Abstract—
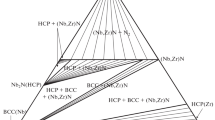
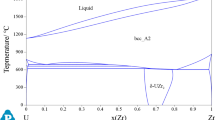
A thermodynamic model is proposed for condensed phases in the ternary system U–Zr–N in the range 298–2800 K. The model is based on previously reported models of the constituent binaries U–Zr, U–N, and Zr–N and available data on the properties of the pseudobinary system UN–ZrN. It allows one to calculate phase equilibria involving liquid phase. The model is used to calculate a number of isobaric–isothermal sections in the U–Zr–N system.





Similar content being viewed by others
REFERENCES
Kovalev, I.A., Shokodko, I.A., Shevtsov, S.V., Ogarkov, A.I., et al., Development of a fuel element on the basis of the composition (Zr,U)N for a high-temperature reactor, J. Phys.: Conf. Ser., 2018, vol. 1134, paper 012075. https://doi.org/10.1088/1742-6596/1134/1/012075
Perrot, P., Nitrogen–uranium–zirconium. Datasheet, Landolt–Börnstein, Group IV Physical Chemistry, vol. 11E3: Refractory Metal Systems, 2010, pp. 475–479. https://doi.org/10.1007/978-3-642-00771-2
Thetford, R. and Mignanelli, M., The chemistry and physics of modelling nitride fuels for transmutation, J. Nucl. Mater., 2003, vol. 320, nos. 1–2, pp. 44–53. https://doi.org/10.1016/S0022-3115(03)00170-3
Ogawa, T. and Akabori, M., Thermodynamic properties and multiphase diffusion paths of the ternary system U–Zr–N, J. Alloys Compd., 1994, vols. 213–214, pp. 173–177. https://doi.org/10.1016/0925-8388(94)90899-0
Ogawa, T., Akabori, M., Kobayashi, F., and Haire, R.G., Thermochemical modeling of actinide alloys related to advanced fuel cycles, J. Nucl. Mater., 1997, vol. 247, pp. 215–221. https://doi.org/10.1016/S0022-3115(97)00047-0
Akabori, M., Itoh, A., and Ogawa, T., Formation of nitrides at the surface of U–Zr alloys, J. Nucl. Mater., 2001, vol. 289, no. 3, pp. 342–345. https://doi.org/10.1016/S0022-3115(01)00426-3
Holleck, H. and Ishii, T., Berechnung von Gleichgewichten in ternaeren Systemen Uran–Uebergangsmetall–Stickstoff, Report no. KFK-1754, Karlsruhe: Kemforschungszentrum, 1973. https://doi.org/10.5445/IR/270005768
Holleck, H., Ternary phase equilibria in the systems actinide–transition metal–carbon and actinide–transition metal–nitrogen, Thermodynamics of Nuclear Materials: Proc. 4th Symp. (Vienna, 1975), Vienna: Int. Atomic Energy Agency, 1975, vol. 2, pp. 213–264. https://publikationen.bibliothek.kit.edu/270009072/ 3811449
Wei Xiong, Wei Xie, Chao Shen, Morgan, D., Thermodynamic modeling of the U–Zr system – A revisit, J. Nucl. Mater., 2013, vol. 443, nos. 1–3, pp. 331–341. https://doi.org/10.1016/j.jnucmat.2013.07.034
Chevalier, P.-Y., Fischer, E., and Cheynet, B., Thermodynamic modelling of the N–U system, J. Nucl. Mater., 2000, vol. 280, no. 2, pp. 136–150. https://doi.org/10.1016/S0022-3115(00)00043-X
Sridar, S., Kumar, R., and Hari Kumar, K.C., Thermodynamic modelling of Ti–Zr–N system, CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2017, vol. 56, pp. 102–107. https://doi.org/10.1016/j.calphad.2016.12.003
Xiaoyan Ma, Changrong Li, Kewu Bai, Ping Wu, and Weijing Zhang, Thermodynamic assessment of the Zr–N system, J. Alloys Compd., 2004, vol. 373, nos. 1–2, pp. 194–201. https://doi.org/10.1016/j.jallcom.2003.10.051
Punni, J.S. and Mignanelli, J.S., Investigation of the stability of U and U, Zr nitrides at high temperatures, Report no. AEAT/R/NS/0443, Harwell: AEAT, 2001.
Voskov, A.L., Dzuban, A.V., and Maksimov, A.I., TernAPI program for the calculation of ternary phase diagrams with isolated miscibility gaps by the convex hull method, Fluid Phase Equilib., 2015, vol. 388, pp. 50–58. https://doi.org/10.1016/j.fluid.2014.12.028
Funding
This work was supported by the Russian Science Foundation, project no. 20-13-00392 (federal state budget-funded science institution Baikov Institute of Metallurgy and Materials Science, Russian Academy of Sciences).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogarkov, A.I., Voskov, A.L., Kovalev, I.A. et al. Thermodynamic Modeling of Phase Equilibria in the U–Zr–N System. Inorg Mater 57, 790–797 (2021). https://doi.org/10.1134/S0020168521080070
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168521080070