.neurobiolaging.2012.02.029
Pezard, L., Jech, R., & Ruzicka, E. (2001). Investigation of non-linear properties of multichannel EEG in the early stages of Parkinson’s disease. Clinical Neurophysiology, 112(1), 38–45. https://doi.org/10.1016/s1388-2457(00)00512-5
Sajda, P. (2006). Machine learning for detection and diagnosis of disease. Annual Review of Biomedical Engineering, 8, 537–565. https://doi.org/10.1146/annurev.bioeng.8.061505.095802
Sarnthein, J., & Jeanmonod, D. (2007). High thalamocortical theta coherence in patients with Parkinson’s disease. Journal of Neuroscience, 27(1), 124–131. https://doi.org/10.1523/JNEUROSCI.2411-06.2007
Siuly, S., Alcin, O. F., Kabir, E., Sengur, A., Wang, H., Zhang, Y., & Whittaker, F. (2020). A new framework for automatic detection of patients with mild cognitive impairment using resting-state EEG signals. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 28(9), 1966–1976. https://doi.org/10.1109/TNSRE.2020.3013429
Tang, Z., Li, C., & Sun, S. (2017). Single-trial EEG classification of motor imagery using deep convolutional neural networks. Optik - International Journal for Light and Electron Optics, 130, 11–18. https://doi.org/10.1016/j.ijleo.2016.10.117
Vabalas, A., Gowen, E., Poliakoff, E., & Casson, A. J. (2019). Machine learning algorithm validation with a limited sample size. PLoS ONE, 14(11), e0224365. https://doi.org/10.1371/journal.pone.0224365
Yi, G. S., Wang, J., Deng, B., & Wei, X. L. (2017). Complexity of resting-state EEG activity in the patients with early-stage Parkinson’s disease. Cognitive Neurodynamics, 11(2), 147–160. https://doi.org/10.1007/s11571-016-9415-z
Yuvaraj, R., Murugappan, M., Mohamed Ibrahim, N., Iqbal, M., Sundaraj, K., Mohamad, K. … Satiyan, M. (2014). On the analysis of EEG power, frequency and asymmetry in Parkinson’s disease during emotion processing. Behavioral and Brain Functions, 10(1), 12. https://doi.org/10.1186/1744-9081-10-12
Acknowledgements
This study was supported in part by research grants from Ministry of Science and Technology (MOST 109 2221-E-130-003), Taiwan.
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics Approval and Consent to Participate
The experimental protocol was established, according to the ethical guidelines of the Helsinki Declaration and was approved by the Human Ethics Committee of Chang Gung Memorial Hospital (IRB number: 107-0857 C). Written informed consent was obtained from individual or guardian participants.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, CY., Huang, YZ. Parkinson’s Disease Classification Using Machine Learning Approaches and Resting-State EEG. J. Med. Biol. Eng. 42, 263–270 (2022). https://doi.org/10.1007/s40846-022-00695-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-022-00695-7