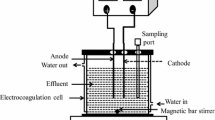
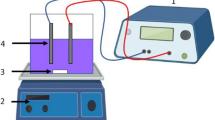
Abstract
Batik wastewater contains organic dyes, waxes, silicate, etc. that are environmentally problematic. Two plain waste aluminum cans were used as electrodes in a batch electrocoagulation (EC) process to remove color, chemical oxygen demand (COD), and silica from batik wastewater. Response surface methodology based on the central composite design was used to demonstrate the dependence of removal efficiency on four key process variables and possible interactions between them. The models were statistically evaluated and conditions for optimized color, COD, and silica removals were verified experimentally. Relatively stronger effects of current density were detected while interactions could be observed on the effects of pH with current density or process time. The floatation of silica scum resulted in increased removal and deviation from the model. The optimal operating conditions were achieved at a current density of 29.91 mA/cm2, an initial pH of 4.51, a reaction time of 24.93 min, and a NaCl dosage of 1.03 g/L that resulted in 88.13 ± 0.51%, 70.50 ± 3.17%, and 100.00 ± 0.00% removals of color, COD, and silica, respectively. The color, COD and pH of the batik wastewater were also reduced to 33 ADMI, 100 mg/L and 6.62 to meet the respective discharge standard limits. Silica content toughened the flocs and increased their buoyancy by strongly interacting with gas bubbles to float to the surface. EC was demonstrated to be an effective and cost-effective treatment technology for the treatment of batik wastewater.
Research Highlights
-
Electrocoagulation treatment of batik wastewater.
-
Color, COD and silica removals.
-
Response surface methodology based on central composite design.
-
Removal efficiency and interaction between process variables.
-
pH as the most influential parameter to cause interactions.
-
Role of silica in the removal mechanism.








Similar content being viewed by others
References
Abbasi S, Mirghorayshi M, Zinadini S, Zinatizadeh AA (2020) A novel single continuous electrocoagulation process for treatment of licorice processing wastewater : optimization of operating factors using RSM. Process Saf Environ Prot 134:323–332. https://doi.org/10.1016/j.psep.2019.12.005
Abdullah AZ, Razali N, Lee KT (2009) Optimization of K/SBA-15 catalyzed transesterification of palm oil using response surface methodology. Fuel Process Technol 90:958–964. https://doi.org/10.1016/j.fuproc.2009.03.023
Amani-Ghadim AR, Olad A, Aber S, Ashassi-Sorkhabi H (2012) Comparison of organic dyes removal mechanism in electrocoagulation process using iron and aluminum anodes. Environ Prog Sustain Energy 00:1–10. https://doi.org/10.1002/ep.11656
Amri N, Abdullah AZ, Ismail S (2020) Removal efficiency of acid red 18 dye from aqueous solution using different aluminium-based electrode materials by electrocoagulation process. Indones J Chem 20:536–544. https://doi.org/10.22146/ijc.43206
APHA, Awwa, WEF (2017) Standard methods for the examination of water and wastewater, 23rd edn. American Public Health Association, Washington D.C.
Arnita Y, Elystia S, Andesgur I (2017) Penyisihan kadar COD dan TSS pada limbah cair pewarnaan batik mengunakan metode elektrokoagulasi. Jom FTEKNIK 4:1–9
Barrera-Díaz C, Varela-Guerrero V, Cuevas-Yáñez E, Martínez-Barrera G, Roa-Morales G, García-Morales MA (2014) Use of recycled aluminum-polyethylene composite films as anodic electrodes for electrocoagulation of wastewater. Int J Electrochem Sci 9:1034–1043
Bener S, Bulca Ö, Palas B, Tekin G, Atalay SA, Ersöz G (2019) Electrocoagulation process for the treatment of real textile wastewater: effect of operative conditions on the organic carbon removal and kinetic study. Process Saf Environ Prot 129:47–54. https://doi.org/10.1016/j.psep.2019.06.010
Buthiyappan A, Raman AAA, Daud WMAW (2016) Development of an advanced chemical oxidation wastewater treatment system for the batik industry. RSC Adv 6:25222–25241. https://doi.org/10.1039/C5RA26775G
Camcioglu S, Ozyurt B, Hapoglu H (2017) Effect of process control on optimization of pulp and paper mill wastewater treatment by electrocoagulation. Process Saf Environ Prot 111:300–319. https://doi.org/10.1016/j.psep.2017.07.014
Castañeda LF, Coreño O, Nava JL (2019) Arsenic and hydrated silica removal from groundwater by electrocoagulation using an up-flow reactor in a serpentine array. J Environ Chem Eng 7:1–7. https://doi.org/10.1016/j.jece.2019.103353
Chantes P, Jarusutthirak C, Kanchanapiya P, Danwittayakul S (2015) Treatment of textile dyeing wastewater by electrocoagulation. Key Eng Mater 659:284–288. https://doi.org/10.4028/www.scientific.net/KEM.659.284
Duman O, Tunç S (2009) Electrokinetic and rheological properties of Na-bentonite in some electrolyte solutions. Micropor Mesopor Mater 117:331–338. https://doi.org/10.1016/j.micromeso.2008.07.007
Fajriati I, Mudasir M, Wahyuni ET (2019) Adsorption and photodegradation of cationic and anionic dyes by TiO2-chitosan nanocomposite. Indones J Chem 19:441–453. https://doi.org/10.22146/ijc.38278
Ghalwa NMA, Saqer AM, Farhat NB (2016) Removal of reactive red 24 dye by clean electrocoagulation process using iron and aluminum electrodes. J Chem Eng Process Technol 7:1–7. https://doi.org/10.4172/2157-7048.1000269
Guzmán A, Nava JL, Coreño O, Rodríguez I, Gutiérrez S (2016) Arsenic and fluoride removal from groundwater by electrocoagulation using a continuous filter-press reactor. Chemosphere 144:2113–2120. https://doi.org/10.1016/j.chemosphere.2015.10.108
Hach (2014a) Chemical oxygen demand, USEPA and reactor digestion method 10, pp 1–10. DOC316.53.01099
Hach (2014b) Method 8185 powder pillows-silicomolybdate method 1, pp 1–6. DOC316.53.01133
Hach (2019) Color, ADMI weighted ordinate method 7, pp 1–6. DOC316.53.01122
Huda N, Raman AAA, Bello MM, Ramesh S (2017) Electrocoagulation treatment of raw landfill leachate using iron-based electrodes: effects of process parameters and optimization. J Environ Manag 204:75–81. https://doi.org/10.1016/j.jenvman.2017.08.028
Environmental Quality Act 1974 (Act 127), Regulations, Rules and Orders (Issue 20th August) (2020) International Law Book Services (ILBS)
Kang J, Fan R, Hu Y, Sun W, Liu R, Zhang Q, Liu H, Meng X (2018) Silicate removal from recycled wastewater for the improvement of scheelite flotation performance. J Clean Prod 195:280–288. https://doi.org/10.1016/j.jclepro.2018.05.215
Khandegar V, Saroha AK (2013) Electrocoagulation for the treatment of textile industry effluent—a review. J Environ Manag 128:949–963. https://doi.org/10.1016/j.jenvman.2013.06.043
Khorram AG, Fallah N (2018) Treatment of textile dyeing factory wastewater by electrocoagulation with low sludge settling time: optimization of operating parameters by RSM. J Environ Chem Eng 6:635–642. https://doi.org/10.1016/j.jece.2017.12.054
Loong NC, Basri M, Fang LF, Masoumi HRF, Tripathy M, Karjiban RA, Abdul-Malek E (2014) Comparison of Box–Behnken and central composite designs in optimization of fullerene loaded palm-based nano-emulsions for cosmeceutical application. Ind Crops Prod 59:309–317. https://doi.org/10.1016/j.indcrop.2014.05.042
Marriaga-Cabrales N, Machuca-Martínez F (2014) Fundamentals of electrocoagulation. Evaluation of electrochemical reactors as a new way to environmental protection. Research Signpost, Kerala, pp 1–16
Mondal B, Srivastava VC, Mall ID (2012) Electrochemical treatment of dye-bath effluent by stainless steel electrodes: multiple response optimization and residue analysis. J Environ Sci Heal Part A Toxic Hazard Subst Environ Eng 47:2040–2051. https://doi.org/10.1080/10934529.2012.695675
Moussa DT, El-Naas MH, Nasser M, Al-Marri MJ (2017) A comprehensive review of electrocoagulation for water treatment: potentials and challenges. J Enviro Manag 186:24–41. https://doi.org/10.1016/j.jenvman.2016.10.032
Nasrullah M, Zularisam AW, Krishnan S, Sakinah M, Singh L, Fen YW (2019) High performance electrocoagulation process in treating palm oil mill effluent using high current intensity application. Chin J Chem Eng 27:208–217. https://doi.org/10.1016/j.cjche.2018.07.021
Riyanto, Puspitasari E (2018) Treatment of wastewater batik by electrochemical coagulation using aluminium (Al) electrodes. IOP Conf Ser Mater Sci Eng 299:1–8. https://doi.org/10.1088/1757-899X/299/1/012081
Rosales M, Coreno O, Nava JL (2018) Removal of hydrated silica, fluoride and arsenic from groundwater by electrocoagulation using a continuous reactor with a twelve-cell stack. Chemosphere 211:149–155. https://doi.org/10.1016/j.chemosphere.2018.07.113
Vakili M, Rafatullah M, Salamatinia B, Ibrahim MH, Abdullah AZ, Gholami Z, Tan KB, Amouzgar P (2015) Elimination of reactive blue 4 from aqueous solutions using 3-aminopropyl triethoxysilane modified chitosan beads. Carbohydr Polym 132:89–96. https://doi.org/10.1016/j.carbpol.2015.05.080
Vepsäläinen M, Sillanpää M (2020) Electrocoagulation in the treatment of industrial waters and wastewaters. Advanced water treatment. Elsevier Inc., pp 1–78. https://doi.org/10.1016/B978-0-12-819227-6.00001-2
Wang CT, Chou WL, Chen LS, Chang SY (2009) Silica particles settling characteristics and removal performances of oxide chemical mechanical polishing wastewater treated by electrocoagulation technology. J Hazard Mater 161:344–350. https://doi.org/10.1016/j.jhazmat.2008.03.099
Acknowledgements
The authors gratefully acknowledge the financial support received from the Ministry of Higher Education (MOE) of Malaysia (LRGS Grant, Project number 67215001) as well as Universiti Teknologi MARA (UiTM) for the study leave of the first author.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Amri, N., Ismail, S., Azha, S.F. et al. Behaviors and Mechanism of Color, COD, and Silica Removals in the Electrocoagulation of Batik Wastewater Using Waste Aluminum Electrodes. Int J Environ Res 15, 509–525 (2021). https://doi.org/10.1007/s41742-021-00329-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41742-021-00329-x