Abstract
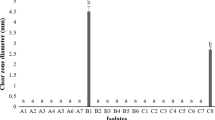
Citrus Bacterial Canker disease, caused by Xanthomonas citri subsp. citri (Xcc), is one of the most devastating diseases that attacks citrus, especially in the southern areas of Iran. The objective of this investigation was to analyze several characteristics involved in Xcc virulence in relation to strain aggressiveness. To achieve this, swarming motility, biofilm formation, resistance to H2O2, and production of xanthan were evaluated considering 44 local strains causing citrus bacterial canker disease in Iran. All strains showed differential swarming motilities, biofilm formation abilities, and xanthan production. The most virulent strain, Xcc-KVXCC1, exhibited the greatest capability to form biofilm on solid surfaces, xanthan production, and pathogenicity on detached leaves of Citrus aurantifolia as well as positive chemotaxis toward C. aurantifolia extract. In this study, a total of 14 common and commercial citrus genotypes were evaluated for resistance to Xcc-KVXCC1. Genotypes were categorized into susceptible and resistant groups based on lesion number per inoculation site. Based on our results, C. aurantifolia was the most susceptible citrus cultivar to Xcc-KVXCC1. In contrast, C. aurantium, C. jumbhori, and C. reticulata cv. ponkan demonstrated high and moderately high resistance against Xcc-KVXCC1.







Similar content being viewed by others
References
Alizadeh A, Rahimian H (1990) Citrus canker in Kerman province. Iranian J Plant Pathol 26:42
Al-Saleh MA, Widyawan A, Saleh AA, Ibrahim YE (2014) Distribution and pathotype identification of Xanthomonas citri subsp. citri recovered from south-western region of Saudi Arabia. Afr J Microbiol Res 8:673–679
Amaral A, Carvalho S, Silva L, Machado M (2010) Reaction of genotypes of Citrus species and varieties to Xanthomonas citri subsp. citri under greenhouse conditions. Plant Pathol 92:519–524
Antúnez-Lamas M, Cabrera-Ordonez E, Lopez-Solanilla E, Raposo R, Trelles-Salazar O, Rodríguez-Moreno A, Rodríguez-Palenzuela P (2009) Role of motility and chemotaxis in the pathogenesis of Dickeya dadantii 3937 (ex Erwinia chrysanthemi 3937). J Microbiol 155:434–442
Bogino PC, Oliva Mde L, Sorroche FG, Giordano W (2013) The role of bacterial biofilms and surface components in plant-bacterial associations. Int J Mol 14:15838–15859
Carignatto CRR, Oliveira KSM, de Lima VMG, de Oliva NP (2011) New culture medium to xanthan production by Xanthomonas campestris pv. campestris. Indian J Med Microbiol 51:283–288
Caswell CC, Baumgartner JE, Martin DW, Roop RM (2012) Characterization of the organic hydroperoxide resistance system of Brucella abortus 2308. J Bacteriol Res 194:5065–5072
Chidamba L, Bezuidenhout CC (2012) Characterisation of Xanthomonas campestris pv. Campestris isolates from South Africa using genomic DNA fingerprinting and pathogenicity tests. Eur J Plant Pathol 133:811–818
Cho JH, Jeong KS, Han JW, Kim WJ, Cha JS (2011) Mutation in clp xoo4158 reduces virulence and resistance to oxidative stress in Xanthomonas oryzae pv. oryzae KACC10859. Plant Pathol J 27:89–92
Civerolo E (1982) Citrus bacterial canker disease: an overview. Pages 390–394. In: Proceedings of the international society of citriculture, November 9–12, 1981, Tokyo, Japan. Vol. 1. International Society of Citriculture
Da Silva A, Ferro J, Reinach F et al (2002) Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature 417:459–463
Das A (2003) Citrus canker-A review J Appl Hortic 5:52–60
de Carvalho SA, de Carvalho Nunes WM, JrJ B, Machado MA, Croce-Filho J, Bock CH, Abdo Z (2015) Comparison of resistance to asiatic citrus canker among different genotypes of citrus in a long-term canker-resistance field screening experiment in Brazil. Plant Dis 99:207–218
Deng Z, Xu L, Li D, Long G, Liu L, Fang F, Shu G (2010) Screening citrus genotypes for resistance to canker disease (Xanthomonas axonopodis pv. citri). Plant Breed 129:341–345
Dunger G, Relling VM, Tondo ML, Barreras M, Ielpi L, Orellano EG, Ottado J (2007) Xanthan is not essential for pathogenicity in citrus canker but contributes to Xanthomonas epiphytic survival. Arch Microbiol 188:127–135
Ference CM, Gochez AM, Behlau F, Wang N, Graham JH, Jones JB (2018) Recent advances in the understanding of Xanthomonas citri ssp. citri pathogenesis and citrus canker disease management. Mol Plant Pathol 19:1302–1318
Fonseca NP, Patané JSL, Varani AM, Felestrino ÉB, Caneschi WL, Sanchez AB, Cordeiro IF, Lemes CGdC, Assis RdAB, Garcia CCM, Belasque J Jr, Martins J Jr, Facincani AP, Ferreira RM, Jaciani FJ, Almeida NFd, Ferro JA, Moreira LM, Setubal JC (2019) Analyses of Seven New Genomes of Xanthomonas citri pv. aurantifolii Strains, Causative Agents of Citrus Canker B and C, Show a Reduced Repertoire of Pathogenicity-Related Genes. Front Microbiol 10:2361
Francis MI, Kostenyuk I, Orbović V, Loskutov A, Zolotukhin M, Graham JH (2011) Automated needle-free injection method for delivery of bacterial suspensions into citrus leaf tissues. J Phytopathol 159:347–351
Garcıa-Ochoa F, Santos V, Casas J, Gomez E (2000) Xanthan gum: production, recovery, and properties. Biotechnol Adv 18:549–579
Gottig N, Garavaglia BS, Garofalo CG, Zimaro T, Sgro GG, Ficarra FA, Dunger G, Daurelio LD, Thomas L, Gehring C, Orellano EG, Ottado J (2010) Mechanisms of infection used by Xanthomonas axonopodis pv. citri in citrus canker disease. Current Research, Technology and Education Topics in Applied Microbiology and Microbial Biotechnology 196–204
Gottwald T, Graham J, Civerolo E, Barrett H, Hearn C (1993) Differential host range reaction of citrus and citrus relatives to citrus canker and citrus bacterial spot determined by leaf mesophyll susceptibility. Plant Dis 77:1004–1009
Garita-Cambronero J, Sena-Vélez M, Ferragud E, Sabuquillo P, Redondo C, Cubero J (2019) Xanthomonas citri subsp. citri and Xanthomonas arboricola pv. pruni: Comparative analysis of two pathogens producing similar symptoms in different host plants. PLoS One 14(7):e0219797
Heindl JE, Wang Y, Heckel BC, Mohari B, Feirer N, Fuqua C (2014) Mechanisms and regulation of surface interactions and biofilm formation in Agrobacterium. Front Plant Sci 5:176
Hyun JW, Myung IS, Lee SC, Kim KS, Lim HC (2003) Evaluation of potential of mandarin hybrid “Shiranuhi” against inoculation of bacterial canker disease pathogen (Xanthomonas axonopodis pv. citri) in citrus field in Jeju Island. Plant Pathol J 19:248–252
Ibrahim YE, El Komy MH, Amer MA et al (2019) Difficulties in identifying Xanthomonas citri subsp. citri A pathotypes. J Plant Pathol 101:927–933
Izadiyan M, Taghavi SM, Farahbakhsh F (2018) Characterization of Xanthomonas citri subsp. CITRI isolated from grapefruit in Iran. J Plant Pathol 100:257–267
Izadiyan M, Taghavi SM (2020) Isolation and characterization of the citrus canker pathogen Xanthomonas citri subsp. citri pathotype A, occurring in imported tangerine (Citrus reticulata Blanco) fruits. J Plant Pathol 102:671–679
Khalaf A, Moore GA, Jones JB, Gmitter FG (2007) New insights into the resistance of Nagami kumquat to canker disease. Phisoil Mol Plant P 71:240–250
Khodakaramian G, Swings J (2011) Genetic diversity and pathogenicity of Xanthomonas axonopodis strains inducing citrus canker disease in Iran and South Korea. Indian J Med Microbiol 51:194–199
Kim S, Cho YJ, Song ES, Lee SH, Kim JG, Kang LW (2016) Time-resolved pathogenic gene expression analysis of the plant pathogen Xanthomonas oryzae pv. oryzae. BMC genomics 17:345
Leduc A, Traoré Y, Boyer K, Magne M, Grygiel P, Juhasz C, Boyer C, Guerin F, Wonni I, Ouedraogo L (2015) Bridgehead invasion of a monomorphic plant pathogenic bacterium: Xanthomonas citri pv. citri, an emerging citrus pathogen in Mali and Burkina Faso. Environ Microbiol 17:4429–4442
Li Y, Yu Q (2020) Role of the lipoprotein VacJ/MlaA in the physiology and virulence of the plant pathogen Xanthomonas citri subsp. Citri. Physiol Mol Plant P 112:101546
Louws FJ, Fulbright DW, Stephens CT, De Bruijn F (1994) Specific genomic fingerprints of phytopathogenic Xanthomonas and Pseudomonas pathovars and strains generated with repetitive sequences and PCR. Appl Environ Microbiol 60:2286–2295
Malamud F, Torres PS, Roeschlin R, Rigano LA, Enrique R, Bonomi HR, Castagnaro AP, Marano MR, Vojnov AA (2011) The Xanthomonas axonopodis pv. citri flagellum is required for mature biofilm and canker development. J Microbiol 157:819–829
Mustafa M, Imran M, Rasool A, Azeem M, Riaz A, Afzal M (2014) Evaluation of commercial citrus cultivars for resistance to citrus leaf miner and its management. J Entomol 2:213–216
O'Toole GA (2011) Microtiter dish biofilm formation assay. J Vis Exp e2437-e2437
Overhage J, Lewenza S, Marr AK, Hancock RE (2007) Identification of genes involved in swarming motility using a Pseudomonas aeruginosa PAO1 mini-Tn5-lux mutant library. J Bacteriol 189:2164–2169
Palleroni NJ (1976) Chamber for bacterial chemotaxis experiments. Appl Environ Microbiol 32:729–730
Park DS, Hyun JW, Park YJ, Kim JS, Kang HW, Hahn JH, Go SJ (2006) Sensitive and specific detection of Xanthomonas axonopodis pv. citri by PCR using pathovar specific primers based on hrpW gene sequences. Microbiol Res 161:145–149
Pitino M, Armstrong C, Duan P (2015) Rapid screening for citrus canker resistance employing pattern-triggered immunity (PTI) responses. Hortic Res 2:1–8
Poplawsky A, Urban S, Chun W (2000) Biological role of xanthomonadin pigments in Xanthomonas campestris pv. campestris. Appl Environ Microbiol 66:5123–5127
Pruvost O, Magne M, Boyer K, Leduc A, Tourterel C et al (2014) A MLVA genotyping scheme for global surveillance of the citrus pathogen Xanthomonas citri pv. citri suggests a worldwide geographical expansion of a single genetic lineage. PLoS One 9:e98129
Pruvost O, Goodarzi T, Boyer K, Soltaninejad H, Escalon A, Alavi S, Javegny S, Boyer C, Cottyn B, Gagnevin L (2015) Genetic structure analysis of strains causing citrus canker in Iran reveals the presence of two different lineages of Xanthomonas citri pv. citri pathotype A. Plant Pathol 64:776–784
Reddy M (1997) Sources of resistance to bacterial canker in citrus. J Mycol Plant Pathol 27:80–81
Rezaei M, Shams-Bakhsh M, Alizadeh A (2012) Genetic diversity among Xanthomonas citri subsp. citri strains in Iran. J Plant Prot Res 52:1–9
Rigano LA, Siciliano F, Enrique R, Sendín L, Filippone P, Torres PS, Qüesta J, Dow JM, Castagnaro AP, Vojnov AA (2007) Biofilm formation, epiphytic fitness, and canker development in Xanthomonas axonopodis pv. citri. Mol Plant Microbe Interact 20:1222–1230
Rohlf F (2002) NTSYSpc: Numerical taxonomy system, ver. 2.1. Setauket, NY: Exeter Publishing Ltd.
Schmidt J, Müsken M, Becker T, Magnowska Z, Bertinetti D, Möller S et al (2011) The Pseudomonas aeruginosa Chemotaxis Methyltransferase CheR1 Impacts on Bacterial Surface Sampling. PLoS One 6(3):e18184
Sena-Velez M, Redondo C, Gell I, Ferragud E, Johnson E, Graham JH et al (2015) Biofilm formation and motility of Xanthomonas strains with different citrus host range. Plant Pathol 64:767–775
Sena-Velez M, Redondo C, Graham JH, Cubero J (2016) Presence of extracellular DNA during biofilm formation by Xanthomonas citri subsp citri strains with different host range. PLoS One 11:e0156695
Schaad NW, Jones JB, Chun W (2001) Laboratory guide for Identification of plant pathogenic bacteria. APS Press, St. Paul, Minnesota, USA
Song X, Guo J, Ma W et al (2015) Identification of seven novel virulence genes from Xanthomonas citri subsp. citri by Tn5-based random mutagenesis. J Microbiol 53:330–336
Stover E, Driggers R, Richardson ML, Hall DG, Duan Y, Lee RF (2014) Incidence and severity of asiatic citrus canker on diverse citrus and citrus-related germplasm in a Florida field planting. HortScience 49:4–9
Tondo ML, Delprato ML, Kraiselburd I, Fernández Zenoff MV, Farías ME, Orellano EG (2016) KatG, the Bifunctional Catalase of Xanthomonas citri subsp. citri, Responds to Hydrogen Peroxide and Contributes to Epiphytic Survival on Citrus Leaves. PLoS One 11(3):e0151657
Viloria Z, Drouillard D, Graham J, Grosser J (2004) Screening triploid hybrids of “Lakeland” limequat for resistance to citrus canker. Plant Dis 88:1056–1060
Ye G, Hong N, Zou LF, Zou HS, Zakria M, Wang GP, Chen GY (2013) tale-based genetic diversity of Chinese isolates of the citrus canker pathogen Xanthomonas citri subsp. citri. Plant Dis 97:1187–1194
Yousefi Koupaei F, Taghavi SM, Shiotani H, Tsuyumu S (2014) A PCRbased assay for differentiating A- and A*-type strains of Xanthomonas citri subsp. citri, the causal agent of Asiatic citrus canker. J Gene Plant Pathol 80:85–89
Yun MH, Torres PS, El Oirdi M, Rigano LA, Gonzalez-Lamothe R, Marano MR, Castagnaro AP, Dankert MA, Bouarab K, Vojnov AA (2006) Xanthan induces plant susceptibility by suppressing callose deposition. Plant Physiol 141:178–187
Zimaro T, Thomas L, Marondedze C, Garavaglia BS, Gehring C, Ottado J, Gottig N (2013) Insights into Xanthomonas axonopodis pv. citri biofilm through proteomics. BMC microbiol 13:186
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
There is no conflict of interest by the co-authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mirzaei-Najafgholi, H., Aeini, M., Tarighi, S. et al. Comparing bacterial properties in relation to the virulence factors of Xanthomonas citri subsp. citri strains and evaluating resistance of subtribe Citrinae cultivars to the most virulent strain. J Plant Pathol 103, 449–460 (2021). https://doi.org/10.1007/s42161-021-00760-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-021-00760-1