Abstract
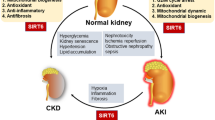
Acute kidney injury (AKI) is a common syndrome associated with high morbidity and mortality, despite progress in medical care. Many studies have shown that there are sex differences and different role of sex hormones particularly estrogens in kidney injury. In this regard, the incidence and rate of progression of kidney diseases are higher in men compared with women. These observations suggest that female sex hormone may be renoprotective. Silent information regulator 2 homolog 1 (SIRT1) is a histone deacetylase, which is implicated in multiple biologic processes in several organisms. In the kidneys, SIRT1 inhibits renal cell apoptosis, inflammation, and fibrosis. Studies have reported a link between SIRT1 and estrogen. In addition, SIRT1 regulates ERα expression and inhibition of SIRT1 activity suppresses ERα expression. This effect leads to inhibition of estrogen-responsive gene expression. In this text, we review the role of SIRT1 in mediating the protective effects of estrogen in the onset and progression of AKI.

Similar content being viewed by others
References
Tao LP, Burdmann E, Mehta R. Acute kidney injury: global health alert. 2013.
National CGCU. Acute Kidney Injury: Prevention, Detection and Management Up to the Point of Renal Replacement Therapy. 2013.
Koza Y (2016) Acute kidney injury: current concepts and new insights. J Injury Violence Res 8(1):58
Hsu C-Y, McCulloch C, Fan D, Ordonez J, Chertow G, Go A (2007) Community-based incidence of acute renal failure. Kidney Int 72(2):208–212
Nash K, Hafeez A, Hou S (2002) Hospital-acquired renal insufficiency. Am J Kidney Dis 39(5):930–936
Hoste EA, Schurgers M (2008) Epidemiology of acute kidney injury: how big is the problem? Crit Care Med 36(4):S146–S151
Rahman M, Shad F, Smith MC (2012) Acute kidney injury: a guide to diagnosis and management. Am Fam Physician 86(7):631–639
Brar H, Olivier J, Lebrun C, Gabbard W, Fulop T, Schmidt D (2008) Predictors of mortality in a cohort of intensive care unit patients with acute renal failure receiving continuous renal replacement therapy. Am J Med Sci 335(5):342–347
Guan Y, Hao C-M (2015) SIRT1 and kidney function. Kidney Diseases 1(4):258–265
Kong L, Wu H, Zhou W, Luo M, Tan Y, Miao L et al (2015) Sirtuin 1: a target for kidney diseases. Mol Med 21(1):87–97
Hasegawa K, Wakino S, Simic P, Sakamaki Y, Minakuchi H, Fujimura K et al (2013) Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing Claudin-1 overexpression in podocytes. Nat Med 19(11):1496–1504
Gao R, Chen J, Hu Y, Li Z, Wang S, Shetty S et al (2014) Sirt1 deletion leads to enhanced inflammation and aggravates endotoxin-induced acute kidney injury. PLoS ONE 9(6):e98909
Neugarten J. Gender and the progression of renal disease. Am Soc Nephrol; 2002.
Dixon A, Maric C (2007) 17β-Estradiol attenuates diabetic kidney disease by regulating extracellular matrix and transforming growth factor-β protein expression and signaling. Am J Physiol-Renal Physiol 293(5):F1678–F1690
Müller V, Szabó A, Viklicky O, Gaul I, Pörtl S, Philipp T et al (1999) Sex hormones and gender-related differences: their influence on chronic renal allograft rejection. Kidney Int 55(5):2011–2020
Park KM, Kim JI, Ahn Y, Bonventre AJ, Bonventre JV (2004) Testosterone is responsible for enhanced susceptibility of males to ischemic renal injury. J Biol Chem 279(50):52282–52292
Metcalfe PD, Leslie JA, Campbell MT, Meldrum DR, Hile KL, Meldrum KK (2008) Testosterone exacerbates obstructive renal injury by stimulating TNF-α production and increasing proapoptotic and profibrotic signaling. Am J Physiol-Endocrinol Metab 294(2):E435–E443
Reed DK, Arany I (2014) Sex hormones differentially modulate STAT3-dependent antioxidant responses during oxidative stress in renal proximal tubule cells. in vivo. 28(6):1097–100.
Ibrahim IY, Elbassuoni EA, Ragy MM, Habeeb WN (2013) Gender difference in the development of cardiac lesions following acute ischemic-reperfusion renal injury in albino rats. Gen Physiol Biophys 32(3):421–428
Ghazvini H, Khaksari M, Esmaeilpour K, Shabani M, Asadi-Shekaari M, Khodamoradi M et al (2016) Effects of treatment with estrogen and progesterone on the methamphetamine-induced cognitive impairment in ovariectomized rats. Neurosci Lett 619:60–67
Khaksari M, Mahmoodi M, Rezvani ME, Sajjadi MA, Karam GA, Hajizadeh S. Differences between male and female students in cardiovascular and endocrine responses to examination stress. Journal of Ayub Medical College Abbottabad. 2005;17(2).
Group KDIGHOW (2012) Definition and classification of acute kidney injury. Kidney Int 2(Suppl 2):19–36.
Makris K, Spanou L (2016) Acute kidney injury: definition, pathophysiology and clinical phenotypes. Clin Biochem Rev 37(2):85
Lameire N, Van Biesen W, Vanholder R (2008) Acute kidney injury. The Lancet 372(9653):1863–1865
Ozer JS, Dieterle F, Troth S, Perentes E, Cordier A, Verdes P et al (2010) A panel of urinary biomarkers to monitor reversibility of renal injury and a serum marker with improved potential to assess renal function. Nat Biotechnol 28(5):486–494
Obermüller N, Geiger H, Weipert C, Urbschat A (2014) Current developments in early diagnosis of acute kidney injury. Int Urol Nephrol 46(1):1–7
Makris K, Kafkas N (2012) Neutrophil gelatinase-associated lipocalin in acute kidney injury. Adv Clin Chem 58:143
Xue W, Xie Y, Wang Q, Xu W, Mou S, Ni Z (2014) Diagnostic performance of urinary kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin for acute kidney injury in an obstructive nephropathy patient. Nephrology 19(4):186–194
Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A, Group NM-aI. (2009) Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis 54(6):1012–24.
Blantz RC (1998) Pathophysiology of pre-renal azotemia. Kidney Int 53(2):512–523
Kaufman J, Dhakal M, Patel B, Hamburger R (1991) Community-acquired acute renal failure. Am J Kidney Dis 17(2):191–198
Basile DP, Anderson MD, Sutton TA (2011) Pathophysiology of acute kidney injury. Comprehensive Physiol 2(2):1303–1353
Rickert E, Fernandez MO, Choi I, Gorman M, Olefsky JM, Webster NJ (2019) Neuronal SIRT1 regulates metabolic and reproductive function and the response to caloric restriction. J Endocrine Soc 3(2):427–445
McBurney MW, Clark-Knowles KV, Caron AZ, Gray DA (2013) SIRT1 is a highly networked protein that mediates the adaptation to chronic physiological stress. Genes Cancer 4(3–4):125–134
Chang H-C, Guarente L (2014) SIRT1 and other sirtuins in metabolism. Trends Endocrinol Metab 25(3):138–145
Bouras T, Fu M, Sauve AA, Wang F, Quong AA, Perkins ND et al (2005) SIRT1 deacetylation and repression of p300 involves lysine residues 1020/1024 within the cell cycle regulatory domain 1. J Biol Chem 280(11):10264–10276
Guarente L (2013) Introduction: sirtuins in aging and diseases. Springer, Sirtuins, pp 3–10
Verdin E, Hirschey MD, Finley LW, Haigis MC (2010) Sirtuin regulation of mitochondria: energy production, apoptosis, and signaling. Trends Biochem Sci 35(12):669–675
Xu D, He H, Jiang X, Hua R, Chen H, Yang L et al (2019) SIRT2 plays a novel role on progesterone, estradiol and testosterone synthesis via PPARs/LXRα pathways in bovine ovarian granular cells. J Steroid Biochem Mol Biol 185:27–38
Firestein R, Blander G, Michan S, Oberdoerffer P, Ogino S, Campbell J, et al. The SIRT1 deacetylase suppresses intestinal tumorigenesis and colon cancer growth. PloS One. 2008;3(4).
Wang R-H, Sengupta K, Li C, Kim H-S, Cao L, Xiao C et al (2008) Impaired DNA damage response, genome instability, and tumorigenesis in SIRT1 mutant mice. Cancer Cell 14(4):312–323
Bindu S, Pillai VB, Gupta MP (2016) Role of sirtuins in regulating pathophysiology of the heart. Trends Endocrinol Metab 27(8):563–573
Yacoub R, Lee K, He JC (2014) The role of SIRT1 in diabetic kidney disease. Front Endocrinol 5:166
Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B et al (2004) Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science (New York, NY) 305(5682):390–392
Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P (2005) Nutrient control of glucose homeostasis through a complex of PGC-1α and SIRT1. Nature 434(7029):113–118
Bordone L, Cohen D, Robinson A, Motta MC, Van Veen E, Czopik A et al (2007) SIRT1 transgenic mice show phenotypes resembling calorie restriction. Aging Cell 6(6):759–767
Kume S, Uzu T, Horiike K, Chin-Kanasaki M, Isshiki K, Araki S-i, et al. Calorie restriction enhances cell adaptation to hypoxia through Sirt1-dependent mitochondrial autophagy in mouse aged kidney. The Journal of clinical investigation. 2010;120(4):1043–55.
Guarente L, Picard F (2005) Calorie restriction—the SIR2 connection. Cell 120(4):473–482
Blander G, Guarente L (2004) The Sir2 family of protein deacetylases. Annu Rev Biochem 73(1):417–435
Imai S-i (2009) The NAD World: a new systemic regulatory network for metabolism and aging—Sirt1, systemic NAD biosynthesis, and their importance. Cell Biochem Biophys 53(2):65
Houtkooper RH, Pirinen E, Auwerx J (2012) Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol 13(4):225–238
Haigis MC, Sinclair DA (2010) Mammalian sirtuins: biological insights and disease relevance. Annu Rev Pathol 5:253–295
Gu X, Wang Z, Ye Z, Lei J, Li L, Su D et al (2014) Resveratrol, an activator of SIRT1, upregulates AMPK and improves cardiac function in heart failure. Genet Mol Res 13(1):323–335
Das DK, Maulik N (2006) Resveratrol in cardioprotection: a therapeutic promise of alternative medicine. Mol Interventions 6(1):36
Labinskyy N, Csiszar A, Veress G, Stef G, Pacher P, Oroszi G et al (2006) Vascular dysfunction in aging: potential effects of resveratrol, an anti-inflammatory phytoestrogen. Curr Med Chem 13(9):989–996
Mattagajasingh I, Kim C-S, Naqvi A, Yamamori T, Hoffman TA, Jung S-B et al (2007) SIRT1 promotes endothelium-dependent vascular relaxation by activating endothelial nitric oxide synthase. Proc Natl Acad Sci 104(37):14855–14860
Stef G, Csiszar A, Lerea K, Ungvari Z, Veress G (2006) Resveratrol inhibits aggregation of platelets from high-risk cardiac patients with aspirin resistance. J Cardiovasc Pharmacol 48(2):1–5
Colak Y, Ozturk O, Senates E, Tuncer I, Yorulmaz E, Adali G, et al. SIRT1 as a potential therapeutic target for treatment of nonalcoholic fatty liver disease. Medical science monitor: international medical journal of experimental and clinical research. 2011;17(5):HY5.
Deng XQ, Chen LL, Li NX (2007) The expression of SIRT1 in nonalcoholic fatty liver disease induced by high-fat diet in rats. Liv Int 27(5):708–715
Chen L, Deng X, Li N (2007) Effects of calorie restriction on SIRT1 expression in liver of nonalcoholic fatty liver disease: experiment with rats. Zhonghua yi xue za zhi 87(20):1434–1437
Imai S-I, Armstrong CM, Kaeberlein M, Guarente L (2000) Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403(6771):795–800
Vaziri H, Dessain SK, Eaton EN, Imai S-I, Frye RA, Pandita TK et al (2001) hSIR2SIRT1 functions as an NAD-dependent p53 deacetylase. Cell 107(2):149–159
Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA et al (2004) Modulation of NF-κB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23(12):2369–2380
Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y et al (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science (New York, NY) 303(5666):2011–2015
Motta MC, Divecha N, Lemieux M, Kamel C, Chen D, Gu W et al (2004) Mammalian SIRT1 represses forkhead transcription factors. Cell 116(4):551–563
Li X, Zhang S, Blander G, Jeanette GT, Krieger M, Guarente L (2007) SIRT1 deacetylates and positively regulates the nuclear receptor LXR. Mol Cell 28(1):91–106
Grimaldi B, Nakahata Y, Kaluzova M, Masubuchi S, Sassone-Corsi P (2009) Chromatin remodeling, metabolism and circadian clocks: the interplay of CLOCK and SIRT1. Int J Biochem Cell Biol 41(1):81–86
Nakahata Y, Kaluzova M, Grimaldi B, Sahar S, Hirayama J, Chen D et al (2008) The NAD+-dependent deacetylase SIRT1 modulates CLOCK-mediated chromatin remodeling and circadian control. Cell 134(2):329–340
Yamazaki Y, Usui I, Kanatani Y, Matsuya Y, Tsuneyama K, Fujisaka S et al (2009) Treatment with SRT1720, a SIRT1 activator, ameliorates fatty liver with reduced expression of lipogenic enzymes in MSG mice. Am J Physiol-Endocrinol Metab 297(5):E1179–E1186
Dong Y-j, Liu N, Xiao Z, Sun T, Wu S-h, Sun W-x, et al. Renal protective effect of sirtuin 1. Journal of diabetes research. 2014;2014.
Chen K-H, Hung C-C, Hsu H-H, Jing Y-H, Yang C-W, Chen J-K (2011) Resveratrol ameliorates early diabetic nephropathy associated with suppression of augmented TGF-β/smad and ERK1/2 signaling in streptozotocin-induced diabetic rats. Chem Biol Interact 190(1):45–53
Xu Y, Nie L, Yin Y-G, Tang J-L, Zhou J-Y, Li D-D et al (2012) Resveratrol protects against hyperglycemia-induced oxidative damage to mitochondria by activating SIRT1 in rat mesangial cells. Toxicol Appl Pharmacol 259(3):395–401
Fan H, Yang H-C, You L, Wang Y-Y, He W-J, Hao C-M (2013) The histone deacetylase, SIRT1, contributes to the resistance of young mice to ischemia/reperfusion-induced acute kidney injury. Kidney Int 83(3):404–413
Simic P, Williams EO, Bell EL, Gong JJ, Bonkowski M, Guarente L (2013) SIRT1 suppresses the epithelial-to-mesenchymal transition in cancer metastasis and organ fibrosis. Cell Rep 3(4):1175–1186
Ohse T, Vaughan MR, Kopp JB, Krofft RD, Marshall CB, Chang AM et al (2010) De novo expression of podocyte proteins in parietal epithelial cells during experimental glomerular disease. American Journal of Physiology-Renal Physiology 298(3):F702–F711
Zhang J, Hansen KM, Pippin JW, Chang AM, Taniguchi Y, Krofft RD et al (2012) De novo expression of podocyte proteins in parietal epithelial cells in experimental aging nephropathy. Am J Physiol-Renal Physiol 302(5):F571–F580
Lin Q, Geng Y, Lin S, Tian Z (2016) Sirtuin1 (SIRT1) regulates tumor necrosis factor-alpha (TNF-α-Induced) aquaporin-2 (AQP2) expression in renal medullary collecting duct cells through inhibiting the NF-κB pathway. Med Sci Monitor Basic Res 22:165
Kim DH, Jung YJ, Lee JE, Lee AS, Kang KP, Lee S et al (2011) SIRT1 activation by resveratrol ameliorates cisplatin-induced renal injury through deacetylation of p53. Am J Physiol-Renal Physiol 301(2):F427–F435
Chuang PY, Dai Y, Liu R, He H, Kretzler M, Jim B, et al. Alteration of forkhead box O (foxo4) acetylation mediates apoptosis of podocytes in diabetes mellitus. PLoS One. 2011;6(8).
Miyazaki R, Ichiki T, Hashimoto T, Inanaga K, Imayama I, Sadoshima J et al (2008) SIRT1, a longevity gene, downregulates angiotensin II type 1 receptor expression in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 28(7):1263–1269
Gao R, Chen J, Hu Y, Li Z, Wang S, Shetty S, et al. Sirt1 deletion leads to enhanced inflammation and aggravates endotoxin-induced acute kidney injury. PLoS One. 2014;9(6).
Hayden MS, Ghosh S (2008) Shared principles in NF-κB signaling. Cell 132(3):344–362
Wu H, Kong L, Zhou S, Cui W, Xu F, Luo M, et al. The role of microRNAs in diabetic nephropathy. Journal of diabetes research. 2014;2014.
Shimo T, Adachi Y, Yamanouchi S, Tsuji S, Kimata T, Umezawa K et al (2013) A novel nuclear factor κB inhibitor, dehydroxymethylepoxyquinomicin, ameliorates puromycin aminonucleoside-induced nephrosis in mice. Am J Nephrol 37(4):302–309
Jung YJ, Lee JE, Lee AS, Kang KP, Lee S, Park SK et al (2012) SIRT1 overexpression decreases cisplatin-induced acetylation of NF-κB p65 subunit and cytotoxicity in renal proximal tubule cells. Biochem Biophys Res Commun 419(2):206–210
Matsushita N, Takami Y, Kimura M, Tachiiri S, Ishiai M, Nakayama T et al (2005) Role of NAD-dependent deacetylases SIRT1 and SIRT2 in radiation and cisplatin-induced cell death in vertebrate cells. Genes Cells 10(4):321–332
Hasegawa K, Wakino S, Yoshioka K, Tatematsu S, Hara Y, Minakuchi H et al (2010) Kidney-specific overexpression of Sirt1 protects against acute kidney injury by retaining peroxisome function. J Biol Chem 285(17):13045–13056
Kaushal GP (2012) Autophagy protects proximal tubular cells from injury and apoptosis. Kidney Int 82(12):1250–1253
Lee IH, Cao L, Mostoslavsky R, Lombard DB, Liu J, Bruns NE et al (2008) A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proc Natl Acad Sci 105(9):3374–3379
Ginsburg ES, Owen Jr WF, editors. Reproductive Endocrinology and Pregnancy in Women on Hemodialysis: Endocrine and Metabolic Function in Renal Failure Garabed Eknoyan, Series Editor. Seminars in Dialysis; 1993: Wiley Online Library.
Ahmed SB, Ramesh S (2016) Sex hormones in women with kidney disease. Nephrol Dial Transpl 31(11):1787–1795
Holley JL, Schmidt RJ, Bender FH, Dumler F, Schiff M (1997) Gynecologic and reproductive issues in women on dialysis. Am J Kidney Dis 29(5):685–690
Anderson GD, Odegard PS (2004) Pharmacokinetics of estrogen and progesterone in chronic kidney disease. Adv Chronic Kidney Dis 11(4):357–360
Mercantepe T, Unal D, Selli J, Mercantepe F, Unal B, Karabiyik TN (2016) Protective effects of estrogen and bortezomib in kidney tissue of post-menopausal rats: an ultrastructural study. Ren Fail 38(7):1129–1135
Neugarten J, Golestaneh L (2013) Gender and the prevalence and progression of renal disease. Adv Chronic Kidney Dis 20(5):390–395
Nielsen CB, Flyvbjerg A, Bruun JM, Forman A, Wogensen L, Thomsen K (2003) Decreases in renal functional reserve and proximal tubular fluid output in conscious oophorectomized rats: normalization with sex hormone substitution. J Am Soc Nephrol 14(12):3102–3110
Gross M-L, Adamczak M, Rabe T, Harbi NA, Krtil J, Koch A et al (2004) Beneficial effects of estrogens on indices of renal damage in uninephrectomized SHRsp rats. J Am Soc Nephrol 15(2):348–358
Potier M, Elliot SJ, Tack I, Lenz O, Striker GE, Striker LJ et al (2001) Expression and regulation of estrogen receptors in mesangial cells: influence on matrix metalloproteinase-9. J Am Soc Nephrol 12(2):241–251
Naderi V, Khaksari M, Abbasi R, Maghool F (2015) Estrogen provides neuroprotection against brain edema and blood brain barrier disruption through both estrogen receptors α and β following traumatic brain injury. Iran J Basic Med Sci 18(2):138
Khaksari M, Abbasloo E, Dehghan F, Soltani Z, Asadikaram G (2015) The brain cytokine levels are modulated by estrogen following traumatic brain injury: Which estrogen receptor serves as modulator? Int Immunopharmacol 28(1):279–287
Mankhey RW, Bhatti F, Maric C (2005) 17β-Estradiol replacement improves renal function and pathology associated with diabetic nephropathy. Am J Physiol-Renal Physiol 288(2):F399–F405
Metcalfe PD, Meldrum KK (2006) Sex differences and the role of sex steroids in renal injury. J Urol 176(1):15–21
Neugarten J, Acharya A, Silbiger SR (2000) Effect of gender on the progression of nondiabetic renal disease: a meta-analysis. J Am Soc Nephrol 11(2):319–329
Silbiger SR, Neugarten J (2003) The role of gender in the progression of renal disease. Adv Ren Replace Ther 10(1):3–14
Cherney DZ, Sochett EB, Miller JA (2005) Gender differences in renal responses to hyperglycemia and angiotensin-converting enzyme inhibition in diabetes. Kidney Int 68(4):1722–1728
Wells CC, Riazi S, Mankhey RW, Bhatti F, Ecelbarger C, Maric C (2005) Diabetic nephropathy is associated with decreasedcirculating estradiol levels and imbalance in the expression of renal estrogen receptors. Gend Med 2(4):227–237
Mankhey RW, Wells CC, Bhatti F, Maric C (2007) 17β-Estradiol supplementation reduces tubulointerstitial fibrosis by increasing MMP activity in the diabetic kidney. Am J Physiol-Regulatory Integrative Comparative Physiol 292(2):R769–R777
Blush J, Lei J, Ju W, Silbiger S, Pullman J, Neugarten J (2004) Estradiol reverses renal injury in Alb/TGF-β1 transgenic mice. Kidney Int 66(6):2148–2154
Dean S, Tan J, O'Brien E, Leenen F (2005) 17beta-estradiol downregulates tissue 1046 angiotensin-converting enzyme and ANG II type 1 receptor in female rats. Am J Physiol Regul 1047 Integr Comp Physiol. 288(R759–766):1048.
Maric C, Sullivan S (2008) Estrogens and the diabetic kidney. Gend Med 5:S103–S113
Xiao S, Gillespie DG, Baylis C, Jackson EK, Dubey RK (2001) Effects of estradiol and its metabolites on glomerular endothelial nitric oxide synthesis and mesangial cell growth. Hypertension 37(2):645–650
Neugarten J, Acharya A, Lei J, Silbiger S (2000) Selective estrogen receptor modulators suppress mesangial cell collagen synthesis. Am J Physiol-Renal Physiol 279(2):F309–F318
Satake A, Takaoka M, Nishikawa M, Yuba M, Shibata Y, Okumura K et al (2008) Protective effect of 17β-estradiol on ischemic acute renal failure through the PI3K/Akt/eNOS pathway. Kidney Int 73(3):308–317
Lecour S, James RW (2011) When are pro-inflammatory cytokines SAFE in heart failure? Eur Heart J 32(6):680–685
Wang M, Wang Y, Abarbanell A, Tan J, Weil B, Herrmann J et al (2009) Both endogenous and exogenous testosterone decrease myocardial STAT3 activation and SOCS3 expression after acute ischemia and reperfusion. Surgery 146(2):138–144
Sehara Y, Sawicka K, Hwang J-Y, Latuszek-Barrantes A, Etgen AM, Zukin RS (2013) Survivin is a transcriptional target of STAT3 critical to estradiol neuroprotection in global ischemia. J Neurosci 33(30):12364–12374
Diaz MN, Rodriguez AR, Martin JF, Arias MS, Rodriguez PM, Andia JC (2007) Effects of estradiol, calcitriol and both treatments combined on bone histomorphometry in rats with chronic kidney disease and ovariectomy. Bone 41(4):614–619
Yap FC, Taylor MS, Lin MT (2014) Ovariectomy-induced reductions in endothelial SK3 channel activity and endothelium-dependent vasorelaxation in murine mesenteric arteries. PLoS ONE 9(8):e104686
Singh AP, Singh N, Bedi PMS (2016) Estrogen attenuates renal IRI through PPAR-γ agonism in rats. J Surg Res 203(2):324–330
Margueron R, Duong V, Castet A, Cavaillès V (2004) Histone deacetylase inhibition and estrogen signalling in human breast cancer cells. Biochem Pharmacol 68(6):1239–1246
Reid G, Metivier R, Lin C-Y, Denger S, Ibberson D, Ivacevic T et al (2005) Multiple mechanisms induce transcriptional silencing of a subset of genes, including oestrogen receptor α, in response to deacetylase inhibition by valproic acid and trichostatin A. Oncogene 24(31):4894–4907
Liarte S, Alonso-Romero JL, Nicolás FJ (2018) SIRT1 and estrogen signaling cooperation for breast cancer onset and progression. Front Endocrinol 9:552
Yao Y, Li H, Gu Y, Davidson NE, Zhou Q (2010) Inhibition of SIRT1 deacetylase suppresses estrogen receptor signaling. Carcinogenesis 31(3):382–387
Moore R, Dai Y, Faller D (2012) Sirtuin 1 (SIRT1) and steroid hormone receptor activity in cancer. J Endocrinol 213(1):37
Sasaki Y, Ikeda Y, Miyauchi T, Uchikado Y, Akasaki Y, Ohishi M. Estrogen-SIRT1 axis plays a pivotal role in protecting arteries against menopause-induced senescence and atherosclerosis. J Atherosclerosis Thrombosis. 2019:47993.
Elangovan S, Ramachandran S, Venkatesan N, Ananth S, Gnana-Prakasam JP, Martin PM et al (2011) SIRT1 is essential for oncogenic signaling by estrogen/estrogen receptor α in breast cancer. Can Res 71(21):6654–6664
Guo JM, Shu H, Wang L, Xu JJ, Niu XC, Zhang L (2017) SIRT 1-dependent AMPK pathway in the protection of estrogen against ischemic brain injury. CNS Neurosci Ther 23(4):360–369
Zheng Y, Hu Q, Manaenko A, Zhang Y, Peng Y, Xu L et al (2015) 17β-Estradiol attenuates hematoma expansion through ERα/Sirt1/NF-κB pathway in hyperglycemic intracerebral hemorrhage mice. Stroke 46(2):485
Shen T, Ding L, Ruan Y, Qin W, Lin Y, Xi C, et al. SIRT1 functions as an important regulator of estrogen-mediated cardiomyocyte protection in angiotensin II-induced heart hypertrophy. Oxidative medicine and cellular longevity. 2014;2014.
Zuo H-L, Xin H, Yan X-N, Huang J, Zhang Y-P, Du H. 17β-Estradiol improves osteoblastic cell function through the Sirt1/NF-κB/MMP-8 pathway. Climacteric. 2020:1–6.
Khan M, Ullah R, Rehman SU, Shah SA, Saeed K, Muhammad T et al (2019) 17β-Estradiol Modulates SIRT1 and halts oxidative stress-mediated cognitive impairment in a male aging mouse model. Cells 8(8):928
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Darvishzadeh Mahani, F., Khaksari, M. & Raji-amirhasani, A. Renoprotective effects of estrogen on acute kidney injury: the role of SIRT1. Int Urol Nephrol 53, 2299–2310 (2021). https://doi.org/10.1007/s11255-020-02761-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-020-02761-y