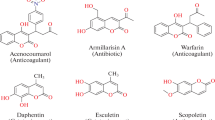
Abstract
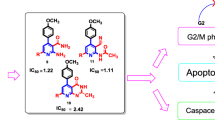
A new series of coumarin-derived molecules have been synthesized and evaluated for their anticancer activity against human hepatocellular liver carcinoma cell line (HepG2). Compound (IX) showed the least IC50 values in MTT colorimetric assay and significantly inhibited topoisomerase IIβ. DNA flow cytometry assay of compound (IX) revealed cell cycle arrest at G2/M phase and activation of apoptosis as verified by changes in cell cycle kinetics. Further mechanism of apoptosis showed that, compound (IX) induced cell apoptosis probably through the intrinsic mitochondrial pathway of apoptosis. This mechanistic pathway was confirmed by a significant increase in the level of p53, Bax and decrease in the level of Bcl-2 compared to control.







Similar content being viewed by others
REFERENCES
Siegel, R.L., Jemal, A., Wender, R.C., Gansler, T., Ma, J., and Brawley, O.W., CA Cancer J. Clin., 2018, vol. 68, pp. 329–339. https://doi.org/10.3322/caac.21460
Zhou, F., Shang, W., Yu, X., and Tian, J., Med. Res. Rev., 2018, vol. 38, pp. 741–767. https://doi.org/10.1002/med.21455
Pommier, Y., Leo, E., Zhang, H., Marchand, C., Chem. Biol., 2010, vol. 17, pp. 421–433. https://doi.org/10.1016/j.chembiol.2010.04.012
Abdelhaleem, E.F., Abdelhameid, M.K., Kassab, A.E., and Kandeel, M.M., Eur. J. Med. Chem., 2018, vol. 143, pp. 1807–1825. https://doi.org/10.1016/j.ejmech.2017.10.075
Nitiss, J.L., Nat. Rev. Cancer, 2009, vol. 9, pp. 338–350. https://doi.org/10.1038/nrc2607
Wang, N., Zhu, M., Tsao, S.-W., Man, K., Zhang, Z., and Feng, Y., Mol. Cancer, 2013, vol. 12, pp. 119. https://doi.org/10.1186/1476-4598-12-119
Sudan, S. and Rupasinghe, H.V., Anticancer Res., 2014, vol. 34, pp. 1691–1699. http://ar.iiarjournals.org/content/34/4/1691.abstract.
Elmore, S., Toxicol. Pathol., 2007, vol. 35, pp. 495–516. https://doi.org/10.1080/01926230701320337
Rashad, M.S., Georgey, H.H., George, R.F., and Abdel-Gawad, N.M., Future Med. Chem., 2018, vol. 10, pp. 1649–1664. https://doi.org/10.4155/fmc-2018-0068
Kim, R., Emi, M., and Tanabe, K., Cancer Biol. Ther., 2005, vol. 4, pp. 924–933. https://doi.org/10.4161/cbt.4.9.2101
Abdelhameid, M.K., Zaki, I., Mohammed, M.R., and Mohamed, K.O., Bioorg. Chem., 2020, vol. 101, pp. 103995–104007. https://doi.org/10.1016/j.bioorg.2020.103995
Zaki, I., Abdelhameid, M.K., El-Deen, I.M., Abdel Wahab, A.H.A., Ashmawy, A.M., and Mohamed, K.O., Eur. J. Med. Chem., 2018, vol. 156, pp. 563–579. https://doi.org/10.1016/j.ejmech.2018.07.003
Liu, Y.-P., Yan, G., Xie, Y.-T., Lin, T.-C., Zhang, W., Li, J., Wu, Y.-J., Zhou, J.-Y., and Fu, Y.-H., Bioorg. Chem., 2020, vol. 97, pp. 103699–103713. https://doi.org/10.1016/j.bioorg.2020.103699
Swain, B., Angeli, A., Singh, P., Supuran, C.T., and Arifuddin, M., Bioorg. Med. Chem., 2020, vol. 28, pp. 115586–115597. https://doi.org/10.1016/j.bmc.2020.115586
Ahmed, E.Y., Abdel Latif, N.A., El-Mansy, M.F., Elserwy, W.S., and Abdelhafez, O.M., Bioorg. Med. Chem., 2020, vol. 28, pp. 115328–115338. https://doi.org/10.1016/j.bmc.2020.115328
Zhang, L. and Xu, Z., Eur. J. Med. Chem., 2019, vol. 181, pp. 111587. https://doi.org/10.1016/j.ejmech.2019.111587
Manolov, I., Maichle-Moessmer, C., and Danchev, N., Eur. J. Med. Chem., 2006, vol. 41, pp. 882–890. https://doi.org/10.1016/j.ejmech.2006.03.007
Wang, J., Lu, M.L., Dai, H.L., Zhang, S.P., Wang, H.X., and Wei, N., Braz. J. Med. Biol. Res., 2015, vol. 48, pp. 245–253. https://doi.org/10.1590/1414-431x20144074
Prasad, K.N., Xie, H., Hao, J., Yang, B., Qiu, S., Wei, X., Chen, F., and Jiang, Y., Food Chem., 2010, vol. 118, pp. 62–66. https://doi.org/10.1016/j.foodchem.2009.04.073
Hejchman, E., Taciak, P., Kowalski, S., Maciejewska, D., Czajkowska, A., Borowska, J., Śladowski, D., and Młynarczuk-Biały, I., Pharmacol. Rep., 2015, vol. 67, pp. 236–244. https://doi.org/10.1016/j.pharep.2014.09.008
Nasr, T., Bondock, S., and Youns, M., Eur. J. Med. Chem., 2014, vol. 76, pp. 539–548. https://doi.org/10.1016/j.ejmech.2014.02.026
Zhao, P., Chen, L., Li, L.-H., Wei, Z.-F., Tong, B., Jia, Y.-G., Kong, L.-Y., Xia, Y.-F., and Dai, Y., BMC Cancer, 2014, vol. 14, pp. 987–1005. https://doi.org/10.1186/1471-2407-14-987
El-Deen, I., Elgareib, M.S., Mahdy, A.R., and Al-Saleem, M.S., Mens Agitat, 2018, vol. 13, pp. 1–5.
Moustafa, A.M.Y. and Bakare, S.B., Res. Chem. Intermed., 2019, vol. 45, pp. 3895–3912. https://doi.org/10.1007/s11164-019-03827-y
Rahman, A.U., Medrano, M.A., and Mittal, O., Recl. Trav. Chim. Pays-Bas, 1960, vol. 79, pp. 188–192.
Menezes, J.C. and Diederich, M., Future Med. Chem., 2019, vol. 11, pp. 1057–1082. https://doi.org/10.4155/fmc-2018-0375
Mohamed, K.O., Zaki, I., El-Deen, I.M., and Abdelhameid, M.K., Bioorg. Chem., 2019, vol. 84, pp. 399–409. https://doi.org/10.1016/j.bioorg.2018.12.007
Wei, H., Ruthenburg, A.J., Bechis, S.K., and Verdine, G.L., J. Biolog. Chem., 2005, vol. 280, pp. 37041–37047. https://doi.org/10.1074/jbc.M506520200
Funding
This work was funded by the authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving animals or human participants performed by any of the authors.
Conflict of Interests
The authors report no conflicts of interest.
Supplementary Information
Rights and permissions
About this article
Cite this article
Zaki, I., El-Sayed, ES.H. & Radwan, E.M. Synthesis and Antiproliferative Activity of Some New Coumarin Derivatives Derived from 8-Hydroxycoumarin. Russ J Bioorg Chem 47, 514–523 (2021). https://doi.org/10.1134/S106816202102028X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106816202102028X