Abstract
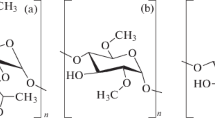
The interaction between 2-hydroxypropyl β-cyclodextrin (HPCD) and a liposomal bilayer has been studied. The main binding sites of HPCD on the surface of neutral liposomes based on dipalmitoylphosphatidylcholine (DPPC) are the phosphate groups of lipids. The complex formation with HPСD leads to the stabilization of the gel state of monocomponent liposomes. The inclusion of the anionic component, cardiolipin (CL, 20%), in the bilayer changes the nature of the liposome-HPCD interaction. The observed lipid disorder disturbs membrane integrity, which is revealed in the release of the included dye (phenolphthalein). The effect of HPCD on the process and parameters of the phase transition of anionic liposomes has been studied using thermograms, which show the change in the position of the absorption bands of lipid acyl chains in the FTIR spectrum of liposomes. A stratification of DPPC/CL (80/20%) bilayer into two microphases with different cardiolipin content has been detected. HPCD causes the more pronounced stratification in the bilayer, i.e., membrane destabilization near the melting point of CL-rich microphase and, vice versa, a decrease in the lipid mobility in regions with a low CL content. We have studied the effect of HPCD on the interaction of an antibacterial drug, levofloxacin (LV), with the lipid bilayer. It has been found that the complexation of the drug molecules with HPCD leads to an increase in the efficiency of drug adsorption on the surface of the bilayer. This may facilitate the transport of the drug through the bilayer due to the formation of defects in the membrane. LV in the complex with HPCD shows a high antibacterial efficiency in vitro against E. coli, which is not lower as compared with free LV.






Similar content being viewed by others
REFERENCES
Loftsson, T., Jarho, P., Masson, M., and Jarvinen, T., Adv. Drug Deliv. Rev., 2005, vol. 36, pp. 335–346. https://doi.org/10.1016/S0169-409X(98)00051-9
Stella, V.J. and He, Q., Toxicol. Pathol., 2008, vol. 36, pp. 30–42. https://doi.org/10.1177/0192623307310945
Crini, G., Fourmentin, S., Fenyvesi, E., Torri, G., Fourmentin, M., and Morin-Crini, N., Environ. Chem. Lett. Springer Int. Publ., 2018, vol. 16, no. 4, pp. 1361–1375. https://doi.org/10.1007/s10311-018-0763-2
Del Valle, E.M.M., Process Biochem., 2004, vol. 39, pp. 1033–1046. https://doi.org/10.1016/S0032-9592(03)00258-9
Davis, M.E. and Brewster, M., Nat. Rev. Drug Discov., 2004, vol. 3, pp. 1023–1035. https://doi.org/10.1038/nrd1576
Le-Deygen, I.M., Skuredina, A.A., Uporov, I.V., and Kudryashova, E.V., Anal. Bioanal. Chem., 2017, vol. 409, pp. 6451–6462. https://doi.org/10.1007/s00216-017-0590-5
Skuredina, A.A., Le-Deygen, I.M., Uporov, I.V., and Kudryashova, E.V., Colloid J., 2017, vol. 79, pp. 668–676.
Skuredina, A.A., Le-Deygen, I.M., and Kudryashova, E.V., Colloid J., 2018, vol. 80, pp. 312–319. https://doi.org/10.1134/S1061933X17050143
Saltzman, W.M. and Kyriakides, T.R., in Principles of Tissue Engineering, 4th ed., Elsevier, 2013, pp. 385–406. https://doi.org/10.1016/B978-0-12-398358-9.00020-3
Schulz, M., Olubummo, A., and Binder, W.H., Soft Matter., 2012, vol. 8, no. 18, pp. 4849–4864. https://doi.org/10.1039/c2sm06999g
Hammoud, Z., Khreich, N., Auezova, L., Fourmentin, S., Elaissari, A., and Greige-Gerges, H., Int. J. Pharm. Elsevier, 2019, vol. 564, pp. 59–76. https://doi.org/10.1016/j.ijpharm.2019.03.063
Challa, R., Ahuja, A., Ali, J., and Khar, R.K., AAPS Pharm. Sci. Tech., 2005, vol. 2, pp. 329–357. https://doi.org/10.1517/17425247.2.1.335
Le-Deygen, I.M., Skuredina, A.A., Safronova, A.S., Yakimov, I.D., Kolmogorov, I.M., Deygen, D.M., Burova, T.V., Grinberg, N.V., Grinberg, V.Y., and Kudryashova, E.V., Chem. Phys. Lipids. Elsevier Ireland Ltd, 2020, vol. 228, p. 104 891. https://doi.org/10.1016/j.chemphyslip.2020.104891
Le-Deigen, I.M., Skuredina, A.A., and Kudryashova, E.V., Russ. J. Bioorg. Chem., 2020 (in press).
Deygen, I.M., Seidl, C., Kolmel, D.K., Bednarek, C., Heissler, S., Kudryashova, E.V., Brase, S., and Schepers, U., Langmuir, 2016, vol. 32, pp. 10 861–10 869. https://doi.org/10.1021/acs.langmuir.6b01023
Piel, G., Piette, M., Barillaro, V., Castagne, D., Evrard, B., and Delattre, L., Int. J. Pharm., 2007, vol. 338, pp. 35–42. https://doi.org/10.1016/j.ijpharm.2007.01.015
Yaroslavov, A.A., Efimova, A.A., Lobyshe, V.I., and Kabanov, V.A., Biochim. Biophys. Acta, 2002, vol. 1560, pp. 14–24. https://doi.org/10.1016/S0005-2736(01)00453-9
Bilge, D., Sahin, I., Kazanci, N., and Severcan, F., Spectrochim. Acta, A: Mol. Biomol. Spectrosc., Elsevier, 2014, vol. 130, pp. 250–256. https://doi.org/10.1016/j.saa.2014.04.027
Donova, M.V., Nikolayeva, V.M., Dovbnya, D.V., Gulevskaya, S.A., and Suzina, N.E., Microbiology, 2007, vol. 153, pp. 1981–1992. https://doi.org/10.1099/mic.0.2006/001636-0
Le-Deygen, I.M., Vlasova, K.Y., Kutsenok, E.O., Usvaliev, A.D., Efremova, M.V., Zhigachev, A.O., Rudakovskaya, P.G., Golovin, D.Y., Gribanovsky, S.L., Kudryashova, E.V., Majouga, A.G., Golovin, Y.I., Kabanov, A.V., and Klyachko, N.L., Nanomed., Nanotechnol., Biol. Med., Elsevier Inc, 2019, vol. 21, p. 102 065. https://doi.org/10.1016/j.nano.2019.102065
Hatzi, P., Mourtas, S.G., Klepetsanis, P., Antimisiaris, S.G., Int. J. Pharm., 2007, vol. 333, pp. 167–176. https://doi.org/10.1016/j.ijpharm.2006.09.059
Angelini, G., Campestre, C., Boncompagni, S., and Gasbarri, C., Chem. Phys. Lipids, Elsevier Ireland Ltd., 2017, vol. 209, pp. 61–65. https://doi.org/10.1016/j.chemphyslip.2017.09.004
Bertucci, C., Pistolozzi, M., and De Simone, A., Anal. Bioanal. Chem., 2010, vol. 398, pp. 155–166. https://doi.org/10.1007/s00216-010-3959-2
Chamseddin, C. and Jira, T., Curr. Pharm. Anal., 2013, vol. 9, pp. 121–129.
Goddard, J.M., Caput, D., Williams, S.R., and Martin, D.M., Proc. Natl. Acad. Sci. U. S. A., vol. 80, pp. 4281–4285. https://doi.org/10.1073/pnas.80.14.4281
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies with the use of humans as objects of research.
Conflict of Interest
The authors state that there is no conflict of interest.
Additional information
Translated by A. Levina
Abbreviations: HPCD, 2-hydroxypropyl β-cyclodextrin; DPPC, dipamitoylphosphatidylcholine; CL, cardiolipin; LV, levofloxacin; MIC, minimal inhibitory concentration; PP, phenolphthalein; CD, cyclodextrin.
Corresponding author: phone: +7 (495) 939-34-34; e-mail: skuredinanna@gmail.com.
Rights and permissions
About this article
Cite this article
Skuredina, A.A., Tychinina, A.S., Le-Deygen, I.M. et al. Regulation of Properties of Lipid Membranes by Interaction with 2-Hydroxypropyl β-Cyclodextrin: Molecular Details. Russ J Bioorg Chem 46, 692–701 (2020). https://doi.org/10.1134/S1068162020050246
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162020050246