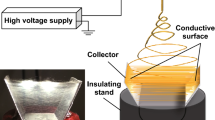
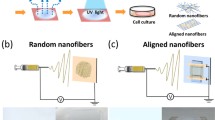
Abstract
Parallel fibrous scaffolds play a critical role in controlling the morphology of cells to be more natural and biologically inspired. Among popular tissue engineering materials, poly(2-hydroxyethyl methacrylate) (pHEMA) has been widely investigated in conventional forms due to its biocompatibility, low toxicity, and hydrophilicity. However, the swelling of pHEMA in water remains a major concern. To address this issue, randomly oriented and aligned as-spun pHEMA nanofibrous scaffolds were first fabricated at speeds of 300 and 2000 rpm in this study, which were then post-treated using either a thermal or a freeze-drying method. In cell assays, human dermal fibroblasts (HDFs) adhered to the freeze-drying treated substrates at a significantly faster rate, whereas they had a higher cell growth rate on thermally-treated substrates. Results indicated that the structural properties of pHEMA nanofibrous scaffolds and subsequent cellular behaviors were largely dependent on post-treatment methods. Moreover, this study suggests that aligned pHEMA nanofibrous substrates tended to induce regular fibroblast orientation and unidirectionally oriented actin cytoskeletons over random pHEMA nanofibrous substrates. Such information has predictive power and provides insights into promising post-treatment methods for improving the properties of aligned pHEMA scaffolds for numerous tissue engineering applications.
Graphic abstract








Similar content being viewed by others
References
Punnakitikashem D, Truong JU, Menon KT, Nguyen YH. Electrospun biodegradable elastic polyurethane scaffolds with dipyridamole release for small diameter vascular grafts. Acta Biomater. 2014;10(11):4618–28.
Jin G, Prabhakaran MP, Ramakrishna S. Stem cell differentiation to epidermal lineages on electrospun nanofibrous substrates for skin tissue engineering. Acta Biomater. 2011;7(8):3113–22.
Norouzi M, Boroujeni SM, Omidvarkordshouli N, Soleimani M. Advances in skin regeneration: application of electrospun scaffolds. Adv Healthc Mater. 2015;4(8):1114–33.
Yin Z, Chen X, Chen JL, Shen WL, Hieu Nguyen TM, Gao L, Ouyang HW. The regulation of tendon stem cell differentiation by the alignment of nanofibers. Biomaterials. 2009;31(8):2163–7215.
De MA, Seifalian AM, Birchall MA. Orchestrating cell/material interactions for tissue engineering of surgical implants. Macromol Biosci. 2012;12(8):1010–21.
Cordie T, Harkness T, Jing X, Carlson-Stevermer J, Mi HY, Turng LS. Nanofibrous electrospun polymers for reprogramming human cells. Cell Mol Bioeng. 2014;7(3):379–93.
Morgado PI, Aguiar-Ricardo A, Correia IJ. Asymmetric membranes as ideal wound dressings: an overview on production methods, structure, properties and performance relationship. J Membr Sci. 2015;490:139–51.
Sheikh FA, Ju HW, Lee JM, Bo MM, Park HJ, Lee OJ. 3D electrospun silk fibroin nanofibers for fabrication of artificial skin. Nanomed Nanotechnol. 2015;11(3):681–91.
Baker BM, Mauck RL. The effect of nanofiber alignment on the maturation of engineered meniscus constructs. Biomaterials. 2007;28(11):1967–77.
Talebian S, Foroughi J, Wade SJ, Vine KL, Dolatshahi-Pirouz A, Mehrali M, Conde J, Wallace GG. Biopolymers for antitumor implantable drug delivery systems: recent advances and future outlook. Adv Mater. 2018;30(31):1706665.
Chen SH, Chang Y, Lee KR, Lai JY. A three-dimensional dual-layer nano/microfibrous structure of electrospun chitosan/poly(d,l-lactide) membrane for the improvement of cytocompatibility. J Membr Sci. 2014;450:224–34.
Choi JS, Lee SJ, Christ GJ, Atala A, Yoo JJ. The influence of electrospun aligned poly(epsilon-caprolactone)/collagen nanofiber meshes on the formation of self-aligned skeletal muscle myotubes. Biomaterials. 2008;29(19):2899–906.
Hutchins-Kumar LD, Wang J, Tuzhi P. Parallel dual-electrode detection based on size exclusion for liquid chromatography/electrochemistry. Anal Chem. 1986;58(6):1019–23.
Katta P, Alessandro M, Ramsier RD, Chase GG. Continuous electrospinning of aligned polymer nanofibers onto a wire drum collector. Nano Lett. 2004;4(11):2215–8.
Liu SJ, Kau YC, Chou CY, Chen JK, Wu RC, Yeh WL. Electrospun plga/collagen nanofibrous membrane as early-stage wound dressing. J Membr Sci. 2010;355:53–9.
Agarwal S, Wendorff JH, Greiner A. Use of electrospinning technique for biomedical applications. Polymer. 2008;49(26):5603–21.
Yang C, Deng G, Chen W, Ye XJ, Mo XM. A novel electrospun-aligned nanoyarn-reinforced nanofibrous scaffold for tendon tissue engineering. Colloid Surf B. 2014;122:270–6.
Hurme T, Kalimo H, Lehto M, Jarvinen M. Healing of skeletal muscle injury: an ultrastructural and immunohistochemical study. Med Sci Sport Exerc. 1991;23(7):801–10.
Chew SY, Mi R, Hoke A, Leong KW. Aligned protein-polymer composite fibers enhance nerve regeneration: a potential tissue engineering platform. Adv Funct Mater. 2007;17(8):1288–96.
Lee CH, Shin HJ, Cho IH, Kang YM, Kim IM, Park KD. Nanofiber alignment and direction of mechanical strain affect the ECM production of human ACL fibroblast. Biomaterials. 2005;26(11):1261–70.
Qin S, Clark RA, Rafailovich MH. Establishing correlations in the en-mass migration of dermal fibroblasts on oriented fibrillar scaffolds. Acta Biomater. 2015;25:230–9.
Kai D, Prabhakaran MP, Jin G, Ramakrishna S. Guided orientation of cardiomyocytes on electrospun aligned nanofibers for cardiac tissue engineering. J Biomed Mater Res B. 2011;98(2):379–86.
Chew SY, Mi R, Hoke A, Leong KW. The effect of the alignment of electrospun fibrous scaffolds on Schwann cell maturation. Biomaterials. 2008;29(6):653–61.
Mi HY, Salick MR, Jing X, Crone WC, Peng XF, Turng LS. Electrospinning of unidirectionally and orthogonally aligned thermoplastic polyurethane nanofibers: fiber orientation and cell migration. J Biomed Mater Res A. 2015;103(2):593–603.
Liu Y, Ji Y, Ghosh K, Clark RAF, Huang L, Rafailovich MH. Effects of fiber orientation and diameter on the behavior of human dermal fibroblasts on electrospun PMMA scaffolds. J Biomed Mater Res A. 2009;90(4):1092–106.
Xu CY, Inai R, Kotaki M, Ramakrishna S. Aligned biodegradable nanofiber structure: a potential scaffold for blood vessel engineering. Biomaterials. 2004;25(5):877–86.
Dragusin DM, Van Vlierberghe S, Dubruel P, Dierick M, Van Hoorebeke L, Declercq HA, Cornelissen MM, Stancu IC. Novel gelatin-PHEMA porous scaffolds for tissue engineering applications. Soft Matter. 2012;8(37):9589–602.
Atzet S, Curtin S, Trinh P, Bryant S, Ratner B. Degradable poly(2-hydroxyethyl methacrylate)-co-polycaprolactone hydrogels for tissue engineering scaffolds. Biomacromol. 2008;9(12):3370–7.
Badea A, Mccracken JM, Tillmaand EG, Kandel ME, Oraham AW, Mevis MB, Rubakhin SS, Popescu G, Sweedler JV, Nuzzo RG. 3D-printed pHEMA materials for topographical and biochemical modulation of dorsal root ganglion cell response. ACS Appl Mater Interfaces. 2017;9(36):30318–28.
Ramalingam N, Natarajan TS, Rajiv S. Development and characterization of electrospun poly(2-hydroxy ethyl methacrylate) for tissue engineering applications. Adv Polym Tech. 2013;32(3):617–29.
Rao SS, Jeyapal SG, Rajiv S. Biodegradable electrospun nanocomposite fibers based on poly(2-hydroxy ethyl methacrylate) and bamboo cellulose. Compos Part B Eng. 2014;60:43–8.
Zhang B, Lalani R, Cheng F, Liu Q, Liu L. Dual-functional electrospun poly(2-hydroxyethyl methacrylate). J Biomed Mater Res A. 2011;99(3):455–66.
Hennink WE, De Jong SJ, Bos GW, Veldhuis TFJ, Van Nostrum CF. Biodegradable dextran hydrogels crosslinked by stereocomplex formation for the controlled release of pharmaceutical proteins. Int J Pharm. 2004;277(1–2):99–104.
Rampichová M, Martinová L, Košťáková E, Filová E, Míčková A, Buzgo M, Buzgo M, Michálek J, Přádný M, Nečas A, Lukáš D, Amler E. A simple drug anchoring microfiber scaffold for chondrocyte seeding and proliferation. J Mater Sci Mater Med. 2012;23(2):555–63.
Mabilleau G, Stancu IC, Honoré T, Legeay G, Cincu C, Baslé MF, Chappard D. Effects of the length of crosslink chain on poly(2-hydroxyethyl methacrylate) (pHEMA) swelling and biomechanical properties. J Biomed Mater Res A. 2006;77(1):35–42.
Yang F, Murugan R, Wang S, Ramakrishna S. Electrospinning of nano/micro scale poly(l-lactic acid) aligned fibers and their potential in neural tissue engineering. Biomaterials. 2005;26(15):2603–10.
Hu W, Yim EKF, Reano RM, Leong KW, Pang SW. Effects of nanoim printed patterns in tissue-culture polystyrene on cell behavior. J Vac Sci Technol B. 2005;23(6):2984–9.
Ramalingam N, Natarajan TS, Rajiv S. Preparation and characterization of electrospun curcumin loaded poly(2-hydroxyethyl methacrylate) nanofiber-A biomaterial for multidrug resistant organisms. J Biomed Mater Res A. 2015;103(1):16–24.
Song J, Saiz E, Bertozzi CR. Preparation of pHEMA-CP composites with high interfacial adhesion via template-driven mineralization. J Eur Ceram Soc. 2003;23(15):2905–19.
Song K, Zhu W, Li X, Yu Z. A novel mechanical robust, self-healing and shape memory hydrogel based on PVA reinforced by cellulose nanocrystal. Mater Lett. 2020;260:126884.
Liu GF, Zhang D, Feng CL. Control of three-dimensional cell adhesion by the chirality of nanofibers in hydrogels. Angew Chem Int Ed. 2014;53(30):7789–93.
Xu CY, Inai R, Kotaki M, Ramakrishna S. Aligned biodegradable nanofibrous structure: a potential scaffold for blood vessel engineering. Biomaterials. 2004;25(5):877–86.
Kurpinski KT, Stephenson JT, Janairo RRR, Lee H, Li S. The effect of fiber alignment and heparin coating on cell infiltration into nanofibrous PLLA scaffolds. Biomaterials. 2010;31(13):3536–42.
Acknowledgements
The authors appreciate Northeastern University for funding. The work is also supported by Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), National Natural Science Foundation of China (Grant no. 11372205 and 31900964), Program of Zhejiang Sci-Tech University (Grant no. 11110231281803), Scientific Research Foundation of Zhejiang Sci-Tech University (Grant no. 11112932618215) and the Fundamental Research Funds of Zhejiang Sci-Tech University (Grant no. 2020Q002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Shen, Q., Shen, J. et al. Multifunctional Fibroblasts Enhanced via Thermal and Freeze-Drying Post-treatments of Aligned Electrospun Nanofiber Membranes. Adv. Fiber Mater. 3, 26–37 (2021). https://doi.org/10.1007/s42765-020-00059-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42765-020-00059-3