Abstract
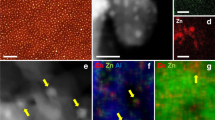
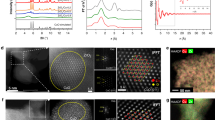
In situ quick X-ray absorption spectroscopy (QXAFS) at the Cu and Zn K-edge under operando conditions has been used to unravel the Cu/Zn interaction and identify possible active site of CuO/ZnO/Al2O3 catalyst for methanol synthesis. In this work, the catalyst, whose activity increases with the reaction temperature and pressure, was studied at calcined, reduced, and reacted conditions. TEM and EDX images for the calcined and reduced catalysts showed that copper was distributed uniformly at both conditions. TPR profile revealed two reduction peaks at 165 and 195 °C for copper species in the calcined catalyst. QXAFS results demonstrated that the calcined form consisted mainly of a mixed CuO and ZnO, and it was progressively transformed into Cu metal particles and dispersed ZnO species as the reduction treatment. It was demonstrated that activation of the catalyst precursor occurred via a Cu+ intermediate, and the active catalyst predominantly consisted of metallic Cu and ZnO even under higher pressures. Structure of the active catalyst did not change with the temperature or pressure, indicating that the role of the Zn was mainly to improve Cu dispersion. This indicates the potential of QXAFS method in studying the structure evolutions of catalysts in methanol synthesis.







Similar content being viewed by others
References
Mauna Loa Observatory, Hawaii. http://co2now.org/(2014)
K.M.K. Yu, I. Curcic, J. Gabriel et al., Recent advances in CO2 capture and utilization. Chem. Sus. Chem. 1, 893–899 (2008). doi:10.1002/cssc.201090023
W. Wang, S.P. Wang, X.B. Ma et al., Recent advances in catalytic hydrogenation of carbon dioxide. Chem. Soc. Rev. 40, 3703–3727 (2011). doi:10.1039/clcs15008a
G. Centi, E.A. Quadrelli, S. Perathoner, Catalysis for CO2 conversion: a key technology for rapid introduction of renewable energy in the value chain of chemical industries. Energy Environ. Sci. 6, 1711–1731 (2013). doi:10.1039/C3EE00056G
G.A. Olah, G.K.S. Prakash, A. Goeppert, Anthropogenic chemical carbon cycle for a sustainable future. J. Am. Chem. Soc. 133, 12881–12898 (2011). doi:10.1021/ja202642y
G.A. Olah, Jenseits von öl und Gas: die Methanolwirtschaft. Angew. Chem. 117, 2692–2696 (2005). doi:10.1002/ange.200462121
G.A. Olah, Beyond oil and gas: the methanol economy. Angew. Chem. Int. Ed. 44, 2636–2639 (2005). doi:10.1002/anie.200462121
P. Gao, F. Li, F.K. Xiao et al., Effect of hydrotalcite-containing precursors on performance of Cu/Zn/Al/Zr catalysts for CO2 hydrogenation: introduction of Cu2+ at different formation stages of precursors. Catal. Today 194, 9–15 (2012). doi:10.1016/j.cattod.2012.06.012
U. Olsbye, S. Svelle, M. BjØgen et al., Conversion of methanol to hydrocarbons: how zeolite cavity and pore size controls product selectivity. Angew. Chem. 51, 5810–5831 (2012). doi:10.1002/aine.201103657
E.E. Ortelli, J. Wambach, A. Wokaun, Methanol synthesis reactions over CuZr based catalyst investigated using periodic variations of reaction concentrations. Appl. Catal. A 216, 227–241 (2001). doi:10.1016/S0926-860X(01)00569-5
A.K. Shukla, P.A. Christensen, A.J. Dickinson et al., A liquid-feed solid polymer electrolyte direct methanol fuel cell operating at near-ambient conditions. J. Power Sources 76, 54–59 (1998). doi:10.1016/S0378-7753(98)00140-2
G.C. Chinchen, K.C. Waugh, D.A. Whan, The activity and state of the copper surface in methanol synthesis catalysts. Appl. Catal. 25, 101–107 (1986). doi:10.1016/S0166-9834(00)81226-9
F. Liao, Y.Q. Huang, J.W. Ge et al., Morphology-dependent interactions of ZnO with Cu nanoparticles at the materials’ interface in selective hydrogenation of CO2 to CH3OH. Angew. Chem. 123, 2210–2213 (2011). doi:10.1002/anie.201007108
P. Gao, F. Li, H.J. Zhan et al., Influence of Zr on the performance of Cu/Zn/Al/Zr catalysts via hydrotalcite-like precursors for CO2 hydrogenation to methanol. J. Catal. 298, 51–60 (2013). doi:10.1016/j.jcat.2012.10.030
M. Behrens, F. Studt, I. Kasatkin et al., The active site of methanol synthesis over Cu/ZnO/Al2O3 industrial catalysts. Science 336, 893–897 (2012). doi:10.1126/science.1219831
Y.Q. Cen, X.N. Li, H.Z. Liu, Preparation of copper-based catalysts for methanol synthesis by acid-alkali-based alternate precipitation method. Chin. J. Catal. 27, 210–216 (2006). doi:10.1016/S1872-2067(06)60014-4
J.J. Wu, X.D. Zhou, Catalytic conversion of CO2 to value added fuels: current status, challenges, and future directions. Chin. J. Catal. 37, 999–1015 (2016). doi:10.1016/S1872-2067(16)62455-5
J. Nakamura, Y. Choi, T. Fujitani, On the issue of the active site and the role of ZnO in Cu/ZnO methanol synthesis catalysts. Top. Catal. 22, 277–285 (2003). doi:10.1023/A:1023588322846
J.D. Grunwaldt, A.M. Molenbroek, N.Y. Topsøe, H. Topsøe, In situ investigations of structural changes in Cu/ZnO catalysts. J. Catal. 194(45), 452–460 (2000). doi:10.1006/jcat.2000.2930
T. Fujitani, J. Nakamura, The effect of ZnO in methanol synthesis catalysts on Cu dispersion and the specific activity. Catal. Lett. 56, 119–124 (1998). doi:10.1023/A:1019000927366
L. Dong, X.J. Yao, Y. Chen, Interactions among supported copper based catalyst components and their effects on performance: a review. Chin. J. Catal. 34, 851–864 (2013). doi:10.1016/S1872-2067(12)60592-0
B. Denise, R.P.A. Sneeden, B. Beguin et al., Supported copper catalysts in the synthesis of methanol: N2O-titrations. Appl. Catal. 30, 353–358 (1987). doi:10.1016/S0166-9834(00)84125-1
T.H. Fleisch, R.L. Miecille, Studies on the chemical state of Cu during methanol synthesis. J. Catal. 90, 165–171 (1984). doi:10.1016/0021-9517(84)90099-X
W.X. Pan, R. Cao, D.L. Roberts et al., Methanol synthesis activity of CuZnO catalysts. J. Catal. 114, 440–444 (1988). doi:10.1016/0021-9517(88)90047-4
J.F. Deng, Q. Sun, Y.L. Zhang et al., A novel process for preparation of a Cu/ZnO/Al2O3 ultrafine catalyst for methanol synthesis from CO2 + H2: comparison of various preparation methods. Appl. Catal. A 139, 75–80 (1996). doi:10.1016/0926-860X(95)00324-X
G.C. Chinchen, K.C. Waugh, D.A. Whan, The activity and state of the copper surface in methanol synthesis catalysts. Appl. Catal. 25, 48–51 (1986). doi:10.1016/S0166-9834(00)81226-9
G.C. Chinchen, M.C. Spencer, K.C. Waugh et al., Promotion of methanol synthesis and the water-gas shift reactions by adsorbed oxygen on supported copper catalysts. J. Chem Soc Farady Trans. I 83, 2193–2200 (1987). doi:10.1039/F19878302193
R.G. Herman, K. Klier, G.W. Simmons et al., Catalytic synthesis of methanol from COH2: I. Phase composition, electronic properties, and activities of the Cu/ZnO/M2O3 catalysts. J. Catal. 56, 407–410 (1979). doi:10.1016/0021-9517(79)90132-5
H.Y. Chen, S.P. Lau, L. Chen et al., Synergism between Cu and Zn sites in Cu/Zn catalysts for methanol synthesis. Appl. Surf. Sci. 152, 193–198 (1999). doi:10.1016/S0169-4332(99)00317-7
Y. Kanai, T. Watanable, T. Fujtitani et al., The synergy between Cu and ZnO in methanol synthesis catalysts. Catal. Lett. 38, 157–163 (1996). doi:10.1007/BF00806562
T. Fujitani, J. Nakamura, The chemical modification seen in the Cu/ZnO methanol synthesis catalysts. Appl. Catal. A 191, 111–129 (2000). doi:10.1016/S0926-860X(99)00313-0
D.E. Sayers, E.A. Stern, F.W. Lytle, New technique for investigating noncrystalline structures: Fourier analysis of the extended X-ray absorption fine structure. Phys. Rev. Lett. 27, 1204–1207 (1971). doi:10.1103/PhysRevLett.27.1204
J. Singh, C. Lamberti, J.A. Bokhoven, Advanced X-ray absorption and emission spectroscopy: in situ catalytic studies. Chem. Soc. Rev. 39, 4754–4766 (2010). doi:10.1039/c0cs00054j
H.L. Bao, X.P. Sun, J. Zheng et al., Structural changes of Rh-Mn nanoparticles inside carbon nanotubes studied by X-ray absorption spectroscopy. Chin. J. Catal. 35, 1418–1427 (2014). doi:10.1016/S1872-2067(14)60081-4
A.E. Russell, A. Rose, X-ray absorption spectroscopy of low temperature fuel cell catalysts. Chem. Rev. 104, 4613–4635 (2004). doi:10.1002/chin.200450216
H.S. Yu, X.J. Wei, J. Li et al., The XAFS beamline of SSRF. Nucl. Sci. Tech. 26, 050102 (2015). doi:10.13538/j.1001-8042/nst.26.050102
K. Evgeny, S. Jacinto, A.D. Jehad et al., Structure of the methanol synthesis catalyst determined by in situ HERFD XAS and EXAFS. Catal. Sci. Technol. 2, 373–378 (2012). doi:10.1039/c1cy00277e
S. Velu, K. Suzuki, C.S. Gopinath et al., XPS, XANES and EXAFS investigations of CuO/ZnO/Al2O3/ZrO2 mixed oxide catalysts. Phys. Chem. Chem. Phys. 4, 1990–1999 (2002). doi:10.1039/b109766k
D. Grandjean, V. Pelipenko, E.D. Batyrev et al., Dynamic Cu/Zn interaction in SiO2 supported methanol synthesis catalysts unraveled by in- situ XAFS. J. Phys. Chem. C 115, 20175–20191 (2011). doi:10.1021/jp201839s
J. Fang, F.C. Shi, H.Z. Bao et al., Evolution of surface and bulk structures of Ce x Ti1−x O2 oxide composites. Chin. J. Catal. 34, 2075–2083 (2013). doi:10.1016/S1872-2067(12)60667-6
K. Okumura, T. Honma, S. Hirayama et al., Stepwise growth of Pd clusters in USY zeolite at room temperature analyzed by QXAFS. J. Phys. Chem. C 112, 16740–16747 (2008). doi:10.1021/jp804381c
Q. Sun, Y.L. Zhang, H.Y. Chen et al., A novel process for the preparation of Cu/ZnO and Cu/ZnO/Al2O3 ultrafine catalyst: structure, surface properties, and activity for methanol synthesis from CO2 + H2. Chen. J. Catal. 167, 92–105 (1997). doi:10.1006/jcat.1997.1554
J. Yoshihara, C.T. Campbell, Methanol synthesis and reverse water–gas shift kinetics over Cu(110) model catalysts: structural sensitivity. J. Catal. 161, 776–782 (1996). doi:10.1006/jcat.1996.0240
X.S. Dong, F. Li, N. Zhao et al., CO2 hydrogenation to methanol over Cu/ZnO/ZrO2 catalysts prepared by precipitation-reduction method. Appl. Catal. B 191, 8–17 (2016). doi:10.1016/j.apcatb.2016.03.014
Z. Li, H.Y. Zheng, K.C. Xie, Surface properties of CuO/ZnO/Al2O3 catalyst for methanol synthesis in slurry reactor. Chin. J. Catal. 29, 431–435 (2008). doi:10.3321/j.issn:0253-9837.2008.05.005
X.P. Sun, F.F. Sun, Z.H. Sun et al., Disorder effects on EXAFS modeling for catalysts working at elevated temperatures. Radiat. Phys. Chem. (2016). doi:10.1016/j.radphyschem.2016.01.039
A. Yevick, A.I. Frenkel, Effect of surface disorder on EXAFS modeling of metallic clusters. Phys. Rev. B 81, 760–762 (2010). doi:10.1103/PhysRevB.81.115451
E.B. Batyrev, J.C.V.D. Heuvel, J. Beckers et al., The effect of the reduction temperature on the structure of Cu/ZnO/SiO2 catalysts for methanol synthesis. J. Catal. 229, 136–143 (2005). doi:10.1016/j.jcat.2004.10.012
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the National Basic Research Program of China (973 Program, 2013CB933104) and the National Natural Science Foundation of China (Nos. 11275258 and 11135008).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, XP., Sun, FF., Gu, SQ. et al. Local structural evolutions of CuO/ZnO/Al2O3 catalyst for methanol synthesis under operando conditions studied by in situ quick X-ray absorption spectroscopy. NUCL SCI TECH 28, 21 (2017). https://doi.org/10.1007/s41365-016-0170-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41365-016-0170-y