Abstract
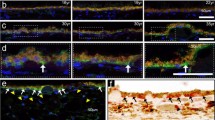
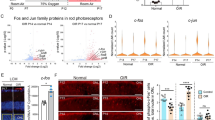
Clinically, a large proportion of glaucoma patients undergo repeated intraocular pressure (IOP) spike (Spike IOP) attacks during their sleep, which may facilitate retinopathy. In this study, we established a mouse model of repeated transient Spike IOP to investigate the direct damage to the retina following Spike IOP attacks, and elucidated the underlying molecular mechanism. We analyzed the changes in the number of retinal ganglion cells (RGCs) via immunofluorescence. Thereafter, we detected retinal cell apoptosis via terminal deoxynucleotidyl transferase deoxyuridine triphosphate (dUTP) nick-end labeling (TUNEL) staining, and performed RNA sequencing (RNA-seq) to reveal the underlying molecular mechanism. Finally, we validated the expression of key molecules in the endoplasmic reticulum (ER) stress pathway using quantitative real-time polymerase chain reaction (qRT-PCR) and western blot analysis. Results revealed a time-dependent RGC loss in Spike IOP, evidenced by a reduction in the number of Brn3a-positive RGCs in experimental eyes following a 7-d continuous treatment with Spike IOP. In addition, TUNEL staining indicated that apoptosis of retinal cells started in the outer nuclear layer (ONL), and then spread to the ganglion cell layer (GCL) with time. RNA-seq analysis revealed that ER stress might be involved in Spike IOP-induced retinal injury. This result was corroborated by western blot, which revealed upregulation of ER stress-related proteins including binding immunoglobulin protein/glucose-regulated protein 78 (BiP/GRP78), phosphorylated inositol-requiring enzyme 1 (p-IRE1), unspliced X-box-binding protein 1 (XBP1-u), spliced X-box-binding protein 1 (XBP1-s), phosphorylated c-Jun N-terminal kinase (p-JNK), C/EBP-homologous protein (CHOP), and B-cell lymphoma 2 (Bcl-2)-associated X protein (Bax). These findings indicate that repeated IOP transients are detrimental to the retina, while ER stress plays an important role in retinal cell apoptosis in this situation. Notably, repeated Spike IOP among glaucoma patients is a crucial factor for progressive retinopathy.
概要
目的
研究和探讨反复瞬时高眼压对视网膜的损伤作用及其潜在的分子机制.
创新点
模拟临床上青光眼患者夜间难以检测到的眼压峰值波动对视网膜的影响, 首次证明反复的瞬时高眼压对视网膜具有直接损伤效应, 其损伤机制与内质网应激通路激活有关.
方法
通过眼前房生理盐水灌注, 建立小鼠反复瞬时高眼压模型(50 mmHg, 1 min× 7次), 连续灌注处理1、 3和7天后, 采用视网膜铺片(Whole-mount retina)评估视网膜神经节细胞(RGC)损伤情况; 用TUNEL法检测视网膜全层细胞凋亡; 用转录组测序(RNA-seq)筛选参与视网膜损伤的分子通路; 用实时荧光定量聚合酶链式反应(qRT-PCR)和蛋白免疫印迹(Western blot)进一步检测内质网应激通路相关分子的表达.
结论
反复瞬时高眼压可以损伤视网膜全层, 并呈时间依赖性的由外核层细胞(ONL)死亡进展至视网膜神经节细胞层(GCL)死亡. 内质网应激相关信号通路中肌醇酶1(IRE1)信号通路激活参与了视网膜的损伤过程.
Similar content being viewed by others
References
Asrani S, Zeimer R, Wilensky J, et al., 2000. Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma. J Glaucoma, 9(2): 134–142. https://doi.org/10.1097/00061198-200004000-00002
Barkana Y, Anis S, Liebmann J, et al., 2006. Clinical utility of intraocular pressure monitoring outside of normal office hours in patients with glaucoma. Arch Ophthalmol, 124(6): 793–797. https://doi.org/10.1001/archopht.124.6.793
Choi SS, Zawadzki RJ, Lim MC, et al., 2011. Evidence of outer retinal changes in glaucoma patients as revealed by ultrahigh-resolution in vivo retinal imaging. Br J Ophthalmol, 95(1):131–141. https://doi.org/10.1136/bjo.2010.183756
di Pierdomenico J, García-Ayuso D, Pinilla I, et al., 2017. Early events in retinal degeneration caused by rhodopsin mutation or pigment epithelium malfunction: differences and similarities. Front Neuroanat, 11:14. https://doi.org/10.3389/fnana.2017.00014
di Pierdomenico J, García-Ayuso D, Agudo-Barriuso M, et al., 2019. Role of microglial cells in photoreceptor degeneration. Neural Regen Res, 14(7):1186–1190. https://doi.org/10.4103/1673-5374.251204
García-Ayuso D, Ortín-Martínez A, Jiménez-López M, et al., 2013. Changes in the photoreceptor mosaic of P23H-1 rats during retinal degeneration: implications for rod-cone dependent survival. Invest Ophthalmol Vis Sci, 54(8): 5888–5900. https://doi.org/10.1167/iovs.13-12643
García-Ayuso D, di Pierdomenico J, Agudo-Barriuso M, et al., 2018. Retinal remodeling following photoreceptor degeneration causes retinal ganglion cell death. Neural Regen Res, 13(11):1885–1886. https://doi.org/10.4103/1673-5374.239436
Hetz C, Saxena S, 2017. ER stress and the unfolded protein response in neurodegeneration. Nat Rev Neurol, 13(8): 477–491. https://doi.org/10.1038/nrneurol.2017.99
Hetz C, Thielen P, Matus S, et al., 2009. XBP-1 deficiency in the nervous system protects against amyotrophic lateral sclerosis by increasing autophagy. Genes Dev, 23(19): 2294–2306. https://doi.org/10.1101/gad.1830709
Hou RW, Zhang Z, Yang DY, et al., 2016. Pressure balance and imbalance in the optic nerve chamber: The Beijing Intracranial and Intraocular Pressure (iCOP) study. Sci China Life Sci, 59(5):495–503. https://doi.org/10.1007/s11427-016-5022-9
Hu Y, Park KK, Yang L, et al., 2012. Differential effects of unfolded protein response pathways on axon injury-induced death of retinal ganglion cells. Neuron, 73(3):445–452. https://doi.org/10.1016/j.neuron.2011.11.026
Kim I, Xu WJ, Reed JC, 2008. Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov, 7(12):1013–1030. https://doi.org/10.1038/nrd2755
Konstas AGP, Quaranta L, Mikropoulos DG, et al., 2012. Peak intraocular pressure and glaucomatous progression in primary open-angle glaucoma. J Ocul Pharmacol Ther, 28(1):26–32. https://doi.org/10.1089/jop.2011.0081
Kroeger H, Messah C, Ahern K, et al., 2012. Induction of endoplasmic reticulum stress genes, BiP and Chop, in genetic and environmental models of retinal degeneration. Invest Ophthalmol Vis Sci, 53(12):7590–7599. https://doi.org/10.1167/iovs.12-10221
Kumar V, Mesentier-Louro LA, Oh AJ, et al., 2019. Increased ER stress after experimental ischemic optic neuropathy and improved RGC and oligodendrocyte survival after treatment with chemical chaperon. Invest Ophthalmol Vis Sci, 60(6):1953–1966. https://doi.org/10.1167/iovs.18-24890
Li J, Yang DY, Kwong JMK, et al., 2020. Long-term follow-up of optic neuropathy in chronic low cerebrospinal fluid pressure monkeys: The Beijing Intracranial and Intraocular Pressure (iCOP) study. Sci China Life Sci, 63(1762–1765. https://doi.org/10.1007/s11427-018-1626-6
Lin T, Lee JE, Kang JW, et al., 2019. Endoplasmic reticulum (ER) stress and unfolded protein response (UPR) in mammalian oocyte maturation and preimplantation embryo development. Int J Mol Sci, 20(2):409. https://doi.org/10.3390/ijms20020409
Liu JHK, Zhang XY, Kripke DF, et al., 2003. Twenty-four-hour intraocular pressure pattern associated with early glaucomatous changes. Invest Ophthalmol Vis Sci, 44(4): 1586–1590. https://doi.org/10.1167/iovs.02-0666
Liu JHK, Medeiros FA, Slight JR, et al., 2010. Diurnal and nocturnal effects of brimonidine monotherapy on intraocular pressure. Ophthalmology, 117(11):2075–2079. https://doi.org/10.1016/j.ophtha.2010.03.026
May CA, Mittag T, 2006. Optic nerve degeneration in the DBA/2NNia mouse: is the lamina cribrosa important in the development of glaucomatous optic neuropathy? Acta Neuropathol, 111(2):158–167. https://doi.org/10.1007/s00401-005-0011-2
Nork TM, ver Hoeve JN, Poulsen GL, et al., 2000. Swelling and loss of photoreceptors in chronic human and experimental glaucomas. Arch Ophthalmol, 118(2):235–245. https://doi.org/10.1001/archopht.118.2.235
Nouri-Mahdavi K, Hoffman D, Coleman AL, et al., 2004. Predictive factors for glaucomatous visual field progression in the advanced glaucoma intervention study. Ophthalmology, 111(9):1627–1635. https://doi.org/10.1016/j.ophtha.2004.02.017
Ortín-Martínez A, Salinas-Navarro M, Nadal-Nicolás FM, et al., 2015. Laser-induced ocular hypertension in adult rats does not affect non-RGC neurons in the ganglion cell layer but results in protracted severe loss of cone-photoreceptors. Exp Eye Res, 132:17–33. https://doi.org/10.1016/j.exer.2015.01.006
Panda S, Jonas JB, 1992. Decreased photoreceptor count in human eyes with secondary angle-closure glaucoma. Invest Ophthalmol Vis Sci, 33(8):2532–2536.
Salminen A, Kauppinen A, Hyttinen JMT, et al., 2010. Endoplasmic reticulum stress in age-related macular degeneration: trigger for neovascularization. Mol Med, 16(11–12): 535–542. https://doi.org/10.2119/molmed.2010.00070
Sim D, Fruttiger M, 2013. Keeping blood vessels out of sight. eLife, 2:e00948. https://doi.org/10.7554/eLife.00948
Sun H, Wang Y, Pang IH, et al., 2011. Protective effect of a JNK inhibitor against retinal ganglion cell loss induced by acute moderate ocular hypertension. Mol Vis, 17: 864–875.
Tham YC, Li X, Wong TY, et al., 2014. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology, 121(11):2081–2090. https://doi.org/10.1016/j.ophtha.2014.05.013
Wang JW, Valiente-Soriano FJ, Nadal-Nicolás FM, et al., 2017. MicroRNA regulation in an animal model of acute ocular hypertension. Acta Ophthalmol, 95(1):e10–e21. https://doi.org/10.1111/aos.13227
Weinreb RN, Aung T, Medeiros FA, 2014. The pathophysiology and treatment of glaucoma: a review. JAMA, 311(18): 1901–1911. https://doi.org/10.1001/jama.2014.3192
Xu SC, Gauthier AC, Liu J, 2016. The application of a contact lens sensor in detecting 24-hour intraocular pressure-related patterns. J Ophthalmol, 2016:4727423. https://doi.org/10.1155/2016/4727423
Xu LJ, Li SL, Zemon V, et al., 2020. Central visual function and inner retinal structure in primary open-angle glaucoma. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(4): 305–314. https://doi.org/10.1631/jzus.B1900506.
Yang L, Li SH, Miao LQ, et al., 2016. Rescue of glaucomatous neurodegeneration by differentially modulating neuronal endoplasmic reticulum stress molecules. J Neurosci, 36(21):5891–5903. https://doi.org/10.1523/jneurosci.3709-15.2016
Yang LP, Wu LM, Guo XJ, et al., 2007. Activation of endoplasmic reticulum stress in degenerating photoreceptors of the rd1 mouse. Invest Ophthalmol Vis Sci, 48(11):5191–5198. https://doi.org/10.1167/iovs.07-0512
Zeimer RC, Wilensky JT, Gieser DK, et al., 1991. Association between intraocular pressure peaks and progression of visual field loss. Ophthalmology, 98(1):64–69. https://doi.org/10.1016/s0161-6420(91)32340-6
Zhao ZN, Yu XW, Yang X, et al., 2020. Elevated intraocular pressure causes cellular and molecular retinal injuries, advocating a more moderate intraocular pressure setting during phacoemulsification surgery. Int Ophthalmol, 40(12): 3323–3336. https://doi.org/10.1007/s10792-020-01519-w
Zhou L, Chen W, Lin DY, et al., 2019. Neuronal apoptosis, axon damage and synapse loss occur synchronously in acute ocular hypertension. Exp Eye Res, 180:77–85. https://doi.org/10.1016/j.exer.2018.12.006
Acknowledgments
This work was supported by the Guangzhou Science and Technology Plan Project (Nos. 201803040020, 201903010065, and 202102021099), the Guangdong Natural Science Foundation (No. 2020A151501168), and the Research Funds of the State Key Laboratory of Ophthalmology (No. PT1001022), China.
Author information
Authors and Affiliations
Contributions
Xue YANG performed this study, collected the data, and drafted the manuscript. Zhenni ZHAO, Jiamin ZHANG, and Xiaoqian SU assisted in carrying out the research. Xiaowei YU and Yuqing HE assisted in analyzing the data. Nannan SUN and Zhigang FAN conceived the study and proofread the manuscript. All authors have read and approved the final manuscript and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Corresponding authors
Ethics declarations
Xue YANG, Xiaowei YU, Zhenni ZHAO, Yuqing HE, Jiamin ZHANG, Xiaoqian SU, Nannan SUN, and Zhigang FAN declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yang, X., Yu, X., Zhao, Z. et al. Endoplasmic reticulum stress is involved in retinal injury induced by repeated transient spikes of intraocular pressure. J. Zhejiang Univ. Sci. B 22, 746–756 (2021). https://doi.org/10.1631/jzus.B2100053
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2100053
Key words
- Endoplasmic reticulum (ER) stress
- Intraocular pressure spike (Spike IOP)
- Retinal injury
- Neuron apoptosis
- Glaucoma