Abstract
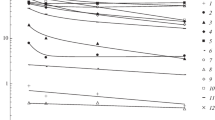
Transformation of the chemical composition of seawater upon interaction with loess, an analog of natural terrigenous aerosols of the Pleistocene, was studied experimentally. It was shown that this process leads to a change in the concentrations of major ions (K, Ca, SO4, and HCO3), nutrients (Si and P), and fluorine, while the chloride, sodium, and magnesium contents do not vary due to their high concentrations in seawater. With increasing weight fraction of the solid phase, the sulfate and hydrocarbonate concentrations increase, while the potassium concentration decreases. The calcium and fluorine concentrations either increase or decrease depending on the content of mobile forms of these elements in the samples. The release of silicon and phosphorus into seawater has an asymptotic limit: 2.5–3.3 mg Si/L and 0.012–0.040 mg P/L. The relationship between the change in concentrations of studied components in seawater and the weight fraction of the solid phase corresponds to a hyperbolic function, but with a relatively small weight fraction, it is described by a linear equation, in which the angular coefficient has a positive correlation with the concentration of the component in an aqueous extract from this loess sample. A change in seawater composition upon interaction with loess can be predicted from the composition of their aqueous extracts.

Similar content being viewed by others
REFERENCES
A. P. Vinogradov, Introduction into Geochemistry of the Ocean (Nauka, Moscow, 1967) [in Russian].
N. V. Komarova and Ya. S. Kamentsev, User’s Guide for Capillary Electrophoresis KAPEL (Veda, St. Petersburg, 2006) [in Russian].
V. N. Konishchev, “New studies of genesis of loess minerals,” Inzh. Geol., No. 5, 22–36 (2015).
N. I. Kriger, Loess: Properties and Relation with Geographic Environment (Nauka, Moscow, 1965) [in Russian].
Yu. Yu. Lur’e, Unified Water Analysis Methods (Khimiya, Moscow, 1971) [in Russian].
V. S. Savenko, Introduction into Ionometry of Natural Waters (Gidrometeoizdat, Leningrad, 1986) [in Russian].
V. S. Savenko and A. V. Savenko, Experimental Study of Low Temperature Geochemical Processes (GEOS, Moscow, 2009) [in Russian].
W. S. Broecker, “Abrupt climate change: causal constraints provided by the paleoclimate record,” Earth-Sci. Rev. 51 (1–4), 137–154 (2000).
B. Herut, R. Collier, and M. D. Krom, “The role of dust in supplying nitrogen and phosphorus to the Southeast Mediterranean,” Limnol. Oceanogr. 47 (3), 870–878 (2002).
H. B. Maring and R. A. Duce, “The impact of atmospheric aerosols on trace metal chemistry in open ocean surface seawater, 1. Aluminum,” Earth Planet. Sci. Lett. 84 (4), 381–392 (1987).
A. Nenes, M. D. Krom, N. Mihalopoulos, et al., “Atmospheric acidification on mineral aerosols: a source of bioavailable phosphorus for the oceans,” Atmos. Chem. Phys. 11 (13), 6265–6272 (2011).
F. Solmon, P. Y. Chuang, N. Meskhidze, and Y. Chen, “Acidic processing of mineral dust iron by anthropogenic compounds over the north Pacific Ocean,” J. Geophys. Res.: Atmos. 114 (2), D02305 (2009).
C.-E. Thuróczy, M. Boye, and R. Losno, “Dissolution of cobalt and zinc from natural and anthropogenic dusts in seawater,” Biogeosciences 7 (6), 1927–1936 (2010).
Funding
This study was supported by the Russian Foundation for Basic Research, project no. 18-05-01133.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by A. Bobrov
Rights and permissions
About this article
Cite this article
Savenko, A.V., Savenko, V.S. On the Transformation of the Chemical Composition of Seawater in Interaction with Terrigenous Aerosols. Oceanology 60, 742–747 (2020). https://doi.org/10.1134/S0001437020050215
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0001437020050215