Abstract
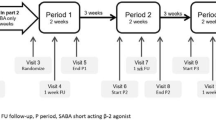
Current asthma treatment guidelines recommend a reduction in inhaled corticosteroid (ICS) doses when appropriate. However, no standardized taper protocols are available to guide ICS tapering in clinical trials or in general practice. This study was designed to determine the safety and effectiveness of tapering the ICS dose in asthmatic patients using specific tapering and rescue protocols. Twenty-two clinically stable asthmatic patients on moderate-to-high doses of ICS were studied. Patients were given either montelukast (10 mg daily) or placebo for 8 weeks and evaluated every 2 weeks. A composite clinical score (0 to 3) comprising lung function, symptoms, and “as-needed” β-agonist use was calculated at each visit. Based on this score, the ICS dose was either tapered, increased, or maintained at each visit. The minimum ICS dose that maintained clinical stability was defined as the last tolerated dose.
The results showed that mean percent change from baseline (1523 μg/day, SD = 536) in the last tolerated dose was 49.8% (SD = 37.4). Sixteen patients tolerated at least one taper; of these, 12 tolerated 2 or more tapers. Seven patients tapered completely off ICS. In contrast, six patients did not tolerate a single taper. Ten patients required rescue from mild worsening with an increase in ICS dose, of which nine were successful and one achieved stability with a second ICS dose increase two weeks later. Overall, patients remained clinically stable during tapering. It is concluded that tapering of ICS guided by the clinical composite score was well tolerated. The rescue protocol was effective in restabilizing patients. The results of this study thus support the use of a composite clinical score in larger clinical trials.
Similar content being viewed by others
References
Lipworth BJ, Wilson AM. Dose response to inhaled corticosteroid: Benefits and Risks. Seminars Respir Critical Care Med. 1998;6:625–646.
Doull IJ, Freezer NJ, Holgate ST. Growth of prepubertal children with mild asthma treated with inhaled beclomethasone dipropionate. Am J Respir Crit Care Med. 1995;151:1715–1719.
National Heart, Lung, and Blood Institute. Expert Panel Report 2: Guidelines for the Diagnosis and Management of Asthma. Bethesda, MD: National Heart, Lung, and Blood Institute: Lund 1997. NIH Publication No. 97–4051.
Santanello NC, Barber BL, Reiss TF, Friedman BS, Juniper EF, Zhang I. Measurement characteristics of two asthma symptom diary scale for use in clinical trials. Eur Respir J. 1997;10:646–651.
Zhang J, Song C, Reiss TF. Development and validation of a sensitive measure of worsening asthma. Am J Resp Crit Care Med. 1997;155:A892.
Erzurum SE, Leff JA, Cochran JE, Ackerson LM, Szefler SJ, Martin RJ, Cott GR. Lack of benefit of methotrexate in severe, steroid-dependent asthma. Ann Int Med. 1991;114:353–360.
Lofdahl C-G, Reiss TF, Leff JA, Israel E, Noonan MJ, Finn AF, Seidenberg BC, Capizzi T Kundu S, Godard P. Randomized, placebo-controlled trial of effect of a leukotriene receptor antagonist, on tapering inhaled corticosteroids in asthmatic patients. Br Med J. 1999;318–87–90.
Price DB, Rouleau MY, Fletcher CP, Patel P, Olivenstein R, Myhr L, Virchow JC, Aitchison WRC, Omenaas ER, Dellea PS, Laurenzi M, Leff JA. Use of Montelukast in tapering inhaled corticosteroid therapy: An open-label, 48-week trial. Current Therapeutic Research; 2002.
Teeter JG, Bleeker ER. Relationship between airway obstruction and respiratory symptoms in adult asthmatics. Chest. 1998;113:272–277.
Shingo S, Zhang J, Reiss TF. Correlation of airway obstruction and patient-reported endpoints in clinical studies. European Resp J. 2001;17:220–224.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was funded by a grant from Merck Research Laboratories, Rahway, NJ, USA.
Rights and permissions
About this article
Cite this article
Shingo, S., Zhang, J., Noonan, N. et al. A Standardized Composite Clinical Score for Inhaled Corticosteroids Taper Studies in Asthma. Ther Innov Regul Sci 36, 501–508 (2002). https://doi.org/10.1177/009286150203600304
Published:
Issue Date:
DOI: https://doi.org/10.1177/009286150203600304