Summary
Synopsis
Lansoprazole is an effective acid pump inhibitor acting at the final enzymatic step of the acid secretory pathway of the parietal cell, decreasing gastric acid secretion regardless of the primary stimulus.
Results of short term (<8 weeks) clinical trials have shown lansoprazole to be significantly superior to placebo and ranitidine in the treatment of duodenal ulcer, both in the rate of healing and in overall healing at 4 weeks. Lansoprazole appears to heal duodenal ulcer more quickly than famotidine, and demonstrates slightly greater efficacy at 4 weeks, although both drugs appear to have equivalent efficacy overall. Gastric ulcers and reflux oesophagitis are also healed by lansoprazole 30 mg/day for 4 to 8 weeks, with healing rates after 8 weeks of approximately 85 to 95% for both indications. Lansoprazole appears to be superior to ranitidine and comparable to omeprazole in treating reflux oesophagitis. Furthermore, lansoprazole has relieved reflux symptoms more quickly than either ranitidine or omeprazole. Preliminary data also indicate that lansoprazole may be effective in the treatment of peptic ulcer disease and reflux oesophagitis refractory to H2-receptor antagonists, and in patients with Zollinger-Ellison syndrome. While direct comparisons with omeprazole are limited, results suggest that lansoprazole, used for short term treatment, is at least as effective as omeprazole in the treatment of peptic ulcer and reflux oesphagitis.
Lansoprazole has been well tolerated in short term clinical trials, with an incidence of adverse effects comparable with that of other agents in its therapeutic class. Trials assessing long term tolerability data are ongoing and will be required as part of the assessment of the safety profile, if lansoprazole is to be used prophylactically to prevent ulcer recurrence.
Thus, by virtue of its ability to heal ulcers and rapidly relieve associated symptomatology, lansoprazole represents a useful alternative for the treatment of acid related disorders.
Pharmacodynamic Properties
Lansoprazole, a substituted benzimidazole derivative, is converted in the acidic environment of the canaliculi of the gastric parietal cell to active sulphenamide derivatives which bind to the sulphydryl group of H+,K+-ATPase, the enzyme catalysing the final step in the acid secretion pathway, thereby inactivating H+,K+-ATPase and producing inhibition of both centrally and peripherally mediated gastric acid secretion.
In in vitro studies lansoprazole appears to be at least as potent as omeprazole at inhibiting gastric acid secretion. In some in vivo animal models lansoprazole is less potent than the H2-receptor antagonists ranitidine and famotidine and as effective as omeprazole. In animal models lansoprazole inhibited gastric acid secretion to a greater extent when administered intravenously rather than orally and was effective regardless of the primary stimulus.
In humans, orally administered lansoprazole causes dose-proportional inhibition of acid secretion over the range of 15 to 60mg. Inhibition is approximately 80% after an initial 30mg dose and 90% after 7 days therapy with a once daily 30mg dose. Unlike H2-receptor antagonists, lansoprazole inhibits daytime and nocturnal acid secretion regardless of whether it is administered in the morning or the evening. Lansoprazole also decreases the volume of gastric acid secretion and inhibits the secretion and activity of pepsin.
Short term clinical studies indicate that lansoprazole 30 mg/day for 2 months causes an approximate doubling in serum gastrin levels in patients with peptic ulcer, but these return to baseline within several days to 4 weeks of discontinuing therapy. A longer term study of 12 months duration has demonstrated that lansoprazole 60mg daily also significantly increases serum gastrin levels. Increased serum gastrin levels occur as a result of gastric acid suppression, whether achieved pharmacologically or surgically, and as yet do not appear to be associated with any significant changes in gastric mucosal morphology in humans. In rats, life-long administration of other acid-suppressing drugs at high doses has resulted in the development of enterochromaffinlike cell carcinoids; however, this has not been reported with lansoprazole to date. The effect of lansoprazole on the gastric mucosa of humans, over periods of greater than 2 months, has not yet been reported.
Lansoprazole appears to prevent experimentally induced ulcers in animals, and in humans a 30 or 60mg daily dose appears to protect against aspirin-induced mucosal injury.
In vitro and preliminary clinical data indicate that lansoprazole may clear Helicobacter pylori, an organism which has been associated with relapse of peptic ulcer, from the gastric mucosa. However, these preliminary findings need confirmation, in particular, whether initial eradication of H. pylori persists after treatment with lansoprazole is completed.
Pharmacokinetic Properties
Because of its instability in acidic conditions, lansoprazole is administered as an enteric-coated formulation. Maximum serum lansoprazole concentrations of 1038 μg/L have been reached within approximately 2 hours of administration of a 30mg encapsulated enteric-coated dose. Concomitant food intake delays absorption and has reduced bioavailability in some studies. Bioavailability varies between individuals but appears to be dose-proportional. Most studies have reported no significant increase in bioavailability after repeated administration of lansoprazole over a 7-day period.
While lansoprazole is converted to the active compounds AG-1812 and AG-2000 in the acidic environment of the parietal cell, in the serum lansoprazole is rapidly and completely metabolised to 2 main excretory metabolites, lansoprazole sulphone and hydroxylansoprazole. Approximately 14 to 23% of a dose is excreted in the urine as conjugated and unconjugated hydroxylated metabolites but unchanged lansoprazole has not been detected in the urine. The mean elimination half-life of lansoprazole is between 1.3 and 1.7 hours in healthy volunteers after a single dose, is about 2 hours in the elderly and in patients with severe hepatic dysfunction is prolonged to 7 hours. Serum lansoprazole concentrations do not correlate with antisecretory activity and indeed, antisecretory activity is apparent for many hours after the drug is undetectable in the serum.
Therapeutic Efficacy
Lansoprazole 30 mg/day heals duodenal ulcers after 2 to 4 weeks, with healing rates of 75 to 100% after 4 weeks. Most studies have found that lansoprazole 30mg once daily provides increased healing rates and more rapid healing than ranitidine: healing rates at 2 weeks were about 75 to 80% with lansoprazole and 45 to 60% with ranitidine, and about 93 to 95% and 80%, respectively, after 4 weeks. A typical study shows that lansoprazole heals more rapidly than famotidine (healing rate at 2 weeks of 52 vs 37%) and is slightly more effective after 4 weeks (healing rates of 89 vs 80%). Lansoprazole also appears to heal more rapidly than orneprazole (healing rate at 2 weeks of 74 vs 58%), but both drugs were equally effective after 4 weeks. Ulcer-related pain is reduced rapidly by lansoprazole 30 and 60 mg/day; in one study lansoprazole 30 mg/day relieved pain more quickly than ranitidine 300 mg/day, producing complete pain relief after a median of 3 days vs 7 for ranitidine. A 60mg daily dose may be superior to ranitidine in relieving epigastric pain; relative effects with lower doses have been equivocal. Some investigators have reported that lansoprazole 30mg daily has a tendency to relieve more patients of ulcer symptomatology than famotidine 40 mg/day after 1 week of treatment, but others report that both drugs were similarly effective.
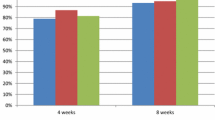
Patients with gastric ulcer require longer term treatment and are generally treated with lansoprazole 30 mg/day for 4 to 8 weeks. Healing rates achieved with lansoprazole 30mg daily after 4 and 8 weeks were 50 to 79% and 87 to 99%, respectively. Lansoprazole 30 mg/day healed gastric ulcer more effectively than ranitidine 300 mg/day at 4 weeks (healing rates of 73 to 78% vs 56 to 61%) and after 8 weeks provided slightly greater healing (99 vs 91%). Lansoprazole 30mg daily was more effective at healing gastric ulcer than famotidine 20mg twice daily after 2, 4 and 8 weeks and reduced symptoms more rapidly, but both drugs provided similar symptomatic relief after 8 weeks. Lansoprazole 30 mg/day was more effective than omeprazole 20mg daily in healing gastric ulcer after 8 weeks (healing rates of 93 vs 82%).
The efficacy of lansoprazole as maintenance therapy for preventing relapse of peptic ulcer has not yet been reported, although studies are in progress. Given the high relapse rate of peptic ulcer when any acid-suppressing therapy, including lansoprazole, is withdrawn, clinical trials to investigate the potential of lansoprazole in this indication are warranted.
Reflux oesophagitis is effectively treated with lansoprazole 30 mg/day with healing rates of 63 to 84% after 4 weeks and 85% to 92% after 8 weeks. Lansoprazole appears to be superior to ranitidine and equivalent to omeprazole in healing patients with this indication. Lansoprazole has relieved symptoms of heartburn significantly better than ranitidine and omeprazole after 4 weeks of treatment.
Preliminary data indicate that lansoprazole is effective in patients with peptic ulcers or reflux oesophagitis refractory to H2-receptor antagonists: at doses of 30 mg/day administered for 8 weeks healing rates of 69 to 100% were achieved. Further healing may result if the dose is increased to 60 mg/day from week 8. Lansoprazole 60 to 120 mg/day (administered once or twice daily) reduced mean gastric acid output in patients with Zollinger-Ellison syndrome to <10 mmol/h in the hour preceding the next dose, and there is some preliminary indirect evidence that lansoprazole may be as effective as omeprazole in controlling acid secretion in patients with this disease.
Tolerability
Lansoprazole is well tolerated by patients in short term (< 8 weeks) clinical trials but there are no published reports of its longer term tolerability. Diarrhoea, headache, nausea and dermatological reactions are the most frequently reported adverse effects, occurring in < 2% of patients, but these are usually mild and transient. Occasional abnormalities in liver function tests and in complete blood counts have also been reported but these have generally been clinically insignificant. Only 9 patients (from a total of about 2100 patients) in clinical studies have discontinued therapy as a result of adverse effects. The incidence of adverse effects due to lansoprazole was not different from that reported in placebo recipients, and in comparative studies was generally similar to the incidence observed in patients treated with histamine H2-receptor antagonists or omeprazole.
Dosage and Administration
A daily dose of lansoprazole 30mg, administered in the morning or the evening, is recommended for the treatment of duodenal ulcer and reflux oesophagitis. Similar regimens have been administered to patients with gastric ulcer or refractory disease. Lansoprazole therapy is usually required for 2 to 4 weeks for the treatment of duodenal ulcer and for 4 to 8 weeks for the treatment of gastic ulcer or reflux oesophagitis. Doses of 60 mg/day have been administered if ulcers are not healed after 6 to 8 weeks. In patients with Zollinger-Ellison syndrome the dose should be individualised to reduce and maintain the basal acid output between 0 and 10 mmol/h, and doses of up to 120 mg/day have been given for this indication.
Similar content being viewed by others
References
Aaronson R, Dorsch E, Padgett C, Jennings D, Greski P, et al. Lansoprazole heals duodenal ulcers. Presented at American College of Gastroenterology Meeting, Boston, October 14–16, 1991
Abe t, Sato S, Suzuki T, Miyajima H. Thirteen-week oral toxicity study of lansoprazole (AG-1749) in rats. Yakuri to Chiryo 18 (Suppl. 10): S2663–S2678, 1990
Andersen DK. Current diagnosis and management of Zollinger-Ellison syndrome. Annals of Surgery 210(6): 685–703, 1989
Andersson T. Omeprazole drug interaction studies. Clinical Pharmacokinetics 21(3): 195–212, 1991
Aoki I, Okumura M, Yashiki T. High-performance liquid Chromatographic determination of lansoprazole and its metabolites in human serum and urine. Journal of Chromatography 571: 283–290, 1991
Arakawa T, Higuchi K, Fukuda T, Nakamura H, Kobayashi K. H2 -receptor antagonist-refractory ulcer: its pathophysiology and treatment. Journal of Clinical Gastroenterology 13 (Suppl. 1): 129–133,1991
Asaka M, Saito M, Takeda H, Murashima Y, Suga T, et-al. Clinical evaluation of lansoprazole on H2 -blocker resistant peptic ulcer. Yakuri to Chiryo 19: 953–966, 1991
Atkinson JE, Daly TW, Bolte HF, Morishima H, Sasaki S. One-year oral gavage toxicity study of lansoprazole (AG-1749) in rats. Yakuri to Chiryo 18 (Suppl. 10): S2713–S2745, 1990a
Atkinson JE, Daly IW, Wooding WL, Morishima H, Sasaki S. One-year oral toxicity study of lansoprazole (AG-1749) in beagle dogs. Yakuri to Chiryo 18 (Suppl. 10): S2747–S2772, 1990b
Bardhan KD, Long R, Hawkey CJ, Wormsley KG, Brocklebank D, et al. Lansoprazole, a new proton-pump inhibitor, vs ranitidine in the treatment of reflux erosive esophagitis. Abstract. Gastroenterology 100: A30, 1991
Benhaim MC, Evreux M, Salducci J, Petite JP, Lemaire M. Lansoprazole and ranitidine in treatment of reflux oesophagitis: double blind comparative trial. Abstract. Gastroenterology 98: A20; 1990
Bigard MA, Joubert M, de Meynard C. Complete prevention by lansoprazole of aspirin induced gastric lesions in healthy subjects. Abstract. Gastroenterology 100: A34, 1991
Cavanaugh JH, Winters EP, Cohen A, Locke CS, Braeckman R. Lack of effect of lansoprazole on steady state warfarin metabolism. Abstract. Gastoenterology 110(5): A40, 1991
Clissold SP, Campoli-Richards DM. Omeprazole: a preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in peptic ulcer disease and Zollinger-Ellison syndrome. Drugs 32: 15–47, 1986
Creutzfeldt W, Lamberts R. Is hypergastrinaemia dangerous to man? Scandinavian Journal of Gastroenterology 26 (Suppl. 180): 179–191, 1991
Daneshmend TK, Sharma HK, Bhaskar NK, Hawthorne AB, Hawkey CJ. Acid inhibition and mucosal protection with a new proton pump inhibitor. Abstract. Gut 30(10): A1483, 1989
Delhotal-Landes B, Cournot A, Vermerie N, Dellatolas F, Benoit M, et al. The effect of food and antacids on lansoprazole absorption and dispostion. European Journal of Drug Metabolism and Pharmacokinetics (Special Issue No. III): 315-320, 1991
Delhotal B, Flouvat B, Cournot A, Duchier J, Dellatolas F. Pharmacocinétique d’un nouvel inhibiteur de la pompe à protons gastrique. Le lansoprazole chez les sujets à risque. In Brès & Panis (Eds) Pharmacocinétique: de la recherche à la clinque. pp. 242–247, John Libbey Eurotext, Paris, 1992
Delhotal Landes B, Miscoria G, Flouvat B. Determination of lansoprazole and its metabolites in plasma by high performance liquid chromatography using a loop column. Journal of Chromatography 577: 117–122; 1992
Dorsch E, Jones J, Padgett C, Jennings D, Greski P, et al. Lansoprazole heals moderate to severe reflux esophagitis. Presented at American College of Gastroenterology Meeting, Boston, October 14–16, 1991
Florent C, Forestier S, Joubert-Coullin M. Lansoprazole versus omeprazole: efficacy and safety in acute gastric ulcer. Abstract. Submitted for presentation at European Gastroenterology week, Athens, Sept. 25–30, 1992
Flouvat B, Delhotal-Landes B, Cournot A, Dellatolas F, Calle-bout B. Pharmacokinetic study of lansoprazole after oral administration to elderly subjects, comparison with young subjects. Poster. Fourth European Congress of Biopharmaceutics and Pharmacokinetics. Genève, April, 1990
Granneman G, Winters EP, Locke CS, Leese PT, Karol MD, et al. Lack of effect of concomitant lansoprazole on steady-state theophylline pharmacokinetics. Abstract. Gastroenterology. 100(5): A75, 1991
Grant SM, Langtry HD, Brogden RN. Ranitidine. An updated review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in peptic ulcer disease and other allied diseases. Drugs 37: 801–870, 1989
Hamamuki S, Tani N, Miwa T. Clinicopharmacological study of lansoprazole on gastric acid secretion: comparison between morning and evening dosage by 24-hour intragastric Ph monitoring. Yakuri to Chiryo 19: 925–931, 1991
Harasawa S, Miwa T. The effect of AG-1749 (lansoprazole) on gastric emptying and serum gastrin concentration in peptic ulcer patients. Yakuri to Chiryo 19: 947–952, 1991
Hatlebakk JG, Berstad A, Carling L, Svedberg L-E, Unge P, et al. Lansoprazole vs omeprazole in short-term treatment of reflux esophagitis- Results of a Scandinavian Multicentre Trial. Abstract. American Gastroenterological Association May 10–13, 1992
Hawkey CJ, Bardhan KD, Long RG, Wormsley KG, Cochran RM, et al. Improved symptom relief and duodenal ulcer healing with lansoprazole compared to ranitidine. Abstract. Gastroenterology 100: A80, 1991
Hochlaf S, Vatier J, Ruszniewski Ph, Poitevin Ch, Lewin MJM, et al. Is lansoprazole as effective as omeprazole in patients with Zollinger-Ellison syndrome? Abstract. Gastroenterology 100: A84, 1991
Hogan DL, Koss MA, Feitelberg S, Hussein Z, Karol MD, et al. Single and repetitive administration of lansoprazole: effects on gastric acid secretion, pharmacokinetics and serum gastrin in old vs young subjects. Abstract. Gastroenterology 100: A84, 1991
Holloway RH, Dent J. Pathophysiology of gastroesophageal reflux. Lower esophageal sphincter dysfunction in gastroesophageal reflux disease. Gastroenterology Clinics of North America 19(3): 517–535, 1990
Holt S. Proton-pump inhibition for acid-related disease. Southern Medical Journal 84(9): 1078–1087, 1991
Hotz J, Kleiner R, Grymbowski T, Hennig U, Schwarz JA. Lansoprazole versus famotidine: efficacy andℴlerance in the acute management of duodenal ulceration. Alimentary Pharmacology and Therapeutics 6: 87–95, 1992
Im WB, Blakeman DP, Davis JP. Irreversible inactivation of rat gastric (H+ -K+ )-ATPase in vivo by omeprazole. Biochemical and Biophysical Research Communications 126.(1): 78–82, 1985
Inatomi N, Nagaya H, Takami K, Shino A, Satoh H. Effects of a proton pump inhibitor, AG-1749 (lansoprazole), on reflux esophagitis and experimental ulcers in rats. Japanese Journal of Pharmacology 55: 437–451, 1991
Iwahi T, Satoh H, Nakao M, Iwasaki T, Yamazaki T, et al. Lansoprazole, a novel benzimidazole proton pump inhibitor, and its related compounds have selective activity against Helicobacter pylori Antimicrobial Agents and Chemotherapy 35(3): 490–496, 1991
Jhala NC, McFarland M, Brightman S, Morale B, Jennings D, et al. Effect of short term treatment with lansoprazole on H. Pylori and antral gastritis in patients with duodenal ulcers. Abstract 675. Meeting of American Gastrenterological Association, San Francisco, May 9–14, 1992
Kodama T, Fuse Y, Ohkawawa Y, Maruyam Y, Tsuji H. Clinical effect of lansoprazole (AG-1749) on patients with gastric and duodenal ulcer. Yakuri to Chiryo 18: 4891–4900, 1990
Licht H, Andrieu J, Bognel JC, Bouvry M, Delas N, et al. Lansoprazole versus ranitidine dans le traitment des ulcers duodenaux: resultats d’un essai multicentrique controle, randomise, en double insu sur groupes paralleles. Medecine et Chirurgie Digestives 19(4): 251–255, 1990
Londong W, Barth H, Dammann HG, Hengels KJ, Kleinen R, et al. Dose-related healing of duodenal ulcer with the proton pump inhibitor lansoprazole. Alimentary Pharmacology and Therapeutics 5: 245–254, 1991
Londong W, Hotz J, Kleinert R, Rehner M, Schwarz JA. Dose finding study in gastric ulcer with lansopraozle, a new proton pump inhibitor. Abstract PP 670. The World Congresses of Gastroenterology, Sydney, Australia, August 26-31, 1990
McTavish D, Buckley MM-T, Heel RC. Omeprazole: an updated review of its pharmacology and therapeutic use in acid-related disorders. Drugs 42(1): 138–170, 1991
Makiyama K, Umene Y, Murata I, Izuno M, Hara K. Clinical study on therapeutic effect of an antiulcer agent, lansoprazole, on gastric ulcer: with special reference to serum gastrin concentration. Yakuri to Chiryo 19(1): 307–325, 1991
Marshall BJ. Campylobacter pylori: its link to gastritis and peptic ulcer disease. Reviews of Infectious Diseases 12 (Suppl. 1): S87–S93, 1990
Maton PN. Omeprazole. New England Journal of Medicine 324(14): 965–975, 1991
Matsuo Y, Takemoto T, Miwa T, Iwasaki A, Asaoka A. Clinical study of the inhibitory effect of AG-1749, a proton pump inhibitor, on nocturnal gastric acid secretion: evaluation of dosage and administration stage. Yakuri to Chiryo 18: 4865–4876, 1990
Megraud F, Boyanova L, Lamouliatte H. Activity of lansoprazole against Helicobacter pylori Letter. Lancet 337: 1486, 1991
Metz DC, Pisegna JR, Ringham GL, Fishbeyn VA, Benya RV, Gardner JD, et al. Efficacy and safety of lansoprazole in patients with Zollinger-Ellison syndrome (ZES). Abstract 1382. Meeting of American Gastroenterological Association, San Francisco, May 9–14, p. A346, 1992
Misawa T, Chijiiwa Y, Imazono Y, Fujishima H, Nawada H. Effects of lansoprazole, a gastric proton pump inhibitor on endocrine function and healing in patients with peptic ulcer. Therapeutic Research 12(1): 175–189, 1991
Mitani M, Tsukamoto T, Yoshida S, Kobayashi T. Metabolic fate of AG-1749, a new proton pump inhibitor, in rats, mice, and dogs. Japanese Pharmacology and Therapeutics 18(9): 3413–3435, 1990
Morise K, Oka Y, Suzuki T, Kusugami K, Inagaki T. Clinical evaluation of lansoprazole (AG-1749) in patients with gastric or duodenal ulcer. Yakuri to Chiryo 19: 327–338, 1991
Müller P, Dammann HG, Leucht U, Simon B. Human gastric acid secretion following repeated doses of AG-1749. Alimentary Pharmacology and Therapeutics 3: 193–198, 1989
Nagaya H, Satoh H, Kubo K, Maki Y. Possible mechanism for the inhibition of gastric (H+ + K+ )-adenosine triphosphatase by the proton pump inhibitor AG-1749. Journal of Pharmacology and Experimental Therapeutics 248(2): 799–805, 1989
Nagaya H, Satoh H, Maki Y. Possible mechanism for the inhibition of acid formation by the proton pump inhibitor AG-1749 in isolated canine parietal cells. Journal of Pharmacology and Experimental Therapeutics 252: 1289–1295, 1990
Nagaya H, Inatomi N, Nohara A, Satoh H. Effects of the enantiomers of lansoprazole (AG-1749) on (H+ + K+ )-ATPase activity in canine gastric microsomes and acid formation in isolated canine parietal cells. Biochemical Pharmacology, 42(10): 1875–1878, 1991a
Nagaya H, Inatomi N, Satoh H. Differences in the antisecretory actions of the proton pump inhibitor AG-1749 (lansoprazole) and the histamine H2 - receptor antagonist famotidine in rats and dogs. Japanese Journal of Pharmacology 55: 425–436, 1991b
Nakagawa A, Ooka T, Kim H, Sato N, Kamada T, et al. Phase I study of lansoprazole (AG-1749), antiulcer agent. Tablet form. Rinsho Iyaku 7: 33–50, 1991
Ogoshi K, Kato T, Saito S, Niwa M, Watanabe H, et al. Clinical study of AG-1749 (lansoprazole): effects on serum gastrin levels and gastric mucosal ECL cell density, etc. Yakuri to Chiryo 19(3): 933–946, 1991
Pace F, Bianchi Porro G. Medical treatment of reflux oesophagitis: review of traditional therapies and omeprazole. Italian Journal of Gastroenterology 20 (Suppl.): 23–29, 1988
Petite JP, Grimaud JC, Rautureau J, Lemaire M. Lansoprazole versus omeprazole dans la traitement de l’ulcère duodenal evolutif. Abstract. Gastroenterologie Clinique et Biologique 15: p. A111, 1991
Ponce J, Rodrigo JM. Therapeutic failure and relapse in peptic ulcer. Methods and Findings in Experimental and Clinical Pharmacology 11 (Suppl. 1): 123–130, 1989
Rauws EAJ, Langenberg W, Houthoff HJ, Zanen HC, Tytgat GNJ. Campylobacter pyloridis-associated chronic active antral gastritis. A prospective study of its prevalence and the effects of antibacterial and antiulcer treatment. Gastroenterology 94: 33–40, 1988
Rauws EAJ, Tytgat GNJ. Cure of duodenal ulcer associated with eradication of Helicobacter pylori Lancet 335: 1233–1235, 1990
Robinson MG, Campbell DR, Sontag S, Sabesin SM, Greski PA, et al. Lansoprazole heals H2 resistant erosive reflux esophagitis. Abstract. Gastroenterology 98: Al 13, 1990
Robinson M, Kogut D, Jennings D, Levy C, Greski-Rose P, et al. Lansoprazole heals erosive reflux esophagitis better than ranitidine. Abstract 2178. Meeting of American Gastroentrological Association, San Francisco, May 9–14, 1992
Sampliner RE, Mackel C, Jennings D, Greski-Rose P. Effect of 12 months of a proton pump inhibitor (lansoprazole) on Barrett’s esophagus — a randomised trial. Abstract 2199. Meeting of American Gastroentrological Association San Francisco, May 9-14, 1992
Satoh H, Inatomi N, Nagaya H, Inada I, Nohara A, et al. Anti-secretory and antiulcer activities of a novel proton pump inhibitor AG-1749 in dogs and rats. Journal of Pharmacology and Experimental Therapeutics 248(2): 806–815, 1989
Sekiguchi T, Matsuzaki T, Horikoshi T, Nishioka T, Kusano M, et al. Clinical effects of lansoprazole in the treatment of reflux esophagitis. Therapeutic Research 12(1): 191–213, 1991
Sontag S, Kurucar C, Murray S, Greski-Rose P, Jennings D, et al. Lansoprazole heals erosive reflux esophagitis resistant to histamine H2 -recptor antagonist therapy. Abstract 2198. Meeting of American Gastroenterological Association San Francisco, May 9–14, 1992
Sugiyama M, Ishikawa T, Aoki T, Kashiwagi H, Watanabe Y, et al. Effect of lansoprazole (AG-1749) on gastric acid secretion — inhibition of insulin-stimulated acid secretion. Shokakika 14(2): 183–193, 1991
Tada M, Murakami A, Karita M, Shiraishi H, Yanai H, et al. A study of gastric juice secretion-inhibitory effect of a proton pump inhibitor, AG-1749. Rynsho-Seijinbyo 21: 633–640, 1991
Takemoto T, Namiki M, Goto Y, Matsuo Y, Miwa T, et al. A study of clinical usefulness of lansoprazole (AG-1749) in treating duodenal ulcer: a comparison with famotidine by the multiclinic, double-blind, controlled technique. Rinshoseijinbyo 21(3): 613–631, 1991a
Takemoto T, Namiki M, Goto Y, Matsuo Y, Miwa T. A study of clinical usefulness of lansoprazole (AG-1749) in treating gastric ulcer: a comparison with famotidine by multiclinic, double-blind technique. Rinshoseijinbyo 21(2): 327–345, 1991b
Takemoto T, Namiki M, Goto Y, Matsuo Y, Miwa T, et al. Studies of clinical usefulness of lansoprazole (AG-1749) in the treatment of gastric and duodenal ulcers. Comparison of lansoprazole at 2 dosages with famotidine. Clinical Adult Diseases 21(4): 743–766, 1991c
Takemoto T, Namiki M, Goto Y, Matsuo Y, Miwa T, et al. Studies of clinical usefulness of lansoprazole (AG-1749) in the treatment of gastric and duodenal ulcers. A study of usefulness of its dosages and administration time. Clinical Adult Diseases 21(5): 995–1013, 1991d
Takemoto T, Namiki M, Goto Y, Matsuo Y, Miwa T, et al. Studies of clinical usefulness of lansoprazole (AG-1749) in the treatment of gastric and duodenal ulcers. A study of usefulness of AG-1749 at a dosage of 30 mg daily, using famotidine as the control drug. Clinical Adult Diseases 21(5): 975–993, 199le
Takemoto T, Okazaki Y, Tada M, Namiki M, Okamura K, et al. Studies of clinical usefulness of lansoprazole (AG-1749) in the treatment of gastric and duodenal ulcers. A clinical early phase II study. Clinical Adult Diseases 21: 769–783, 1991f
Tamura K, Yamamoto A, Shinoyama N, Tanaka M. Clinical effect of AG-1749 on gastric ulcer: histological change of gastric mucosa and effect on Helicobacter pylori Yakuri to Chiryo 18: 4877–4890, 1990
Tateno M, Nakamura N. Phase I study of lansoprazole (AG-1749) antiulcer agent. Capsule form. Rinsho Iyaku 7: 51–62, 1991
Walsh JH. Role of gastrin as a trophic hormone. Digestion 47 (Suppl. 1): 11–16, 1990
Wolfe MM, Jensen RT. Zollinger-Ellison syndrome. Current concepts in diagnosis and management. New England Journal of Medicine 317(19): 1200–1209, 1987
Wormsley KG, Bardhan KD, Morgan AG, Hislop W, Long R, et al. Lansoprazole (LAN) is more effective than ranitidine (RAN) in the healing of gastric ulcer. Abstract T190. Gut 33 (Suppl. 1): 548, 1992
Yasutake K, Yoshimura Y, Oya M, Nishisaki H, Masuta S, et al. Efficacy of lansoprazole in the treatment of peptic ulcer refractory to H2 receptor antagonists. Shokakika 13: 602–610, 1990
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: K.D. Bardhan, Rotherham District General Hospital Rotherham England; A. Berstad, Section of Gastroenterology, Medical Department, Haukeland Hospital, Bergen, Norway G. Bianchi Porro, Gastrointestinal Unit, L. Sacco Hospital, Milan, Italy; W. Creutzfeldt, Department of Gastroenterology and Endocrinology, Department of Medicine, George-August Universtiy, Gottingen, Federal Republic ot Germany; T.K. Daneshmend, Royal Devon and Exeter Hospital, Exeter, England; B. Delhotal Landes Laboratoire de Toxicologie et de Pharmacocinetique, Hôpitaux de Paris, Paris, France; S. Hamamuki, School of Medicine, Tokai University, Kanagawa, Japan; D.L. Hogan, Division of Gastroenterology, University of California San Diego Medical Center, San Diego, California, USA; R.H. Hunt, Division of Gastroenterology, Department of Medicine, McMaster University, Hamilton, Ontario, Canada; W. Londong, Second Medical Department Krankenhaus am Urban, Berlin, Federal Republic of Germany; K. Makiyama, Second Department of Internal Medicine Nagasaki University School of Medicine, Nagasaki, Japan; R. E. Pounder, Royal Free Hospital, London England; T. Takemoto, Yamaguchi Rohsai Hospital, Onoda, Japan; M.M. Wolfe, Gastroenterology Division’ Harvard Medical School, Boston, Massachusetts, USA.
Rights and permissions
About this article
Cite this article
Barradell, L.B., Faulds, D. & McTavish, D. Lansoprazole. Drugs 44, 225–250 (1992). https://doi.org/10.2165/00003495-199244020-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199244020-00007