Abstract
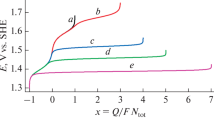
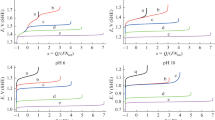
The kinetics of the formation of sodium chlorate during electrolysis of chloride solutions with use of dimensionally stable anodes is studied. An analysis and calculations suggest that the experimental results are well described by the proposed set of differential equations under the assumption that the homogeneous chemical reaction of chlorate formation has second, rather than third, order by active chlorine.
Similar content being viewed by others
REFERENCES
Evdokimov, S.V., Elektrokhimiya, 1999, vol. 35, p. 366.
Evdokimov, S.V., Elektrokhimiya, 1999, vol. 35, p. 202.
Kokoulina, D.V. and Krishtalik, L.I., Elektrokhimiya, 1971, vol. 7, p. 346.
Kheifets, L.I., Gol'dberg, A.B., and Mazanko, A.F., Itogi Nauki Tekh., Ser.: Elektrokhimiya, 1983, p. 244.
Yakimenko, L.M. and Seryshev, G.A., Elektrokhimicheskii sintez neorganicheskikh soedinenii (The Inorganic Electrosynthesis), Moscow: Khimiya, 1984, p. 35.
Vetter, K., Elektrochemische Kinetik, Berlin: Springer, 1961.
Damaskin, B.B. and Petrii, O.A., Vvedenie v elektrokhimicheskuyu kinetiku (Electrochemical Kinetics: An Introduction), Moscow: Vysshaya Shkola, 1975, p. 319.
Bagotzky, V.S., Osnovy elektrokhimii (Fundamentals of Electrochemistry), Moscow: Khimiya, 1988, p. 272.
Antropov, L.I., Teoreticheskaya electrokhimiya (Theoretical Electrochemistry), Moscow: Vysshaya Shkola, 1984, p. 324.
Rotinyan, A.L., Tikhonov, K.I., and Shoshina, I.A., Teoreticheskaya elektrokhimiya (Theoretical Electrochemistry), Leningrad: Khimiya, 1981, pp. 268, 302.
Evdokimov, S.V., Elektrokhimiya, 2001, vol. 37, p. 420.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Evdokimov, S.V. Kinetics of the Sodium Chlorate Formation in Electrolysis of Chloride Solutions with Use of Dimensionally Stable Anodes. Russian Journal of Electrochemistry 37, 786–791 (2001). https://doi.org/10.1023/A:1016774717213
Issue Date:
DOI: https://doi.org/10.1023/A:1016774717213