Abstract
Introduction/objectives
The objective of our systematic review and network meta-analysis (NMA) is to investigate which vitamin D and/or calcium regimen would yield the greatest increase in lumbar spine, femoral neck, and total hip bone mineral density (BMD) in adult patients undergoing glucocorticoid therapy.
Method
We performed NMAs based on a prospectively developed protocol. A database search of MEDLINE, EMBASE, Web of Science, CINAHL, CENTRAL and Chinese databases were conducted for relevant randomized controlled trials (RCTs). Outcomes were percentage change in lumbar spine, femoral neck, and total hip BMD from baseline.
Results
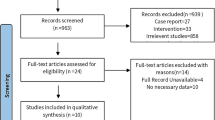
We included 16 RCTs containing 1073 eligible patients in our analysis. We found alfacalcidol+calcium to yield the greatest percentage increase in lumbar spine BMD (MD 6.05, 95% credible interval [CrI] − 4.18 to 16.18) compared to no treatment, and calcitriol+calcium to yield the greatest percentage increase in femoral neck BMD (MD 8.46, 95% CrI − 4.74 to 21.51) compared to no treatment. Cholecalciferol+calcium ranked first in terms of its ability to increase total hip BMD; however this finding needs to be interpreted with caution due to low sample sizes in the cholecalciferol+calcium treatment arm. None of the treatment arms ruled out the possibility of no effect for any outcome.
Conclusions
Alfacalcidol and calcitriol were the most efficacious treatment arms for increasing lumbar spine and femoral neck BMD, respectively. Our findings need to be validated by further investigations using larger, better-designed RCTs.
Key Points •The efficacy of calcium/vitamin D compounds was examined using network meta-analyses. •Alfacalcidol + calcium yielded the greatest increase in lumbar spine BMD, calcitriol + calcium yielded the greatest increase in femoral neck BMD. •Future guidelines should place greater emphasis on the efficacy of different vitamin D compounds. |




Similar content being viewed by others
References
Golder V, Hoi A (2017) Systemic lupus erythematosus: an update. Med J Aust 206:215–220. https://doi.org/10.5694/mja16.01229
Becker DE (2013) Basic and clinical pharmacology of glucocorticosteroids. Anesth Prog 60:25–31. https://doi.org/10.2344/0003-3006-60.1.25
Criswell LA, Saag KG, Sems KM, et al (2000) Moderate-term, low-dose corticosteroids for rheumatoid arthritis. Cochrane Database Syst Rev CD001158. https://doi.org/10.1002/14651858.CD001158
The Cochrane Collaboration (1996) Short-term low-dose corticosteroids vs placebo and nonsteroidal antiinflammatory drugs in rheumatoid arthritis. In: The Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd, Chichester, UK, p 523
Burns CM (2016) The history of cortisone discovery and development. Rheum Dis Clin N Am 42(1–14):vii–14. https://doi.org/10.1016/j.rdc.2015.08.001
Overman RA, Yeh J-Y, Deal CL (2013) Prevalence of oral glucocorticoid usage in the United States: a general population perspective. Arthritis Care Res 65:294–298. https://doi.org/10.1002/acr.21796
Huscher D, Thiele K, Gromnica-Ihle E, Hein G, Demary W, Dreher R, Zink A, Buttgereit F (2009) Dose-related patterns of glucocorticoid-induced side effects. Ann Rheum Dis 68:1119–1124. https://doi.org/10.1136/ard.2008.092163
Wang M (2005) The role of glucocorticoid action in the pathophysiology of the metabolic syndrome. Nutr Metab 2:3. https://doi.org/10.1186/1743-7075-2-3
Reid IR (1998) Glucocorticoid effects on bone. J Clin Endocrinol Metab 83:1860–1862. https://doi.org/10.1210/jcem.83.6.4911
Kuchuk NO, Hoes JN, Bijlsma JWJ, Jacobs JWG (2014) Glucocorticoid-induced osteoporosis: an overview. Int J Clin Rheumatol 9:311–326
Buckley L, Guyatt G, Fink HA, Cannon M, Grossman J, Hansen KE, Humphrey MB, Lane NE, Magrey M, Miller M, Morrison L, Rao M, Robinson AB, Saha S, Wolver S, Bannuru RR, Vaysbrot E, Osani M, Turgunbaev M, Miller AS, McAlindon T (2017) 2017 American College of Rheumatology Guideline for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Rheumatol 69:1521–1537. https://doi.org/10.1002/art.40137
Canalis E, Mazziotti G, Giustina A, Bilezikian JP (2007) Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 18:1319–1328. https://doi.org/10.1007/s00198-007-0394-0
Hahn TJ, Halstead LR, Baran DT (1981) Effects off short term glucocorticoid administration on intestinal calcium absorption and circulating vitamin D metabolite concentrations in man. J Clin Endocrinol Metab 52:111–115. https://doi.org/10.1210/jcem-52-1-111
Wajchenberg BL, Pereira VG, Kieffer J, Ursic S (1969) Effect of dexamethasone on calcium metabolism and 47Ca kinetics in normal subjects. Acta Endocrinol 61:173–192. https://doi.org/10.1530/acta.0.0610173
Huybers S, Naber THJ, Bindels RJM, Hoenderop JGJ (2007) Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am J Physiol Gastrointest Liver Physiol 292:G92–G97. https://doi.org/10.1152/ajpgi.00317.2006
Shi X-M, Chutkan N, W. M, M. C (2012) Mechanism of glucocorticoid-induced osteoporosis: an update. In: Qian X (ed) Glucocorticoids - new recognition of our familiar friend. InTech
Deng J, Silver Z, Huang E, Zheng E, Kavanagh K, Wen A, Cheng W, Dobransky J, Sanger S, Grammatopoulos G (2020) Pharmacological prevention of fractures in patients undergoing glucocorticoid therapies: a systematic review and network meta-analysis. Rheumatology. https://doi.org/10.1093/rheumatology/keaa228
Faltinsen EG, Storebø OJ, Jakobsen JC, Boesen K, Lange T, Gluud C (2018) Network meta-analysis: the highest level of medical evidence? BMJ Evid Based Med 23:56–59. https://doi.org/10.1136/bmjebm-2017-110887
Higgins JPT, Thomas J (2019) Cochrane handbook for systematic reviews of interventions. John Wiley & Sons
Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, Ioannidis JPA, Straus S, Thorlund K, Jansen JP, Mulrow C, Catalá-López F, Gøtzsche PC, Dickersin K, Boutron I, Altman DG, Moher D (2015) The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 162:777–784. https://doi.org/10.7326/M14-2385
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JPA, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. https://doi.org/10.1136/bmj.b2700
Haney EM, Huffman LH, Bougatsos C, et al (2016) Screening for lipid disorders in children and adolescents (07-0598-EF-1). Agency for Healthcare Research and Quality (US), Rockville, MD
Higgins JPT, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
van Valkenhoef G, Lu G, de Brock B et al (2012) Automating network meta-analysis. Res Synth Methods 3:285–299. https://doi.org/10.1002/jrsm.1054
Willink R, Lira I (2005) A united interpretation of different uncertainty intervals. Measurement 38:61–66
Mbuagbaw L, Rochwerg B, Jaeschke R, Heels-Andsell D, Alhazzani W, Thabane L, Guyatt GH (2017) Approaches to interpreting and choosing the best treatments in network meta-analyses. Systematic Reviews 6:79
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Chaimani A, Higgins JPT, Mavridis D, Spyridonos P, Salanti G (2013) Graphical tools for network meta-analysis in STATA. PLoS One 8:e76654. https://doi.org/10.1371/journal.pone.0076654
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634. https://doi.org/10.1136/bmj.315.7109.629
Nikolakopoulou A, Higgins JPT, Papakonstantinou T, Chaimani A, del Giovane C, Egger M, Salanti G (2020) CINeMA: an approach for assessing confidence in the results of a network meta-analysis. PLoS Med 17:e1003082. https://doi.org/10.1371/journal.pmed.1003082
Papakonstantinou T, Nikolakopoulou A, Higgins JPT, Egger M, Salanti G (2020) CINeMA: software for semiautomated assessment of the confidence in the results of network meta-analysis. Campbell Syst Rev 16:1. https://doi.org/10.1002/cl2.1080
Puhan MA, Schünemann HJ, Murad MH et al (2014) A GRADE working group approach for rating the quality of treatment effect estimates from network meta-analysis. BMJ 349:g5630. https://doi.org/10.1136/bmj.g5630
Sterne JAC, Sutton AJ, Ioannidis JPA, Terrin N, Jones DR, Lau J, Carpenter J, Rucker G, Harbord RM, Schmid CH, Tetzlaff J, Deeks JJ, Peters J, Macaskill P, Schwarzer G, Duval S, Altman DG, Moher D, Higgins JPT (2011) Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 343:d4002. https://doi.org/10.1136/bmj.d4002
Bernstein CN, Seeger LL, Anton PA et al (1996) A randomized, placebo-controlled trial of calcium supplementation for decreased bone density in corticosteroid-using patients with inflammatory bowel disease: a pilot study. Aliment Pharmacol Ther 10:777–786. https://doi.org/10.1046/j.1365-2036.1996.63205000.x
Li Z, Chines AA, Meredith MP (2004) Statistical validation of surrogate endpoints: is bone density a valid surrogate for fracture? J Musculoskelet Neuronal Interact 4:64–74
Deng J, Abbas U, Chang O, Dhivagaran T, Sanger S, Bozzo A (2020) Antidiabetic and antiosteoporotic pharmacotherapies for prevention and treatment of type 2 diabetes-induced bone disease: protocol for two network meta-analyses. BMJ Open 10:e034741. https://doi.org/10.1136/bmjopen-2019-034741
Wu T, Li Y, Bian Z, Liu G, Moher D (2009) Randomized trials published in some Chinese journals: how many are randomized? Trials 10:46. https://doi.org/10.1186/1745-6215-10-46
Acknowledgments
We would like to acknowledge Mr. Wenteng Hou and Mr. Aaron Wen of Faculty of Health Sciences, McMaster University for their assistance during the data extraction portion of this study. Their willingness to donate their time has been very much appreciated.
Availability of data and material
All results from our analyses are published in the supplementary material. Aggregated patient data supplied by the included RCTs and extracted by our investigators is available upon reasonable request. We are happy to provide these data to help validate and replicate the findings of our study. Please contact the corresponding or first authors for data requests.
Code availability
The R code used in this study is available upon reasonable request. Please contact the corresponding or first authors.
Author information
Authors and Affiliations
Contributions
Jiawen Deng: conceptualization, methodology, software, formal analysis, resources, data curation, writing—original draft, visualization, supervision, project administration. Zachary Silver: resources, data curation, writing—review and editing. Emma Huang: resources, data curation, writing—review and editing. Elena Zheng: data curation, writing—review and editing. Kyra Kavanagh: data curation, writing—review and editing. Jannusha Panicker: data curation, writing—review and editing.
Corresponding author
Ethics declarations
Disclosures
None.
Ethics approval
None required. The manuscript does not contain clinical studies or patient data.
Consent to participate
None required. The manuscript does not contain clinical studies or patient data.
Consent for publication
All authors had reviewed the final manuscript and give consent for submission and publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1088 kb)
Rights and permissions
About this article
Cite this article
Deng, J., Silver, Z., Huang, E. et al. The effect of calcium and vitamin D compounds on bone mineral density in patients undergoing glucocorticoid therapies: a network meta-analysis. Clin Rheumatol 40, 725–734 (2021). https://doi.org/10.1007/s10067-020-05294-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05294-y