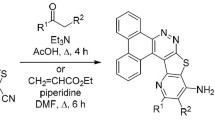
New di-, tri-, tetra-, and pentanorlabdane compounds with thiosemicarbazone and 1,3-thiazole fragments were synthesized. Their antifungal and antibacterial activities were studied. The main advantages of this research were the available starting material, i.e., the natural labdane diterpenoid (–)-sclareol, which was isolated from renewable resources, and the high probability of biological activity combined with the low toxicity of these compounds because of their natural origin.
Similar content being viewed by others
References
J. R. Hanson, Nat. Prod. Rep., 29 (8), 890 (2012).
I. Chinou, Curr. Med. Chem., 12, 1295 (2005).
M. Singh, M. Pal, and R. P. Sharma, Planta Med., 65 (1), 2 (1999).
R. R. Sharipova, O. V. Andreeva, B. F. Garifullin, I. Yu. Strobykina, A. S. Strobykina, A. D. Voloshina, M. A. Kravchenko, and V. E. Kataev, Chem. Nat. Compd., 54, 92 (2018).
L. E. Catalan, E. B. Maturana, K. C. Marin, M. O. Olivares, H. C. Altamirano, M. C. Cuellar Fritis, and J. V. Garcia, Molecules, 15 (9), 6502 (2010).
K. Kuchkova, A. Aricu, A. Barba, P. Vlad, S. Shova, E. Secara, N. Ungur, Gh. Zbancioc, and I. I. Mangalagiu, Synlett, 24, 697 (2013).
A. Aricu, A. Ciocarlan, L. Lungu, A. Barba, S. Shova, G. Zbancioc, I. I. Mangalagiu, M. D’Ambrosio, and N. Vornicu, Med. Chem. Res., 25 (10), 2316 (2016).
H. Beraldo and D. Gambino, Mini-Rev. Med. Chem., 4, 31 (2004).
A. I. Matesanz, I. Leitao, and P. Souza, J. Inorg. Biochem., 125, 26 (2013).
A. Rajasekaran and S. Murugesan, J. Indian Chem. Soc., 79 (6), 544 (2002).
F. Vandresen, H. Falzirolli, S. A. Almeida Batista, A. P. B. da Silva-Giardini, D. N. de Oliveira, R. R. Catharino, A. L. T. G. Ruiz, J. E. de Carvalho, M. A. Foglio, and C. Conceicao da Silva, Eur. J. Med. Chem., 79, 110 (2014).
V. P. Kirin, V. A. Maksakov, A. V. Virovets, S. A. Popov, and A. V. Tkachev, Inorg. Chem. Commun., 5, 32 (2002).
S. Blaja, Chem. J. Mold., 14 (2), 72 (2019).
J. J. Liu, Y. J. Lu, Z. D. Zhao, S. C. Xu, and L. W. Bi, Chem. Nat. Compd., 54, 695 (2018).
S. K. Hua, J. Wang, X. B. Chen, Z. Y. Xu, and B. B. Zeng, Tetrahedron, 67, 1142 (2011).
C. Cucicova, A. Aricu, E. Secara, P. Vlad, and N. Ungur, MD Pat. No. 8, 22 (2013).
R. M. Mohareb, W. W. Wardakhan, G. A. Elmegeed, and R. M. S. Ashour, Steroids, 77, 1560 (2012).
K. Kuchkova, Y. M. Chumakov, Y. A. Simonov, G. Bocelli, A. A. Panasenko, and P. F. Vlad, Synthesis, 9, 1045 (1997).
M. N. Koltsa, G. N. Mironov, S. T. Malinovskii, and P. F. Vlad, Russ. Chem. Bull., 45 (1), 208 (1996).
A. Aricu, A. Ciocarlan, L. Lungu, S. Shova, G. Zbancioc, I. Mangalagiu, and N. Vornicu, Med. Chem. Res., 25 (10), 2316 (2016).
Acknowledgment
We thank the World Federation of Scientists for financial support of scientific research on the Project “Synthesis, characterization and biological activity of di- and trinorlabdane compounds with 1,3-thiazole units.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2021, pp. 84–92.
Rights and permissions
About this article
Cite this article
Blaja, S.P., Lungu, L.V., Kuchkova, K.I. et al. Norlabdane Compounds Containing Thiosemicarbazone or 1,3-Thiazole Fragments: Synthesis and Antimicrobial Activity. Chem Nat Compd 57, 101–110 (2021). https://doi.org/10.1007/s10600-021-03292-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-021-03292-3