Abstract
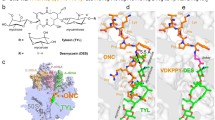
Fourteen new functionally active amino acid and peptide derivatives of the antibiotics tylosin, desmycosin, and 5-O-mycaminosyltylonolide were synthesized in order to study the interaction of the growing polypeptide chain with the ribosomal tunnel. The conjugation of various amino acids and peptides with a macrolide aldehyde group was carried out by two methods: direct reductive amination with the isolation of the intermediate Schiff bases or through binding via oxime using the preliminarily obtained derivatives of 2-aminooxy-acetic acid.
Similar content being viewed by others
Abbreviations
- Aoac:
-
2-aminooxyacetyl
- Des:
-
desmycosin
- DIEA:
-
N,N-diisopropylethylamine
- HBTU:
-
O-(benzotriazolyl)-1,1,3,3-tetramethyluronium hexafluorophosphate
- HOBt:
-
1-hydroxybenzotriazol
- OMT:
-
5-O-mycaminosyltylonolide
- PTC:
-
peptidyltransferase center
- RT:
-
ribosomal tunnel
- Tyl:
-
tylosi
- Ual:
-
3-(uracilyl-1-yl)alanine
References
Nissen, P., Hansen, J., Ban, N., Moore, P.B., and Steitz, T.A., Science, 2000, vol. 289, pp. 920–930.
Hardesty, B. and Kramer, G., Progr. Nucleic Acid Res. Mol. Biol., 2000, vol. 66, pp. 41–66.
Bogdanov, A.A., Molekulyarnaya biologiya, 2003, vol. 37, pp. 1–4.
Nakatogawa, H. and Ito, K., Cell, 2002, vol. 109, pp. 629–636.
Jenni, S. and Ban, N., Curr. Opin. Struct. Biol., 2003, vol. 13, pp. 212–219.
Gong, F. and Yanofsky, C., Science, 2002, vol. 297, pp. 1864–1867.
Yonath, A., Annu. Rev. Biochem., 2005, vol. 74, pp. 649–679.
Mankin, A.S., Trends Biochem. Sci., 2006, vol. 31, pp. 11–13.
Schlunzen, R., Zarivach, F., Harms, J., Bashan, A., Tocilj, A., Albrecht, R., Yonath, A., and Franceschi, F., Nature, 2001, vol. 413, pp. 814–821.
Hansen, J., Ippolito, J.A., Ban, N., Nissen, P., Moore, P.B., and Steitz, T.A., Moll. Cell, 2002, vol. 10, pp. 117–128.
Tenson, T., Lovmar, M., and Ehrenberg, M., J. Mol. Biol., 2003, vol. 330, pp. 1005–1014.
Tenson, T. and Ehrenberg, M., Cell, 2002, vol. 108, pp. 591–594.
Sumbatyan, N.V., Korshunova, G.A., and Bogdanov, A.A., Biokhimiya, 2003, vol. 68, pp. 1436–1438.
Korshunova, G.A., Sumbatyan, N.V., Fedorova, N.V., Kuznetsova, I.V., Shishkina, A.V., and Bogdanov, A.A., Bioorg. Khim., 2007, vol. 33, pp. 235–244 [Russ. J. Bioorg. Chem. (Engl. Tranl.), 2007, vol. 33, pp. 218–226].
McGuire, J., Bonience, W., Higgens, C., Hoehn, M., Stark, W., Westhead, J., and Wolfe, R., Antibiot. Chemother, 1961, vol. 11, pp. 320–327.
Hamill, R.L., Haney, M.E., McGuire, J.M., and Stamper, M.C., Antibiotics Tylosin and Desmycosin and Derivatives Thereof, US Patent No. 3 178 341, 1965.
Morin, R. and Gorman, M., O-Micaminosyl Tylonolide and Process for the Preparation Thereof, US Patent No. 3459853, 1969.
Vasquez-Laslop, N., Thum, C., and Mankin, A.S., Mol. Cell, 2008, vol. 30, pp. 190–202.
Debono, M., Willard, K.E., Kirst, H.A., Wind, J.A., Crouse, G.D., Tao, E.V., Vicenzi, J.T., Counter, F.T., Ott, J.L., and Ose, E.E., J. Antibiot., 1989, vol. 42, pp. 1253–1267.
Kirst, H.A., Willard, K.E., Debono, M., Toth, J.E., Truedell, B.A., Leeds, J.P., Ott, J.L., Felty-Duckworth, A.M., Counter, F.T., Ose, E.E., Crouse, G.D., Tustin, J.M., and Omura, S., J. Antibiot., 1989, vol. 42, pp. 1673–1683.
Mutak, S., Marsik, N., Kramaric, M.D., and Pavlovic, D., J. Med. Chem., 2004, vol. 47, pp. 411–431.
Lundi, K.M. and Vu, C.B., Amide derivatives of 16-Membered Ring Antibiotic Macrolides, US Patent No. 5716939, 1998.
Yang, D., Ng, F.F., and Li, Z.J., J. Am. Chem. Soc., 1996, vol. 118, pp. 9794–9795.
Woolhead, C.A., McCormick, P.J., and Johnson, A.E., Cell, 2004, vol. 116, pp. 725–736.
Lu, J. and Deutsch, C., Nat. Struct. Mol. Biol., 2005, vol. 12, pp. 1123–1129.
Finkel’shtein, A.V. and Ptitsin, O.B., Fizika belka: Kurs lektsii s tsvetnymi i stereoskopicheskimi illyustratsiyami i zadachami: 3-e izd (Protein Physics: A Course of Lectures with Color Stereoscopic Illustrations and Problems, 3rd ed.), Moscow: KDU, 2005.
Bonora, G.M., Nisato, D., and Toniolo, C., Makromol. Chem., 1975, vol. 176, pp. 2535–2545.
Lorenzi, G.P., Rizzo, V., Thoresen, F., and Tomasic, L., Macromolecules, 1979, vol. 12, pp. 870–874.
Han, S.Y. and Kim, Y.A., Tetrahedron, 2004, vol. 60, pp. 2447–2467.
Pshenichnikova, A.B., Karnaukhova, E.N., Mitsner, B.I., and Shchvets, V.I., Bioorg. Khim., 1996, vol. 10, pp. 852–855.
Debono, M. and Kirst, H.A., C-20-Dihydro-Deoxy-(Cyclic Amino)-Derivatives of Macrolide Antibiotics, US Patent No. 4820695, 1989.
Rossi, C., Donati, A., Picci, M.P., Sansoni, M.R., Corbini, G., and Corti, P., Magn. Reson. Chem., 1992, vol. 30, pp. 954–957.
Naranda, A. and Lopotar, N., J. Antibiot., 1999, vol. 52, pp. 68–70.
RTS 100 E. coli HY Kit. Instruction Manual, Mannhein: Roche Diagnostics, 2007.
Dinos, G., Wilson, D.N.., Teraoka, Y., Szaflarski, W., Fucini, P., Kalpaxis, D., and Nierhaus, K.H., Molecular Cell, 2004, vol. 13, pp. 113–124.
Harmsen, D., Erker, G., Froehlich, R., and Kehr, G., Eur. J. Inorg. Chem., 2002, pp. 3156–3171.
Shields, J.E., McDowell, S.T., Pavlos, J., and Gray, G.R., J. Am. Chem. Soc., 1968, vol. 90, pp. 3549–3556.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.V. Sumbatyan, I.V. Kuznetsova, V.V. Karpenko, N.V. Fedorova, V.A. Chertkov, G.A. Korshunova, A.A. Bogdanov, 2010, published in Bioorganicheskaya Khimiya, 2010, Vol. 36, No. 2, pp. 265–276.
Rights and permissions
About this article
Cite this article
Sumbatyan, N.V., Kuznetsova, I.V., Karpenko, V.V. et al. Amino acid and peptide derivatives of the tylosin family of macrolide antibiotics modified by aldehyde function. Russ J Bioorg Chem 36, 245–256 (2010). https://doi.org/10.1134/S1068162010020159
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162010020159