Abstract
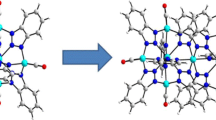
The reaction of Ni(OAc)2·4H2O and 5-aminodiacetic isophthalate (H4adip) in the aqueous solution to produce a discrete nickel coordination compound, {[Ni1.5(H2O)9][Ni(adip)(H2O)3][Ni1.5(adip)(H2O)5]·10H2O}2 (1), whereas another nickel compound was isolated by assembling the same reactants at 80 °C in the mixed solvents of H2O and DMA, {[Ni(H2O)6][Ni3(adip)2(H2O)8]·7.5H2O}2 (2). Both 1 and 2 were structurally characterized by single-crystal X-ray diffraction, and other methods. The skeleton unit of 1 consisted of five crystallographically independent Ni2+ ions and two types of adip4− ligands, and the adjacent units were further generated a 3D supramolecular architecture through hydrogen bonds and π···π stacking interactions. Analogously, four types of six-coordinated Ni2+ ions and two kinds of adip4− ligands in 2 also formed a 3D supramolecular structure by hydrogen bonds and π···π stacking interactions. Moreover, the magnetic properties of 1 and 2 had been examined, and the results suggested that the presence of antiferromagnetic interactions among Ni2+ ions in both compounds.




Similar content being viewed by others
References
P.-Q. Liao, N.-Y. Huang, W.-X. Zhang, J.-P. Zhang, and X.-M. Chen (2017). Science 356, 1193.
S. Zhang, W. Shi, and P. Cheng (2017). Coord. Chem. Rev. 352, 108.
J. Dong, P. Cui, P.-F. Shi, P. Cheng, and B. Zhao (2015). J. Am. Chem. Soc. 137, 15988.
S.-L. Li, M. Han, Y. Zhang, G.-P. Li, M. Li, G. He, and X.-M. Zhang (2019). J. Am. Chem. Soc. 141, 12663.
J.-H. Wang, Y. Zhang, M. Li, S. Yan, D. Li, and X.-M. Zhang (2017). Angew. Chem. Int. Ed. 56, 6478.
Y. Zhang, S. Yuan, G. Day, X. Wang, X. Yang, and H.-C. Zhou (2018). Coord. Chem. Rev. 354, 28.
H. Xu, C.-S. Cao, H.-S. Hu, S.-B. Wang, J.-C. Liu, P. Cheng, N. Kaltsoyannis, J. Li, and B. Zhao (2019). Angew. Chem. Int. Ed. 58, 6022.
S. Zhang and P. Cheng (2016). Chem. Rec. 16, 2077.
Q. Ba, J. Qian, and C. Zhang (2019). J. Clust. Sci. 30, 747.
S.-J. Liu, C. Cao, C.-C. Xie, T.-F. Zheng, X.-L. Tong, J.-S. Liao, J.-L. Chen, H.-R. Wen, Z. Chang, and X.-H. Bu (2016). Dalton Trans. 45, 9209.
J. Gu, Y. Cui, X. Liang, J. Wu, D. Lv, and A. M. Kirillov (2016). Cryst. Growth Des. 16, 4658.
K. Liu, X. Zhang, X. Meng, W. Shi, P. Cheng, and A. K. Powell (2016). Chem. Soc. Rev. 45, 2423.
J. Liu, L. Chen, H. Cui, J. Zhang, L. Zhang, and C.-Y. Su (2014). Chem. Soc. Rev. 43, 6011.
J.-Y. Zou, L. Li, S.-Y. You, H.-M. Cui, Y.-W. Liu, K.-H. Chen, Y.-H. Chen, J.-Z. Cui, and S.-W. Zhang (2018). Dyes Pigm. 159, 429.
Z.-L. Wu, C.-H. Wang, B. Zhao, J. Dong, F. Lu, W.-H. Wang, W.-C. Wang, G.-J. Wu, J.-Z. Cui, and P. Cheng (2016). Angew. Chem. Int. Ed. 55, 4938.
H.-L. Wang, L.-B. Sheng, H.-H. Zou, K. Wang, B. Li, M.-S. Chen, and F.-P. Liang (2018). J. Clust. Sci. 29, 1313.
J.-Y. Zou, L. Li, S.-Y. You, Y.-W. Liu, H.-M. Cui, K.-H. Chen, and S.-W. Zhang (2019). J. Inorg. Organomet. Polym. Mater. 29, 359.
J. Li, R. Wan, H. Li, Y. Liu, S. Zhang, and P. Ma (2019). J. Mol. Struct. 1181, 142.
S.-Y. Zhang, Z.-Y. Wang, J. Gao, K. Wang, E. Gianolio, S. Aime, W. Shi, Z. Zhou, P. Cheng, and M. J. Zaworotko (2019). Chem 5, 1609.
Y. Wang, P. Xu, Q. Xie, Q.-Q. Ma, Y.-H. Meng, Z.-W. Wang, S. Zhang, X.-J. Zhao, J. Chen, and Z.-L. Wang (2016). Chem. Eur. J. 22, 10459.
C.-S. Liu, Z.-H. Zhang, M. Chen, H. Zhao, F.-H. Duan, D.-M. Chen, M.-H. Wang, S. Zhang, and M. Du (2017). Chem. Commun. 53, 3941.
S.-D. Su, X.-M. Ou-Yang, K.-K. Guo, Q.-M. Lin, Y. Li, K. Wang, S.-Y. Luo, and F.-P. Liang (2019). J. Clust. Sci. 30, 863.
L. Ungur, S.-Y. Lin, J. Tang, and L. F. Chibotaru (2014). Chem. Soc. Rev. 43, 6894.
S. Zhang, H. Li, E. Duan, Z. Han, L. Li, J. Tang, W. Shi, and P. Cheng (2016). Inorg. Chem. 55, 1202.
Y.-X. Wang, Y. Ma, Y. Chai, W. Shi, Y. Sun, and P. Cheng (2018). J. Am. Chem. Soc. 140, 7795.
D.-M. Chen, C.-X. Sun, C.-S. Liu, and M. Du (2018). Inorg. Chem. 57, 7975.
Z. Shao, C. Huang, J. Dang, Q. Wu, Y. Liu, J. Ding, and H. Hou (2018). Chem. Mater. 30, 7979.
H. Wang, W. Meng, J. Wu, J. Ding, H. Hou, and Y. Fan (2016). Coord. Chem. Rev. 307, 130.
X. Zhao, S. Zhang, J. Yan, L. Li, G. Wu, W. Shi, G. Yang, N. Guan, and P. Cheng (2018). Inorg. Chem. 57, 5030.
S. Zhang and P. Cheng (2016). ChemPlusChem 81, 811.
X. Meng, W. Shi, and P. Cheng (2019). Coord. Chem. Rev. 378, 134.
M. Sun, S. Zhang, J. Zhang, W. Xia, J. Chen, and X. Yu (2019). J. Coord. Chem. 72, 1899.
X.-Y. Zheng, X.-J. Kong, Z. Zheng, L.-S. Long, and L.-S. Zheng (2018). Acc. Chem. Res. 51, 517.
S. Ning, H. Chen, S. Zhang, and P. Cheng (2018). Polyhedron 155, 457.
P. Chen, S. Zhang, J. Zhang, W. Xia, and X. Yu (2019). J. Coord. Chem. 72, 239.
S.-J. Liu, C. Cao, F. Yang, M.-H. Yu, S.-L. Yao, T.-F. Zheng, W.-W. He, H.-X. Zhao, T.-L. Hu, and X.-H. Bu (2016). Cryst. Growth Des. 16, 6776.
L. Li, J.-Y. Zou, S.-Y. You, Y.-W. Liu, H.-M. Cui, and S.-W. Zhang (2019). Dyes Pigm. https://doi.org/10.1016/j.dyepig.2019.108004.
B. Zheng, X. Luo, Z. Wang, S. Zhang, R. Yun, L. Huang, W. Zeng, and W. Liu (2018). Inorg. Chem. Front. 5, 2355.
J.-Y. Zou, L. Li, S.-Y. You, Y.-W. Liu, H.-M. Cui, J.-Z. Cui, and S.-W. Zhang (2018). Dalton Trans. 47, 15694.
B. Zheng, H. Wang, Z. Wang, N. Ozaki, C. Hang, X. Luo, L. Huang, W. Zeng, M. Yang, and J. Duan (2016). Chem. Commun. 52, 12988.
Z. Tang, H. Chen, Y. Zhang, B. Zheng, S. Zhang, and P. Cheng (2019). Cryst. Growth Des. 19, 1172.
W. Xu, Z. Shao, C. Huang, R. Xu, B. Dong, and H. Hou (2019). Inorg. Chem. 58, 3959.
Y. Xie, S. Ning, Y. Zhang, Z. Tang, S. Zhang, and R. Tang (2018). Dyes Pigm. 150, 36.
S.-L. Yao, S.-J. Liu, X.-M. Tian, T.-F. Zheng, C. Cao, C.-Y. Niu, Y.-Q. Chen, J.-L. Chen, H. Huang, and H.-R. Wen (2019). Inorg. Chem. 58, 3578.
R.-X. Yao, X. Cui, J. Wang, and X.-M. Zhang (2015). Chem. Commun. 51, 5108.
S.-Q. Lu, K. Fang, Y.-Y. Liu, M.-X. Li, S.-J. Liu, and X. He (2018). CrystEngComm 20, 5045.
S.-J. Liu, X.-R. Xie, T.-F. Zheng, J. Bao, J.-S. Liao, J.-L. Chen, and H.-R. Wen (2015). CrystEngComm 17, 7270.
H. Chen, P. Fan, X. Tu, H. Min, X. Yu, X. Li, J.-L. Zeng, S. Zhang, and P. Cheng (2019). Chem. Asian J. 14, 3611.
S. A. Sotnik, R. A. Polunin, M. A. Kiskin, A. M. Kirillov, V. N. Dorofeeva, K. S. Gavrilenko, I. L. Eremenko, V. M. Novotortsev, and S. V. Kolotilov (2015). Inorg. Chem. 54, 5169.
M. N. Kopylovich, E. A. Tronova, M. Haukka, A. M. Kirillov, V. Y. Kukushkin, J. J. R. Fraústo da Silva, and A. J. L. Pombeiro (2007). Eur. J. Inorg. Chem. 2007, 4621.
M. V. Kirillova, A. M. Kirillov, M. F. C. Guedes da Silva, M. N. Kopylovich, J. J. R. Fraústo da Silva, and A. J. L. Pombeiro (2008). Inorg. Chim. Acta 361, 1728.
S. Zhang, J. Ma, X. Zhang, E. Duan, and P. Cheng (2015). Inorg. Chem. 54, 586.
S. Zhang, W. Shi, L. Li, E. Duan, and P. Cheng (2014). Inorg. Chem. 53, 10340.
S. Zhang, E. Duan, Z. Han, L. Li, and P. Cheng (2015). Inorg. Chem. 54, 6498.
F.-L. Ma, L. Jiang, M.-Y. Chen, S.-Q. Wu, D. Zhang, R. Feng Han, and M.-C. Hong (2011). Cryst. Growth Des. 11, 3273.
J. Ma, F.-L. Jiang, L. Chen, M.-Y. Wu, S.-Q. Zhang, K.-C. Xiong, D. Han, and M.-C. Hong (2012). CrystEngComm 14, 6055.
Y. Xu, B. Chen, Y. Gong, D. Yuan, F. Jiang, and M. Hong (2006). J. Mol. Struct. 789, 220.
Y. Xu, D. Yuan, B. Wu, L. Han, M. Wu, F. Jiang, and M. Hong (2006). Cryst. Growth Des. 6, 1168.
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann (2009). J. Appl. Crystallogr. 42, 339.
G. Sheldrick (2015). Acta Crystallogr. Sect. C Struct. Chem. 71, 3.
L. Liu, C. Huang, X. Xue, M. Li, H. Hou, and Y. Fan (2015). Cryst. Growth Des. 15, 4507.
F. Guo, B. Zhu, M. Liu, X. Zhang, J. Zhang, and J. Zhao (2013). CrystEngComm 15, 6191.
J. Cano, G. De Munno, F. Lloret, and M. Julve (2000). Inorg. Chem. 39, 1611.
Y.-P. Li, Y. Chai, G.-P. Yang, H.-H. Miao, L. Cui, Y.-Y. Wang, and Q.-Z. Shi (2014). Dalton Trans. 43, 10947.
S. Zhang, H. Chen, H. Tian, X. Li, and X. Yu (2017). Inorg. Chem. Commun. 86, 87.
Q. Chu, Z. Su, J. Fan, T.-A. Okamura, G.-C. Lv, G.-X. Liu, W.-Y. Sun, and N. Ueyama (2011). Cryst. Growth Des. 11, 3885.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21601058), the Natural Science Foundation of Hunan Province (2018JJ3146), Hunan Provincial Innovation Foundation for Postgraduate (CX2018B672), and Hunan University of Science and Technology (E51677).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tu, X., Chen, H., Zhang, S. et al. Temperature/Solvent-Induced Two Magnetic Nickel Coordination Compounds with 5-Aminodiacetic Isophthalate. J Clust Sci 31, 1199–1206 (2020). https://doi.org/10.1007/s10876-019-01726-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01726-1