Abstract—
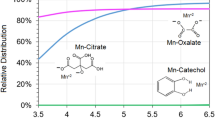
Granite is an important soil-forming rock and is widely distributed in the cold-temperate forest area of northeast China, such as northern Greater Hinggan, where its biochemical weathering plays an important role in the formation of local soils. Due to the special latitude and elevation, lichen plays a crucial role in the biochemical weathering of granite here, of which the role of low molecular organic acids (short for organic acids) is of interest. We simulated the concentrations of oxalic acid (OA) and succinic acid (SA) in the local lichens of Northeast China, applied OA or SA (10, 20, 30, 40, and 50 mmol L–1) to granite powders for 1 (10 min), 10, 20, 30, 40, 50, or 60 days, and studied the effects of organic acids on the bio-weathering process of granite, such as the dissolution regularity of various ions and the morphological changes of rock surfaces. OA and SA both induced the weathering and dissolving of granite, significantly promoted the release of Na+, K+, Al3+, Fe3+, Mg2+, Mn2+, Ca2+, and \({\text{SiO}}_{3}^{{2 - }}\) from granite powders. For the ion dissolution, the effect of OA was stronger than that of SA. Infrared analysis showed that both organic acids did not change the structures of their own groups, and the weathering mechanism was mainly the complexation of organic acids. The results of electron microscopy also presented the dissolution of granite powders by organic acids, and the effects were enhanced as the treatment time increased. After organic acid applications, the concentrations of dissolved ions from the granite powder varied with the type and concentration of organic acids, and treatment time. The concentration of each ion usually reached its maximum at 50 or 40 mmol L–1 OA, and 50 mmol L–1 SA (except for Fe3+ of some treatments). At day 1, the concentrations of \({\text{SiO}}_{3}^{{2 - }}\), Al3+, Fe3+, and Mn2+ treated with OA were higher than those of other times. Compared with SA, OA could dissolve more ions from granite powders, especially at day 1. However, the metal ions dissolved by OA were easy to complex with OA, so the concentrations of ions treated by SA were higher than those of OA, and the ions dissolved by OA would enter into the soils more frequently in OA–metal complexes.







Similar content being viewed by others
REFERENCES
M. Abdel-Hameed, R. L. Bertrand, M. D. Piercey-Normore, and J. L. Sorensen, “Putative identification of the usnic acid biosynthetic gene cluster by de novo whole-genome sequencing of a lichen-forming fungus,” Fungal Biol. 120, 306–316 (2016).
P. Adamo and P. Violante, “Weathering of rocks and neogenesis of minerals associated with lichen activity,” Appl. Clay Sci. 16, 229–256 (2000).
J. M. Arocena, T. Siddique, R. W. Thring, and S. Kapur, “Investigation of lichens using molecular techniques and associated mineral accumulations on a basaltic flow in a Mediterranean environment,” Catena 70, 356–365 (2007).
W. W. Barker and J. F. Banfield, “Biologically versus inorganically mediated weathering reactions: relationships between minerals and extracellular microbial polymers in lithobiontic communities,” Chem. Geol. 132, 55–69 (1996).
P. C. Bennett, “Quartz dissolution in organic-rich aqueous systems,” Geochim. Cosmochim. Acta 55, 1781–1797 (1991).
D. Białońska and F. E. Dayan, “Chemistry of the lichen Hypogymnia physodes transplanted to an industrial region,” J. Chem. Ecol. 31, 2975–2991 (2005).
S. L. Brantley and L. L. Stillings, “Feldspar dissolution at 25°C and low pH,” Am. J. Sci. 296, 101–127 (1996).
J. Cama, J. Ganor, C. Ayora, and C. A. Lasaga, “Smectite dissolution kinetics at 80°C and pH 8.8,” Geochim. Cosmochim. Acta 64 (15), 2701–2717 (2001).
J. Chen and H. P. Blume, “Rock-weathering by lichens in Antarctic: patterns and mechanisms,” J. Geogr. Sci. 12, 387–396 (2002).
J. Chen, H. P. Blume, and L. Beyer, “Weathering of rocks induced by lichen colonization-a review,” Catena 39, 121–146 (2000).
M. J. Eick, P. R. Grossl, D. C. Golden, D. L. Sparks, and D. W. Ming, “Dissolution of a lunar simulant as affected by pH and organic anions,” Geoderma 74, 139–160 (1996).
S. E. Favero-Longo, D. Castelli, O. Salvadori, E. Belluso, and R. Piervittori, “Pedogenetic action of the lichens Lecidea atrobrunnea, Rhizocarpon geographicum gr. and Sporastatia testudinea on serpentinized ultramafic rocks in an alpine environment,” Int. Biodeter. Biodegrad. 56, 17–27 (2005).
S. E. Favero-Longo, M. Girlanda, R. Honegger, B. Fubini, and R. Piervittori, “Interactions of sterile-cultured lichen-forming Ascomycetes with asbestos fibers,” Mycol. Res. 111, 473–481 (2007).
J. B. Fein and P. V. Brady, “Mineral surface controls on the diagenetic transport of oxalate and aluminum,” Chem. Geol. 121, 11–18 (1995).
G. Furrer and W. Stumm, “The coordination chemistry of weathering: I. Dissolution kinetics of δ-Al2O3 and BeO,” Geochim. Cosmochim. Acta 50, 1847–1860 (1986).
M. Hauck, J. Böning, M. Jacob, S. Dittrich, I. Feussner, and C. Leuschner, “Lichen substance concentrations in the lichen Hypogymnia physodes are correlated with heavy metal concentrations in the substratum,” Environ. Exp. Bot. 85, 58–63 (2013).
M. Hauck and S.-R. Jürgens, “Usnic acid controls the acidity tolerance of lichens,” Environ. Pollut. 156, 115–122 (2008).
S. J. M. Holmström, U. S. Lundström, R. D. Finlay, and P. A. W. van Hees, “Siderophores in forest soil solution,” Biogeochemistry 71, 247–258 (2004).
F. H. Hu, Dissertation (Henan Agricultural University, Zhengzhou, 2004).
Z. M. Li, Z. L. Song, J. F. Parr, and H. L. Wang, “Occluded C in rice phytoliths: implications to biogeochemical carbon sequestration,” Plant Soil 370, 615–623 (2013).
X. P. Liu, Laboratory Simulation of the Weathering Process of Granite with Oxalic Acid and Succinic Acid (Northeast Forestry University, Heilongjiang Sheng, 2017).
D. A. C. Manning, E. I. C. Rae, and J. S. Small, “An exploratory study of acetate decomposition and dissolution and Pb-rich potassium feldspar at 150°C, 50 MPa (500 bar),” Mineral. Mag. 55, 183–195 (1991).
B. Pawlik-Skowrońska and M. Bačkor, “Zn/Pb-tolerant lichens with higher content of secondary metabolites produce less phytochelatins than specimens living in unpolluted habitats,” Environ. Exp. Bot. 72, 64–70 (2001).
S. Payette and A. Delwaide, “Tamm review: The North-American lichen woodland,” For. Ecol. Manage. 417, 167–183 (2018).
F. Scarciglia, N. Saporito, M. F. La Russa, E. Le Pera, M. Macchione, D. Puntillo, M. G. Crisci, and A. Pezzino, “Role of lichens in weathering of granodiorite in the Sila uplands (Calabria, southern Italy),” Sediment. Geol. 280, 119–134 (2012).
M. R. D. Seaward, “Major impact made by lichens in biodederioration processes,” Int. Biodeter. Biodegrad. 40, 269–273 (1997).
I. G. Shirokikh, A. A. Shirokikh, and T. Y. Ashikhmina, “Assessing the antagonistic potential and antibiotic resistance of actinomycetes isolated from two zheltozems of southeastern China,” Eurasian Soil Sci. 51, 857–864 (2018).
W. Shotyk and W. H. Nesbitt, “Incongruent and congruent dissolution of plagioclase feldspar: effect of feldspar composition and ligand complexation,” Geoderma 55, 55–78 (1992).
M. M. Smits, A. M. Herrmann, M. Duane, W. O. Duckworth, S. Bonneville, G. L. Benning, and U. Lundström, “The fungal-mineral interface: challenges and considerations of microanalytical developments,” Fungal Biol. Rev. 23, 122–131 (2001).
T. A. Sokolova, I. I. Tolpeshta, L. V. Lysak, Y. A. Zavgorodnyaya, T. S. Chalova, M. M. Karpukhin, and Y. G. Izosimova, “Biological characteristics and concentrations of extractable Fe, Al, and Si compounds in spruce rhizosphere in podzolic soil,” Eurasian Soil Sci. 51, 1317–1325 (2018).
F. J. Stevenson, “Organic acids in soil,” in Soil Biochemistry, Ed. by A. D. Me Laren, and G. H. Peterson (Marcel Dekker, New York, 1967), pp. 110–146.
S. X. Tu, Z. F. Guo, and J. H. Sun, “Effect of oxalic acid on potassium release from typical Chinese soils and minerals,” Pedosphere 17, 457–466 (2007).
Y. Wang, F. H. Ren, X. F. Zhou, X. P. Yang, and Y. C. Li “Geochemical research on tectonic environment of early Cambrian granites in the Huzhong area of Heilongjiang province,” Geoscience 19, 141–146 (2005).
S. A. Welch and W. J. Ullman, “Feldspar dissolution in acidic and organic solutions: compositional and pH dependence of dissolution rate,” Geochim. Cosmochim. Assoc. 60, 2939–2948 (1996).
Q. F. Wu, H. B. Hu, and X. Zhang, “Effect of Aspergillus niger and its metabolites on weathering of granite,” J. Nanjing For. Univ., Nat. Sci. 42, 81–88 (2018).
J. Yan, X. Z. Han, Z. J. Ji, Y. Li, E. T. Wang, Z. H. Xie, and W. F. Chen, “Abundance and diversity of soybean-nodulating rhizobia in black soil are impacted by land use and crop management,” Appl. Environ. Microbiol. 80, 5394–5402 (2014).
Y. F. Zhou, X. C. Lu, R. C. Wang, and J. J. Lu, “Recent progress in the study of microbiomineralogy of feldspar,” Adv. Earth Sci. 23, 80–86 (2008).
ACKNOWLEDGMENTS
We thank Dr. Guoyou Chen (Heilongjiang Academy of Agricultural Sciences, China) for his assistance in the determination of ion concentrations. We are grateful to the editors and reviewers for their help and valuable suggestions.
Funding
This work was financially supported by the National Natural Science Foundation of China (31370613), the Fundamental Research Funds for the Central Universities (2572019CP15).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Song, J.F., Ru, J.X., Liu, X.P. et al. Oxalic Acid and Succinic Acid Mediate the Weathering Process of Granite in the Cold-Temperate Forest Regions of Northeast China. Eurasian Soil Sc. 52, 903–915 (2019). https://doi.org/10.1134/S1064229319080131
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1064229319080131