Abstract
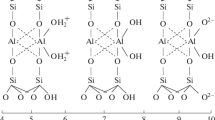
The mechanism of adsorption of anionic dyes on montmorillonite modified with sodium metasilicate is investigated. The kinetics of dye adsorption is approximated by a pseudo–second order model, and the adsorption isotherm is analyzed on the basis of the Langmuir and Dubinin–Radushkevich models. The value of average free energy of adsorption E > 8 kJ/mol for all studied dyes, testifying to a surface chemical reaction between dye molecules and protonated silanol and aluminol groups on the surface of the edges of the aluminosilicate layers of the mineral. The adsorption of dyes is directly proportional to the increase in the temperature, mass, and charge of the organic anion and is inversely proportional to the increase in the ionic strength of the solution.





Similar content being viewed by others
REFERENCES
M. F. Elahi, J. Innov. Dev. Strategy 5, 9 (2011).
B. de Campos Ventura-Camargo and M. A. Marin-Morales, TLIST 2, 85 (2013).
F. Bergayaa, G. Lagaly, and M. Vayer, in Handbook of Clay Science, Ed. by F. Bergaya, B. K. G. Theng, and G. Lagaly (Elsevier, Amsterdam, 2006), Vol. 1, p. 979.
B. Ali Fil, C. Özmetin, and M. Korkmaz, Bull. Korean Chem. Soc. 33, 3184 (2012).
P. Sharma, D. J. Borah, P. Das, et al., J. Desalin. Water Treatm. 57, 8372 (2015).
D. Bendaho, T. Ainad, and D. Djillali, J. Environ. Anal. Chem. 2 (2015). https://doi.org/10.4172/2380-2391.1000130
M. M. F. Silva, M. M. Oliveira, M. C. Avelino, et al., Chem. Eng. J. 203, 259 (2012).
T. V. Kon’kova, A. P. Rysev, and E. V. Mishchenko, Persp. Mater., No. 1, 13 (2020).
M. A. Akl, A. M. Youssef, and M. M. Al-Awadhi, J. Anal. Bioanal. Tech. 4 (2013). https://doi.org/10.4172/2155-9872.1000174
A. S. Ozcan, B. Erdem, and A. Ozcan, Thin Solid Films 280, 44 (2004).
B. Muir, D. Andrunik, J. Hyla, et al., Appl. Clay Sci. 136, 8 (2017).
T. V. Kon’kova and A. P. Rysev, Colloid. J. 82, 130 (2020).
L. Ammann, F. Bergaya, and G. Lagaly, Clay Miner. 40, 441 (2005).
Y. S. Ho and G. McKay, Process Biochem. 34, 451 (1999).
O. O. Krizhanovskaya, L. A. Sinyaeva, S. I. Karpov, et al., Sorbtsion. Khromatogr. Protsessy 14, 784 (2014).
O. V. Lebedeva, E. I. Sipkina, and Yu. N. Pozhidaev, Prot. Met. Phys. Chem. Surf. 53, 80 (2017).
M. A. Shumilova and V. G. Petrov, Teor. Prikl. Ekol., No. 4, 32 (2017).
B. Sarkara, Y. Xia, M. Megharaja, et al., J. Hazard. Mater. 183, 87 (2010).
A. D. Zimon, Colloid Chemistry, The General Course (KRASAND, Moscow, 2015) [in Russian].
T. Theophanides, Infrared Spectroscopy - Materials Science, Engineering, and Technology (InTech., Rijeka, Croatia, 2012), p. 524.
J. Temuujin, Ts. Jadamba, G. Burma, et al., Ceram. Int. 30, 251 (2004).
The Science of Clays Applications in Industry, Engineering, and Environment, Ed. by S. Mukherjee (Springer, Kolkata, 2013), p. 351.
M. Erdemoglu, S. Erdemoglu, F. Sayllkan, et al., Appl. Clay Sci. 27, 41 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by D. Kharitonov
Rights and permissions
About this article
Cite this article
Kon’kova, T.V., Rysev, A.P. & Mal’kova, Y.O. Kinetics and Mechanism of Adsorption of Anionic Dyes on Montmorillonite Modified with Sodium Metasilicate. Russ. J. Phys. Chem. 95, 43–48 (2021). https://doi.org/10.1134/S003602442101012X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602442101012X