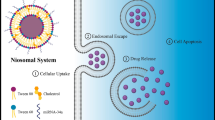
Abstract
Objective
Tumor suppressor miRNAs, miR-15a and miR-16–1, with high-specificity and oncogenic targeting of Bcl-2, can target tumor tissues. Disadvantages of the clinical application of free miRNAs include poor cellular uptake and instability in plasma, which can be partially improved by using nanocarriers to deliver anti-cancer agents to the tumor cell.
Method
In this study, cationic niosomes were designed and optimized with the specific formulation. Then, the physical characteristics, the cytotoxicity, the impact of transfected miRNAs on the expression of the Bcl-2 gene, and the apoptosis rate of the different formulation into prostate cancer cell were determined.
Results
The optimum formulation containing tween-60: cholesterol: DOTAP: DSPE-PEG2000 at 70:30:25:5 demonstrated that the vesicle size and zeta potentials were 69.7 nm and + 14.83 mV, respectively. Additionally, noisome-loaded miRNAs had higher toxicity against cancer cells comparing with free forms. The transfection of PC3 cells with the combination therapy of nanocarriers loaded of two miRNAs led to a significant decrease in the expression of the Bcl-2 gene and increased the degree of cell death in PC3 cells compared with other treatment groups, and the synergistic effects of co-delivery of miR-15a and miR-16–1 on prostate cancer cells were shown.
Conclusion
According to the results, it seems the designed niosomes containing miR-15a and miR-16–1 can target the Bcl-2 gene and provide a cheap, applicable, cost-effective, and safe drug delivery system against prostate cancer.






Similar content being viewed by others
References
Acunzo M, Croce CM (2016) Downregulation of miR-15a and miR-16–1 at 13q14 in chronic lymphocytic leukemia. Clin Chem 62:655–656. https://doi.org/10.1373/clinchem.2015.240036
Albamonte MS, Willis MA, Albamonte MI, Jensen F, Espinosa MB, Vitullo AD (2008) The developing human ovary: immunohistochemical analysis of germ-cell-specific VASA protein, BCL-2/BAX expression balance and apoptosis. Human Reprod 23:1895–1901
Aqeilan R, Calin G, Croce C (2009) Aqeilan RI, Calin GA, Croce CM. miR-15a and miR-16–1 in cancer: discovery, function and future perspectives. Cell Death Differ 17:215–220. https://doi.org/10.1038/cdd.2009.69
Aqeilan RI, Calin GA, Croce CM (2010) miR-15a and miR-16–1 in cancer: discovery, function and future perspectives. Cell Death Differ 17:215–220. https://doi.org/10.1038/cdd.2009.69
Bai Z et al (2019) Non-viral nanocarriers for intracellular delivery of microRNA therapeutics. J Mater Chem B 7:1209–1225. https://doi.org/10.1039/C8TB02946F
Bandi N et al (2009) miR-15a and miR-16 are implicated in cell cycle regulation in a Rb-dependent manner and are frequently deleted or down-regulated in non–small cell lung cancer. Cancer Res 69:5553–5559
Baraban JM, Shah A, Fu X (2018) Chapter one - multiple pathways mediate microRNA degradation: focus on the translin/trax RNase complex. In: Pasternak GW, Coyle JT (eds) Advances in pharmacology, vol 82. Academic Press, New York, pp 1–20
Bonci D et al (2008) The miR-15a–miR-16–1 cluster controls prostate cancer by targeting multiple oncogenic activities. Nat Med 14:1271. https://doi.org/10.1038/nm.1880
Cai CK et al (2012) miR-15a and miR-16–1 downregulate CCND1 and induce apoptosis and cell cycle arrest in osteosarcoma. Oncol Rep 28:1764–1770. https://doi.org/10.3892/or.2012.1995
Campbell KJ, Tait SWG (2018) Targeting BCL-2 regulated apoptosis in cancer. Open Biol 8:180002. https://doi.org/10.1098/rsob.180002
Chen L, Tang L, Calin G, Croce CM, Kipps TJ (2006) Expression of microRNA (miR) miR-15a/miR-16–1 downregulates expression of BCL-2 protein in chronic lymphocytic leukemia. Blood 108:2796–2796. https://doi.org/10.1182/blood.V108.11.2796.2796
Cimmino A et al (2005) miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl Acad Sci U S A 102:13944–13949. https://doi.org/10.1073/pnas.0506654102
Ding L, Hu X-M, Wu H, Liu G-X, Gao Y-J, He D-M, Zhang Y (2013) Combined transfection of Bcl-2 siRNA and miR-15a oligonucleotides enhanced methotrexate-induced apoptosis in Raji cells. Cancer Biol Med 10:16
Guo WT, Wang Y (2019) Dgcr8 knockout approaches to understand microRNA functions in vitro and in vivo. Cell Mol Life Sci 76:1697–1711. https://doi.org/10.1007/s00018-019-03020-9
Haghiralsadat F, Amoabediny G, Helder MN, Naderinezhad S, Sheikhha MH, Forouzanfar T, Zandieh-Doulabi B (2018a) A comprehensive mathematical model of drug release kinetics from nano-liposomes, derived from optimization studies of cationic PEGylated liposomal doxorubicin formulations for drug-gene delivery. Artif Cells Nanomed Biotechnol 46:169–177. https://doi.org/10.1080/21691401.2017.1304403
Haghiralsadat F, Amoabediny G, Naderinezhad S, Forouzanfar T, Helder MN, Zandieh-Doulabi B (2018b) Preparation of PEGylated cationic nanoliposome-siRNA complexes for cancer therapy. Artif Cells Nanomed Biotechnol 46:684–692. https://doi.org/10.1080/21691401.2018.1434533
Haghiralsadat F, Amoabediny G, Naderinezhad S, Zandieh-Doulabi B, Forouzanfar T, Helder MN (2018c) Codelivery of doxorubicin and JIP1 siRNA with novel EphA2-targeted PEGylated cationic nanoliposomes to overcome osteosarcoma multidrug resistance. Int J Nanomed 13:3853–3866. https://doi.org/10.2147/ijn.S150017
Hemati M, Haghiralsadat F, Jafary F, Moosavizadeh S, Moradi A (2019) Targeting cell cycle protein in gastric cancer with CDC20siRNA and anticancer drugs (doxorubicin and quercetin) co-loaded cationic PEGylated nanoniosomes. Int J Nanomed 14:6575–6585
Hsu S-h et al (2013) Cationic lipid nanoparticles for therapeutic delivery of siRNA and miRNA to murine liver tumor. Nanomed Nanotechnol Biol Med 9:1169–1180
Huang W (2017) MicroRNAs: biomarkers, diagnostics, and therapeutics. Methods Mol Biol (Clifton, NJ) 1617:57–67. https://doi.org/10.1007/978-1-4939-7046-9_4
Kang W et al (2015) Targeting of YAP1 by microRNA-15a and microRNA-16–1 exerts tumor suppressor function in gastric adenocarcinoma. Mol Cancer 14:52
Kopper L, Tímár J (2005) Genomics of prostate cancer: Is there anything to „translate”? Pathol Oncol Res 11:197–203. https://doi.org/10.1007/BF02893851
Liu J et al (2014) Loss of p53 and altered miR15-a/16–1 MCL-1 pathway in CLL: insights from TCL1-Tg: p53−/− mouse model and primary human leukemia cells. Leukemia 28:118
Naderinezhad S, Amoabediny G, Haghiralsadat F (2017) Co-delivery of hydrophilic and hydrophobic anticancer drugs using biocompatible pH-sensitive lipid-based nano-carriers for multidrug-resistant cancers RSC. Advances 7:30008–30019. https://doi.org/10.1039/C7RA01736G
Palanichamy JK, Rao DS (2014) miRNA dysregulation in cancer: towards a mechanistic understanding. Front Genet 5:54–54. https://doi.org/10.3389/fgene.2014.00054
Patel N, Garikapati KR, Ramaiah MJ, Polavarapu KK, Bhadra U, Bhadra MP (2016) miR-15a/miR-16 induces mitochondrial dependent apoptosis in breast cancer cells by suppressing oncogene BMI1. Life Sci 164:60–70. https://doi.org/10.1016/j.lfs.2016.08.028
Pejin B, Jovanovic KK, Mojovic M, Savic AG (2013) New and highly potent antitumor natural products from marine-derived fungi: Covering the period from 2003 to 2012. Curr Top Med Chem 13:2745–2766
Pejin B, Jovanovic KK, Savic AG (2015) New antitumour natural products from marine red algae: covering the period from 2003 to 2012. Mini Rev Med Chem 15:720–730
Porkka KP et al (2011) The miR-15a-miR-16-1 locus is homozygously deleted in a subset of prostate cancers. Genes Chromos Cancer 50:499–509
Qian H, Tay CY, Setyawati MI, Chia SL, Lee DS, Leong DT (2017) Protecting microRNAs from RNase degradation with steric DNA nanostructures. Chem Sci 8:1062–1067. https://doi.org/10.1039/c6sc01829g
Sambri I, Capasso R, Pucci P, Perna AF, Ingrosso D (2011) The microRNA 15a/16–1 cluster down-regulates protein repair isoaspartyl methyltransferase in hepatoma cells: implications for apoptosis regulation. J Biol Chem 286:43690–43700. https://doi.org/10.1074/jbc.M111.290437
Sun CY et al (2013) miR-15a and miR-16 affect the angiogenesis of multiple myeloma by targeting VEGF. Carcinogenesis 34:426–435. https://doi.org/10.1093/carcin/bgs333
Xia L et al (2008) miR-15b and miR-16 modulate multidrug resistance by targeting BCL2 in human gastric cancer cells. Int J Cancer 123:372–379. https://doi.org/10.1002/ijc.23501
Yang N (2015) An overview of viral and nonviral delivery systems for microRNA. Int J Pharm Investig 5:179–181. https://doi.org/10.4103/2230-973X.167646
Zhang XJ, Ye H, Zeng CW, He B, Zhang H, Chen YQ (2010) Dysregulation of miR-15a and miR-214 in human pancreatic cancer. J Hematol Oncol 3:46
Zhou K, Liu M, Cao Y (2017) New insight into microRNA functions in cancer: oncogene-microRNA-tumor suppressor gene network. Front Mol Biosci 4:46–46. https://doi.org/10.3389/fmolb.2017.00046
Zhu C-S, Zhu L, Tan D-A, Qiu X-Y, Liu C-Y, Xie S-S, Zhu L-Y (2019) Avenues toward microRNA detection in vitro: a review of technical advances and challenges. Comput Struct Biotechnol J 17:904–916. https://doi.org/10.1016/j.csbj.2019.06.018
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghaffari, M., Kalantar, S.M., Hemati, M. et al. Co-delivery of miRNA-15a and miRNA-16–1 using cationic PEGylated niosomes downregulates Bcl-2 and induces apoptosis in prostate cancer cells. Biotechnol Lett 43, 981–994 (2021). https://doi.org/10.1007/s10529-021-03085-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-021-03085-2