Abstract
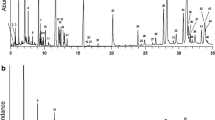
Gonipterus weevils have been a taxonomic challenge for many years, with implications on our understanding of invasive species, host plant relationships and natural enemies. We assessed cuticular hydrocarbon (CHC) analysis as a tool for discrimination of some of the many species of Gonipterus occurring in Australia. Weevils were collected across several localities and kept under identical conditions prior to a whole-body wash for extraction of CHCs in hexane. Weevil identifications were confirmed using morphology and molecular tools. CHC extracts were analyzed by gas chromatography–mass spectrometry (GC–MS) and the relative peak areas in profiles were compared; compounds were identified according to MS fragmentation and retention indices. CHC profiles of the seven species of Gonipterus analyzed differed from each other, and from another weevil genus (Oxyops), used as an outgroup. The compounds that contributed most to species differences were alkanes, alkenes and methyl branched alkanes. Within some species, locality of collection affected CHC profiles. Our study presents CHC analysis as a promising tool for distinction of Gonipterus species.


Similar content being viewed by others
Availability of data and material
Relevant data available as Online Resources.
References
Arzone A, Meotto F (1978) Reperti biologici su Gonipterus scutellatus Gyll. (Col. Curculionidae) infestante gli eucalipti della Riviera Ligure Redia 61:205–222
Barratt BI, Cock MJ, Oberprieler RG (2018) Weevils as targets for biological control, and the importance of taxonomy and phylogeny for efficacy and biosafety. Diversity 10:73. https://doi.org/10.3390/d10030073
Bernhardt V, Pogoda W, Verhoff MA, Toennes SW, Amendt J (2017) Estimating the age of the adult stages of the blow flies Lucilia sericata and Calliphora vicina (Diptera: Calliphoridae) by means of the cuticular hydrocarbon n-pentacosane. Sci Justi 57:361–365. https://doi.org/10.1016/j.scijus.2017.04.007
Blomquist GJ, Bagnères AG (2010) Introduction: history and overview of insect hydrocarbons. In: Bagnères AG, Blomquist GJ (eds) Insect Hydrocarbons: Biology, Biochemistry, and Chemical Ecology. Cambridge University Press, Cambridge, pp 3–18. https://doi.org/10.1017/CBO9780511711909.002
Buczkowski G, Kumar R, Suib SL, Silverman J (2005) Diet-related modification of cuticular hydrocarbon profiles of the Argentine ant Linepithema humile, diminishes intercolony aggression. J Chem Ecol 31:829–843. https://doi.org/10.1007/s10886-005-3547-7
Buellesbach J, Whyte BA, Cash E, Gibson JD, Scheckel KJ, Sandidge R, Tsutsui ND (2018) Desiccation resistance and micro-climate adaptation: cuticular hydrocarbon signatures of different argentine ant supercolonies across California. J Chem Ecol 44:1101–1114. https://doi.org/10.1007/s10886-018-1029-y
Calderon-Fernandez GM, Girotti JR, Juarez MP (2011) Cuticular hydrocarbons of Triatoma dimidiata (Hemiptera: Reduviidae): intraspecific variation and chemotaxonomy. J Med Entomol 48:262–271. https://doi.org/10.1603/me10141
Calderon-Fernandez GM, Girotti JR, Juarez MP (2012) Cuticular hydrocarbon pattern as a chemotaxonomy marker to assess intraspecific variability in Triatoma infestans, a major vector of Chagas’ disease. Med Veter Entomol 26:201–209. https://doi.org/10.1111/j.1365-2915.2011.00978.x
Chen Y, Dallara PL, Nelson LJ, Coleman TW, Hishinuma SM, Carrillo D, Seybold SJ (2017b) Comparative morphometric and chemical analyses of phenotypes of two invasive ambrosia beetles (Euwallacea spp.) in the United States. Insect Science 24:647–662. https://doi.org/10.1111/1744-7917.12329
Chen N, Bai Y, Fan YL, Liu TX (2017a) Solid-phase microextraction-based cuticular hydrocarbon profiling for intraspecific delimitation in Acyrthosiphon pisum. PLoS ONE. https://doi.org/10.1371/journal.pone.0184243
Chung H, Carroll SB (2015) Wax, sex and the origin of species: Dual roles of insect cuticular hydrocarbons in adaptation and mating. BioEssays 37:822–830. https://doi.org/10.1002/bies.201500014
Chung H, Loehlin DW, Dufour HD, Vaccarro K, Millar JG, Carroll SB (2014) A single gene affects both ecological divergence and mate choice in Drosophila. Science 343:1148–1151. https://doi.org/10.1126/science.1249998
Clarke KR (1993) Non-parametric multivariate analysis of changes in community structure. Aust J Ecol 18:117–143
Clarke KR, Gorley RN, Somerfield PJ, Warwick RM (2014) Change in marine communities: an approach to statistical analysis and interpretation, 3rd edn. PRIMER-E, Plymouth
El-Sayed AM (2020) Pherobase https://www.pherobase.com/ Accessed 23 Nov 2020
Everaerts C, Farine JP, Cobb M, Ferveur JF (2010) Drosophila cuticular hydrocarbons revisited: mating status alters cuticular profiles. PLoS ONE. https://doi.org/10.1371/journal.pone.0009607
Garcia A et al (2019) Biological control of Gonipterus: Uncovering the associations between eucalypts, weevils and parasitoids in their native range. For Ecol Manage 443:106–116. https://doi.org/10.1016/j.foreco.2019.04.004
Geiselhardt S, Otte T, Hilker M (2012) Looking for a similar partner: host plants shape mating preferences of herbivorous insects by altering their contact pheromones. Ecol Lett 15:971–977. https://doi.org/10.1111/j.1461-0248.2012.01816.x
Gemeno C, Laserna N, Riba M, Valls J, Castane C, Alomar O (2012) Cuticular hydrocarbons discriminate cryptic Macrolophus species (Hemiptera: Miridae). Bull Entomol Res 102:624–631. https://doi.org/10.1017/s0007485312000193
Ginzel MD, Blomquist GJ, Millar JG, Hanks LM (2003) Role of contact pheromones in mate recognition in Xylotrechus colonus. J Chem Ecol 29:533–545. https://doi.org/10.1023/a:1022894419521
Guillem RM, Drijfhout FP, Martin SJ (2012) Using chemo-taxonomy of host ants to help conserve the large blue butterfly. Biol Cons 148:39–43. https://doi.org/10.1016/j.biocon.2012.01.066
Guillem RM, Drijfhout FP, Martin SJ (2016) Species-specific cuticular hydrocarbon stability within European Myrmica Ants. J Chem Ecol 42:1052–1062. https://doi.org/10.1007/s10886-016-0784-x
Hanks LM, Millar JG, Paine TD, Campbell CD (2000) Classical biological control of the Australian weevil Gonipterus scutellatus (Coleoptera : Curculionidae) in California. Environ Entomol 29:369–375. https://doi.org/10.1603/0046-225x(2000)029[0369:cbcota]2.0.co;2
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Ann Rev Entomol 50:371–393. https://doi.org/10.1146/annurev.ento.50.071803.130359
ICA (2016) Reporte de Gonipterus platensis (Marelli, 1926) en el departamento de Antioquia. Instituto Colombiano Agropecuario. https://www.ica.gov.co/Alertas-Fitosanitarias/Notificacion-Oficial.aspx Accessed 28 Feb 2017
Kather R, Martin SJ (2015) Evolution of cuticular hydrocarbons in the hymenoptera: a meta-analysis. J Chem Ecol 41:871–883. https://doi.org/10.1007/s10886-015-0631-5
Kelstrup HC, Hartfelder K, Nascimento FS, Riddiford LM (2014) Reproductive status, endocrine physiology and chemical signaling in the Neotropical, swarm-founding eusocial wasp Polybia micans. J Experim Biol 217:2399–2410. https://doi.org/10.1242/jeb.096750
Kühbandner S, Sperling S, Mori K, Ruther J (2012) Deciphering the signature of cuticular lipids with contact sex pheromone function in a parasitic wasp. J Exp Biol 215:2471–2478
Lea AM (1927) Australian Curculionidae of the subfamily Gonipterides. Proc Roy Soc Victoria (New Series) 39:76–112
Lelito JP, Boroczky K, Jones TH, Fraser I, Mastro VC, Tumlinson JH, Baker TC (2009) Behavioral evidence for a contact sex pheromone component of the Emerald Ash Borer Agrilus planipennis Fairmaire. J Chem Ecol 35:104–110. https://doi.org/10.1007/s10886-008-9583-3
Liang D, Silverman J (2000) “You are what you eat”: Diet modifies cuticular hydrocarbons and nestmate recognition in the Argentine ant Linepithema humile. Naturwissenschaften 87:412–416. https://doi.org/10.1007/s001140050752
Liebig J, Eliyahu D, Brent CS (2009) Cuticular hydrocarbon profiles indicate reproductive status in the termite Zootermopsis nevadensis. Behav Ecol Sociobiol 63:1799–1807. https://doi.org/10.1007/s00265-009-0807-5
Mapondera TS, Burgess T, Matsuki M, Oberprieler RG (2012) Identification and molecular phylogenetics of the cryptic species of the Gonipterus scutellatus complex (Coleoptera: Curculionidae: Gonipterini). Aust J Entomol 51:175–188. https://doi.org/10.1111/j.1440-6055.2011.00853.x
Marelli CA (1928) Estudio sobre una peste de los eucaliptos descubierta en la Argentina Memorias del Jardin Zoológico de La Plata (Rep Argentina) 3:51–183
Michelutti KB, Soares ERP, Sguarizi-Antonio D, Piva RC, Suarez YR, Cardoso CAL, Antonialli-Junior WF (2018) Influence of temperature on survival and cuticular chemical profile of social wasps. J Therm Biol 71:221–231. https://doi.org/10.1016/j.jtherbio.2017.11.019
Miller D (1927) The gum-tree weevil and its parasites. N Zeal J Agric 35:283–289
Moore HE, Butcher JB, Adam CD, Day CR, Drijfhout FP (2016) Age estimation of Calliphora (Diptera: Calliphoridae) larvae using cuticular hydrocarbon analysis and Artificial Neural Networks. Foren Sci Int 268:81–91. https://doi.org/10.1016/j.forsciint.2016.09.012
Mothapo NP, Wossler TC (2016) "You are not always what you eat": diet did not override intrinsic nestmate recognition cues in Argentine ants from two supercolonies in South Africa. African Zool 51:161–171 https://doi.org/10.1080/15627020.2016.1236670
Otte T, Hilker M, Geiselhardt S (2018) Phenotypic plasticity of cuticular hydrocarbon profiles in insects. J Chem Ecol 44:235–247. https://doi.org/10.1007/s10886-018-0934-4
Pokorny T, Lunau K, Quezada-Euan JJG, Eltz T (2014) Cuticular hydrocarbons distinguish cryptic sibling species in Euglossa orchid bees. Apidologie 45:276–283. https://doi.org/10.1007/s13592-013-0250-5
Pullen KR, Jennings D, Oberprieler RG (2014) Annotated catalogue of Australian weevils (Coleoptera: Curculionoidea) Zootaxa 3896:1–481
Salerno G, Frati F, Conti E, De Pasquale C, Peri E, Colazza S (2009) A finely tuned strategy adopted by an egg parasitoid to exploit chemical traces from host adults. J Exp Biol 212:1825–1831. https://doi.org/10.1242/jeb.028308
Schlechter-Helas J, Schmitt T, Peschke K (2012) Learning individual signatures: rove beetle males discriminate unreceptive females by cuticular hydrocarbon patterns. Anim Behav 84:369–376. https://doi.org/10.1016/j.anbehav.2012.05.003
Schröder ML, Slippers B, Wingfield MJ, Hurley BP (2019) Invasion history and management of Eucalyptus snout beetles in the Gonipterus scutellatus species complex. J Pest Sci 1–15
Seppä P, Helanterä H, Trontti K, Punttila P, Chernenko A, Martin SJ, Sundström L (2011) The many ways to delimit species: hairs, genes and surface chemistry. Myrmecolog News 15:31–41
Spikes AE, Paschen MA, Millar JG, Moreira JA, Hamel PB, Schiff NM, Ginzel MD (2010) First contact pheromone identified for a longhorned beetle (Coleoptera: Cerambycidae) in the subfamily Prioninae. J Chem Ecol 36:943–954. https://doi.org/10.1007/s10886-010-9837-8
Stennett M, Etges W (1997) Premating isolation is determined by larval rearing substrates in cactophilic Drosophila mojavensis. III Epicuticular hydrocarbon variation is determined by use of different host plants in Drosophila mojavensis and Drosophila arizonae. J Chem Ecol 23:2803–2824. https://doi.org/10.1023/A:1022519228346
Tooke FGC (1955) The eucalyptus snout-beetle, Gonipterus scutellatus Gyll. Entomology Memoirs, vol 3. Department of Agriculture, Union of South Africa, Pretoria
Turillazzi S, Sledge MF, Moneti G (1998) Use of a simple method for sampling cuticular hydrocarbons from live social wasps. Ethol Ecol Evol 10:293–297. https://doi.org/10.1080/08927014.1998.9522859
Wicker-Thomas C, Chertemps T (2010) Molecular biology and genetics of hydrocarbon production. In: Bagnères AG, Blomquist GJ (eds) Insect Hydrocarbons: Biology, Biochemistry, and Chemical Ecology. Cambridge University Press, Cambridge, pp 53–74. https://doi.org/10.1017/CBO9780511711909.005
Xue HJ, Wei J, Huang ZZ, Li WZ, Yang XK (2018) Your chemical coat tells me you are my delicacy: a predatory stink bug uses cuticular hydrocarbons to identify prey. Chemoecology 28:69–73. https://doi.org/10.1007/s00049-018-0255-7
Acknowledgements
We thank Simon Lawson for research and travel funds through Forest and Wood Products Australia Limited (project number VNC 418-1617) (supported by Biological Control of Eucalypt Pests Alliance, Industry Plantation Management Group and University of the Sunshine Coast) towards a postgraduate research scholarship to NMS. MLS was in receipt of an Australia Awards Fellowship and funded by the South African Department of Science and Technology—Sector-Specific Innovation Fund and Centre of Excellence in Tree Health Biotechnology—Tree Protection Co-Operative Programme. We also thank Jacques Schröder and Hoan Le Ngoc for their help with the collections across eastern Australia; Stephen Elms and Dianne Patzel for their help with collections in Blue Gum plantations in Victoria and South Australia; Francisco Tovar and IPMG for help with collection and access to plantations in south Western Australia, Lone Pine Koala Sanctuary for site access and Rolf Oberprieler for help with weevil taxonomy.
Funding
MLS received Department of Foreign Affairs and Trade, Australian Government (7202). NMS received a postgraduate research scholarship from Forest and Wood Products Australia Limited (project number VNC 418–1617) (supported by Biological Control of Eucalypt Pests Alliance, Industry Plantation Management Group and University of the Sunshine Coast) and MLS received an Australia Awards Fellowship and was funded by the South African Department of Science and Technology—Sector-Specific Innovation Fund and Centre of Excellence in Tree Health Biotechnology—Tree Protection Co-Operative Programme (DST-NRF Centre of Excellence In Tree Health Biotechnology).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Günther Raspotnig.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Souza, N.M., Schröder, M.L., Hayes, R.A. et al. Cuticular hydrocarbons of Gonipterus weevils: are there species differences?. Chemoecology 31, 159–167 (2021). https://doi.org/10.1007/s00049-021-00337-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-021-00337-5